Abstract
Introduction
The present study evaluated whether frontal lobe cerebral oxygenation (ScO2), as assessed by near-infrared spectroscopy (NIRS), can detect cerebral autoregulation in patients undergoing orthotopic liver transplantation.
Methods
We studied changes in frontal lobe ScO2 assessed in 33 patients, 19 females, who underwent orthotopic liver transplantation (OLT). We evaluated whether ScO2 would remain stable over a wide range of MAP and whether an eventual drop in ScO2 could be related to a low MAP.
Results
Among the 31 of 33 patients for whom a NIRS signal could be detected, ScO2 varied in parallel with mean arterial pressure (MAP) for 3 patients and, therefore, an autoregulation curve could not be established and yet, there was detected no change in ScO2 to a lowest MAP ranging from 42 to 66 mmHg for 20 patients, while for 8 patients a decrease in ScO2 was detected at a MAP of 69 (50–90) mmHg; (median and range). As detected by NIRS, the present study confirms that some patients undergoing liver transplantation do not demonstrate cerebral autoregulation but for the majority of the patients, ScO2 was stable over a wide range of MAP suggesting that ScO2 detects cerebral autoregulation.
Conclusion
We find that NIRS is a ready available non-invasive technology for evaluation of cerebral autoregulation in patients undergoing orthotopic liver transplantation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cerebral autoregulation describes that cerebral blood flow (CBF) stays relatively stable within a mean arterial pressure (MAP) from approximately 60–150 mmHg [1] but for some patients with liver disease, there is no effective cerebral autoregulation [2, 3] and death from acute liver failure may be by a cerebral catastrophe related to cerebral hyperperfusion [2]. Also during orthotopic liver transplantation, cerebral autoregulation may be affected although the hepatectomy seems to correct the failing autoregulatory capacity of the brain circulation as detected by transcranial Doppler [4, 5] and similar observations are available with a 133Xenon clearance determined CBF [2]. During the operation, however, neither an evaluation of cerebral perfusion with transcranial Doppler nor of CBF with 133Xenon clearance are readily available as both techniques require a trained operator.
Furthermore, evaluation of CBF is a discontinuous measure and although information of CBF is important, we consider that the purpose of monitoring CBF is to secure cerebral oxygenation (ScO2). Near-infrared spectroscopy (NIRS) is applicable for routine monitoring of the brain during surgery and ScO2 reflects changes in brain capillary saturation and mitochondrial oxygen tension [6].
In the present study, changes in frontal lobe ScO2 were assessed in patients who underwent orthotopic liver transplantation (OLT). We evaluated whether ScO2 would remain stable over a wide range of MAP and whether an eventual drop in ScO2 could be related to a low MAP. Such a correlation analysis for evaluation of cerebral autoregulation has been applied, based on internal jugular venous oxygen saturation, in healthy humans [7].
Methods
ScO2 was related to MAP in 33 patients, 19 females, undergoing liver transplantation at a Child-Pugh class A (n = 4), B (n = 16), and C (n = 13) (Table 1). Anesthesia was induced with fentanyl (0.3–0.5 mg) and propofol (1.5–2.5 mg/kg) and was maintained with remifentanil (1mcg/kg/min) and propofol (25–42 mcg/kg/min) infusions and oral intubation of the trachea was facilitated with cisatracurium (0.15 mg/kg). The patients were ventilated with an oxygen/air mixture to oxygen fraction of 0.8 [8] and ventilation was adjusted to maintain an end-tidal CO2 tension (etCO2) of 3.5–4.5 kPa. To attenuate bleeding, aprotinin (Trasylol® Bayer Healthcare Pharmaceutical, Montville, NJ) infusion was administered at 20 ml/h (40 ml/h during the anhepatic phase) and to prevent spasm in the hepatic artery, infusion of epoprostenol (Flolan® GlaxoSmithKline Pharma, Brentford, UK; 6 ml/h) was added before arterial reperfusion of the liver. Blood pressure was controlled by nitroglycerine, phenylephrine, or ephedrine and plasma calcium was kept within the normal range. Central blood volume (CBV) was maintained by infusion of plasma and saline–adenine–glucose–mannitol erythrocyte suspension to maintain a hematocrit of 33%. All patients, except for one had a venovenous bypass. The median anesthesia duration was 6 h 40 min, range 4–11 h 10 min.
Arterial blood pressure was obtained from the right femoral artery and cardiac output (CO) is reported as estimated from the arterial pressure wave with the Modelflow method [9] that has been satisfactory compared to a thermodilution estimate in patients undergoing liver transplantation [10]. A pulmonary artery catheter (Swan-Ganz, 93A-831H-7.5F; Baxter Healthcare Corp., Irvine, CA.) was used for a thermodilution estimate of CO, pulmonary arterial mean pressure (PAMP), central venous pressure (CVP), and mixed venous oxygen saturation (SvO2). Heart rate (HR) and arterial oxygen saturation (SaO2) were monitored by an electrocardiogram and pulsoximetry, respectively.
Changes in the CBV were assessed by thoracic electrical admittance (TA) and expressed in Siemens (mS) (C-Guard, Danmeter, Odense, Denmark). At a low (1.5 kHz) and a high frequency (100 kHz) current, TA distinguishes between the extracellular (TA1.5) and total water (TA100) content. Accordingly, changes in the difference between TA1.5 and TA100 were considered to reflect changes in the intracellular water content (TAICW) [11], i.e., red cell volume within the thoracic region [11, 12].
The ScO2 and muscle oxygen saturation (SmO2) were monitored by NIRS (INVOS Cerebral Oximeter, Somanetics, Troy, MI.) with optodes attached to the forehead and the skin over the left biceps muscle, respectively. Monitoring of frontal lobe oxygenation by NIRS is a non-invasive alternative to recording of changes in CBF [13]. Changes in ScO2 parallel those in internal jugular venous O2 saturation and middle cerebral artery mean flow velocity [4, 14] and NIRS detects cerebral hypoperfusion during surgery [15]. The NIRS determined ScO2 is based on the absorption of light in the spectra for oxygenated and deoxygenated hemoglobin and reports ScO2 and SmO2 as a percentage of light absorption by oxygenated to total hemoglobin. An emitter generates light at 733 and 808 nm and the reflection is registered by two sensors placed at a distance of 3 and 4 cm from the emitter. This placement of the optodes allows for the subtraction of reflections derived from superficial tissues of the scalp and the skull for ScO2 [16] and subcutaneous tissue for SmO2. With increasing distance between the emitter and the optodes, light penetrates deeper into the tissues and with evaluation of absorption at two distances (spatial resolution), absorption in deep tissue, i.e., brain and skeletal muscle, is appreciated. Thus, values reported for ScO2 and SmO2 account predominantly for hemoglobin oxygenation in the frontal lobe cortex and left biceps muscle, respectively. To take into account attenuation of ScO2 by individual high levels of bilirubin in the blood [17], NIRS data are presented as changes from baseline (ΔScO2) defined as the first stable ScO2 recorded when the patient was fully monitored before surgery.
To evaluate whether ScO2 could detect cerebral autoregulation, for each patient, ScO2 was related to MAP by computer-assisted iteration of crossing between a line based on the lowest versus the highest MAP values [7]. If an insertion could be detected between the two lines, the MAP at the insertion was taken to define the lower limit of cerebral autoregulation [7]. Conversely, if no such insertion could be defined between the two lines, cerebral autoregulation was not considered to be detected by ScO2, or if there was no significant change in ScO2 over the range of manifested MAP, cerebral autoregulation was considered to be maintained to the lowest recorded level of MAP.
For presentation of the data, surgical procedures were divided into the dissection phase (from the beginning of surgery until clamps were placed on the portal and the inferior caval veins), the anhepatic phase (where the liver was bypassed and ending when flow through the inferior caval and portal veins was re-established), the early reperfusion phase (with full portal flow) followed by the late reperfusion phase (to the end of surgery). Mean values for each phase were compared by one-way ANOVA for repeated measures on ranks and Dunn’s test evaluated post-hoc comparisons. Statistical significance was set at the 95% confidence limit (P < 0.05) and data are presented as median with range.
Results
The dissection phase lasted 131 (40–312) min, the anhepatic phase 80 (30–180) min, the early reperfusion phase 22 (20–30) min, and the late reperfusion phase 125 (50–350) min. High levels of bilirubin rendered the NIRS measurements impossible in two patients who, therefore, were excluded from the study. During the four phases of surgery, no significant changes in MAP (72 (70–73) mmHg) and HR (82 (80–83) beats/min) were detected (Fig. 1). Following clamping of the portal and the inferior caval veins in the anhepatic phase of surgery, blood flow to the heart was impeded. Thus, as expected, when compared with the dissection phase, CO and TAICW decreased (from 7.9 (3.5–16.7) to 6.2 (3.1–10.2) l/min and 45 (21–76) to 43 (19–73) mS, respectively). Despite the decrease in CO and TAICW, ΔScO2 remained stable at 7 (−1–20)% vs. 8 (−9–30)%, as did ΔSmO2 at 2 (−12–22)% vs. 4 (−24–18)% and etCO2 was 3.9 (3.1–5.9) vs. 3.9 (3.0–7.4) kPa.
In the early reperfusion phase, ΔScO2 increased to 16 (−6–36)%, ΔSmO2 to 9 (−27–24)% and etCO2 to 4.3 (3.4–5.2) kPa. That was the cases as CO increased to 9.9 (4.3–18.8) l/min and TAICW returned to the level of the dissection phase (45 (22–78) mS).
ΔScO2 and ΔSmO2 tended to decrease in the late reperfusion phase, 11 (−6–25)% and 2 (−20–28)%, respectively, although etCO2 and CO remained higher than during the dissection phase (4.2 (3.2–5) kPa and 9.3 (5.1–13.2) l/min, respectively). Compared with the dissection phase at 46 (22–99) mS., the TAICW was not significantly different.
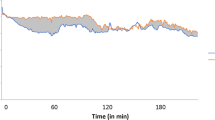
Even though MAP, on an average, remained stable during surgery, some patients demonstrated hypotensive episodes. To evaluate whether there was any individual change in ScO2 in response to a lowering of MAP, for each patient ScO2 was related to MAP. From that perspective eight patients exhibited cerebral autoregulation with a defined lower level of ~69 (range 50–90) mmHg (Table 2 and Fig. 2a). In contrast, for three patients, a positive regression between ScO2 and MAP continued for the full range of MAP recorded (Fig. 2b). No statistical significant correlation between ScO2 and MAP could be established for 20 patients who demonstrated a lowest MAP of 55 (range 42–66) mmHg (Fig. 2c).
Changes in frontal lobe cerebral oxygenation (ΔScO2) in relation to mean arterial pressure (MAP). a Cerebral autoregulation for patient # 29. The intersection between the regression and the horizontal line indicates a lower limit of cerebral autoregulation at 62 mmHg. b Impaired cerebral autoregulation for patient # 31. ΔScO2 is positively related to MAP throughout the full range of MAP. c No lower limit of cerebral autoregulation could be established for patient # 13
Discussion
On an average MAP did not change significantly during the operation while ScO2 increased in the early reperfusion phase, indicating hyperperfusion of the brain [2, 14] and also SmO2 increased. Despite the stable MAP, individual variation in ScO2 between patients was significant and 3 patients exhibited a linear relation between ScO2 and MAP, indicating that brain perfusion was not autoregulated. Conversely, a lower limit of cerebral autoregulation was defined in 8 patients while the lowest recorded MAP did not affect ScO2 for 20 patients.
During OLT, ScO2 detects changes in cerebral perfusion related to the arterial carbon dioxide tension (PaCO2) [14] and affected mental function after the operation has been related to a sudden drop in ScO2 probably because of an embolus [18]. Also the NIRS signal becomes affected by enhanced bilirubin concentration in blood likely to be elevated for the liver transplant patient, although even low values appear to respond to changes in the PaCO2 [17]. However, plasma bilirubin may be so high (>370 mM) that no NIRS signal can be detected.
The capability of NIRS to determine adequate cerebral perfusion has been shown in other studies [19–21]. From the present study, we consider the findings to indicate that NIRS detects cerebral autoregulation for most patients undergoing liver transplantation, the exception being those patients with so high a bilirubin level that a reading of ScO2 by NIRS, at least with the applied apparatus, becomes impossible as was the case for 2 of the 33 studied patients (6%). For those patients for whom a NIRS signal could be detected, two responses in ScO2 to a low MAP were detected. For the majority of the patients a low level of MAP was of no consequence for ScO2. On the other hand, for the remaining of the patients, a low MAP was associated with a reduction in ScO2 and the difference between the two groups of patients appeared, although the central blood volume was not affected, as detected with transthoracic electric admittance and SmO2 indicated no periferal vasoconstriction [13]. Under circumstances were MAP is reduced by lowering of the central blood volume by lower body negative pressure [22], head-up tilt [23, 24], and hemorrhage [13], jugular venous saturation and ScO2 are affected, while the same MAP may be of no consequence for ScO2 if CBV is maintained [25]. Yet the present results indicate that for some patients cerebral oxygenation becomes affected with even a small reduction in MAP at a maintained central blood volume.
During anesthesia MAP is often the only indirect measure to indicate whether CBF is preserved. CBF is held relatively stable by cerebral autoregulation, and only if MAP exceeds the lower limit of cerebral autoregulation CBF is considered to decrease [1]. Conversely, with a maintained central blood volume cerebral autoregulation may still be intact with a MAP of 37 mmHg as indicated by ScO2 [25]. For the present patients, MAP offered little if any indication for whether ScO2 was maintained in that there was a substantial overlap between the lowest MAP recorded for the patients with maintained ScO2 and the MAP at which a lower limit of cerebral autoregulation appeared to be detected in other patients (Fig. 3). It has furthermore been demonstrated that administration of epinephrine through its α1-adrenergic agonist vasopressure actions may produce increased perfusion pressure and flow in large vessels, but nevertheless decreases the cerebral microcirculatory blood flow [26].
Filled bars, number of patients demonstrating a lower limit of cerebral oxygenation as detected by near-infrared frontal lobe oxygenation (ScO2) during orthotopic liver transplantation plotted at the mean arterial pressure (MAP) of the limit. Open bars, number of patients for whom ScO2 was independent of MAP plotted against the lowest recorded MAP during the operation
For three patients it was not possible to determine a plateau where ScO2 remained stable. ScO2 kept rising through the full range of MAP recorded indicating that cerebral autoregulation was impaired. In contrast to the patients for whom an autoregulatory plateau could be established, not only a low MAP challenges cerebral perfusion, but also in case of a high MAP, the brain circulation will not be protected and, in turn, constitute a risk of cerebral hyperperfusion that may lead to cerebral edema [27].
It has been reported that postoperative neurological complications is seen in up to 33% of the patients undergoing OLT [28]. During surgery the ScO2 should not be allowed to decrease more than 10% from baseline as this is the level associated with brain dysfunction [13] and for (cardiac) surgery preserved ScO2 appears to reduce postoperative complications [29]. Only one patient exceeded the 10% limit drop in ScO2 and thus may have had an episode of cerebral hypoperfusion to a level where brain dysfunction would be suspected [23]. The drop in ScO2 was by 21% during reperfusion of the liver and associated with a concomitant decrease in MAP and CO, although TAICW remained stable. ScO2, MAP, and CO normalized within 20 min and ScO2 remained stable around baseline for the remaining part of the operation. Accordingly, some cardio-inhibiting factor of consequence for not only CO but also for cerebral perfusion, may have been released when normal splanchnic circulation was reestablished.
When there was a ScO2 determined cerebral autoregulation, it was possible to approximate the lower limit of cerebral autoregulation. For the eight patients we found the lower limit was at a MAP of ~69 mmHg but there was considerable variation among the patients. Variation in PaCO2 during the operation could influence the lower limit of cerebral autoregulation as hypercapnia leads to cerebral vasodilatation and hypocapnia to vasoconstriction producing, not only changes in CBF [30], but also different levels of ScO2 for the same MAP [13]. Hypercapnia in the reperfusion phase could have shifted the autoregulatory curve upwards and the lower limit towards a higher MAP because of the dilated cerebral vessels.
In conclusion, ScO2 was kept at an acceptable level during surgery even though the lower limit of cerebral autoregulation was exceeded or autoregulation was impaired for some patients. Only one patient (#31) exhibited a critical drop in ScO2 not associated with a drop in TA but with a reduction in MAP, CO, and SvO2 in the early reperfusion phase and ScO2, as well as the other parameters returned to the normal level after 20 min.
This study indicates that ScO2 detect effective cerebral autoregulation for most patients undergoing liver transplantation, but with a highly variable lower limit, i.e., ScO2 may be maintained at lowest MAP of 42 mmHg, while for another patient ScO2 decreased when MAP became lower than 90 mmHg. The implication of the present observations is that it remains unknown whether ScO2 is maintained during the operation unless it is measured when MAP is lower than 90 mmHg. We therefore recommend that regional cerebral oxygenation be monitored throughout the operation.
Abbreviations
- CBF:
-
Cerebral blood flow
- CBV:
-
Central blood volume
- CO:
-
Cardiac output
- MAP:
-
Mean arterial pressure
- NIRS:
-
Near-infrared spectroscopy
- OLT:
-
Orthotopic liver transplantation
- ScO2 :
-
Frontal lobe cerebral oxygenation
- SvO2 :
-
Mixed venous oxygen saturation
- TA:
-
Thoracic electric admittance
References
Paulson OB, Strandgaard S, Edvinsson L. Cerebral autoregulation. Cerebrovasc Brain Metab Rev. 1990;2(2):161–92.
Larsen FS, Ejlersen E, Strauss G, et al. Cerebrovascular metabolic autoregulation is impaired during liver transplantation. Transplantation. 1999;68(10):1472–6. doi:10.1097/00007890-199911270-00007.
Pere P, Hockerstedt K, Isoniemi H, Lindgren L. Cerebral blood flow and oxygenation in liver transplantation for acute or chronic hepatic disease without venovenous bypass. Liver Transpl. 2000;6(4):471–9. doi:10.1053/jlts.2000.8186.
Pott F, Larsen FS, Ejlersen E, Linkis P, Jorgensen LG, Secher NH. Cerebral perfusion during human liver transplantation. Clin Physiol. 1995;15(2):119–30. doi:10.1111/j.1475-097X.1995.tb00436.x.
Ardizzone G, Arrigo A, Panaro F, et al. Cerebral hemodynamic and metabolic changes in patients with fulminant hepatic failure during liver transplantation. Transplant Proc. 2004;36(10):3060–4. doi:10.1016/j.transproceed.2004.11.014.
Rasmussen P, Dawson EA, Nybo L, van Lieshout JJ, Secher NH, Gjedde A. Capillary-oxygenation-level-dependent near-infrared spectrometry in frontal lobe of humans. J Cereb Blood Flow Metab. 2007;27(5):1082–93.
Schmidt JF, Waldemar G, Vorstrup S, Andersen AR, Gjerris F, Paulson OB. Computerized analysis of cerebral blood flow autoregulation in humans: validation of a method for pharmacologic studies. J Cardiovasc Pharmacol. 1990;15(6):983–8. doi:10.1097/00005344-199006000-00017.
Greif R, Akca O, Horn EP, Kurz A, Sessler DI. Supplemental perioperative oxygen to reduce the incidence of surgical-wound infection. Outcomes research group. N Engl J Med. 2000;342(3):161–7. doi:10.1056/NEJM200001203420303.
Wesseling KH, Jansen JR, Settels JJ, Schreuder JJ. Computation of aortic flow from pressure in humans using a nonlinear, three-element model. J Appl Physiol. 1993;74(5):2566–73.
Nissen P, van Lieshout JJ, Novovic S, Bundgaard-Nielsen M, Secher NH. Techniques of cardiac output measurement during liver transplantation: arterial pulse wave versus thermodilution. Liver Transpl. 2009;15(3):287–91. doi:10.1002/lt.21689.
Cai Y, Holm S, Jenstrup M, et al. Electrical admittance for filling of the heart during lower body negative pressure in humans. J Appl Physiol. 2000;89(4):1569–76.
Cai Y, Boesen M, Stromstad M, Secher NH. An electrical admittance based index of thoracic intracellular water during head-up tilt in humans. Eur J Appl Physiol. 2000;83(4–5):356–62.
Madsen PL, Secher NH. Near-infrared oximetry of the brain. Prog Neurobiol. 1999;58(6):541–60. doi:10.1016/S0301-0082(98)00093-8.
Skak C, Rasmussen A, Kirkegaard P, Secher NH. Cerebral oxygen saturation and blood flow during liver transplantation. Anesth Analg. 1997;84(4):730–3. doi:10.1097/00000539-199704000-00005.
Plachky J, Hofer S, Volkmann M, Martin E, Bardenheuer HJ, Weigand MA. Regional cerebral oxygen saturation is a sensitive marker of cerebral hypoperfusion during orthotopic liver transplantation. Anesth Analg. 2004;99(2):344–9. doi:10.1213/01.ANE.0000124032.31843.61.
Grubhofer G, Lassnigg A, Manlik F, Marx E, Trubel W, Hiesmayr M. The contribution of extracranial blood oxygenation on near-infrared spectroscopy during carotid thrombendarterectomy. Anaesthesia. 1997;52(2):116–20. doi:10.1111/j.1365-2044.1997.20-az020.x.
Madsen PL, Skak C, Rasmussen A, Secher NH. Interference of cerebral near-infrared oximetry in patients with icterus. Anesth Analg. 2000;90(2):489–93. doi:10.1097/00000539-200002000-00046.
Madsen PL, Secher NH. Postoperative confusion preceded by decreased frontal lobe haemoglobin oxygen saturation. Anaesth Intensive Care. 2000;28(3):308–10.
Bein B, Meybohm P, Cavus E, et al. A comparison of transcranial Doppler with near infrared spectroscopy and indocyanine green during hemorrhagic shock: a prospective experimental study. Crit Care. 2006;10(1):R18. doi:10.1186/cc3980.
Steiner LA, Pfister D, Strebel SP, Radolovich D, Smielewski P, Czosnyka M. Near-infrared spectroscopy can monitor dynamic cerebral autoregulation in adults. Neurocrit Care. 2009;10(1):122–8. doi:10.1007/s12028-008-9140-5.
Vernieri F, Tibuzzi F, Pasqualetti P, et al. Transcranial Doppler and near-infrared spectroscopy can evaluate the hemodynamic effect of carotid artery occlusion. Stroke. 2004;35(1):64–70. doi:10.1161/01.STR.0000106486.26626.E2.
Giller CA, Levine BD, Meyer Y, Buckey JC, Lane LD, Borchers DJ. The cerebral hemodynamics of normotensive hypovolemia during lower-body negative pressure. J Neurosurg. 1992;76(6):961–6.
Madsen P, Lyck F, Pedersen M, Olesen HL, Nielsen H, Secher NH. Brain and muscle oxygen saturation during head-up-tilt-induced central hypovolaemia in humans. Clin Physiol. 1995;15(5):523–33. doi:10.1111/j.1475-097X.1995.tb00541.x.
Jorgensen LG, Perko M, Perko G, Secher NH. Middle cerebral artery velocity during head-up tilt induced hypovolaemic shock in humans. Clin Physiol. 1993;13(4):323–36. doi:10.1111/j.1475-097X.1993.tb00333.x.
Nissen P, Nielsen HB, van Lieshout JJ, Secher NH. Frontal lobe oxygenation is maintained during hypotension following propofol-phentanyl anesthesia. AANA J. 2009 (in press).
Ristagno G, Tang W, Huang L, et al. Epinephrine reduces cerebral perfusion during cardiopulmonary resuscitation. Crit Care Med. 2009;37(4):1408–15.
Larsen FS. Cerebral circulation in liver failure: Ohm’s law in force. Semin Liver Dis. 1996;16(3):281–92. doi:10.1055/s-2007-1007241.
Adams DH, Ponsford S, Gunson B, et al. Neurological complications following liver transplantation. Lancet. 1987;1(8539):949–51. doi:10.1016/S0140-6736(87)90294-7.
Murkin JM, Adams SJ, Novick RJ, et al. Monitoring brain oxygen saturation during coronary bypass surgery: a randomized, prospective study. Anesth Analg. 2007;104(1):51–8. doi:10.1213/01.ane.0000246814.29362.f4.
Jalan R, Olde Damink SW, Deutz NE, Hayes PC, Lee A. Restoration of cerebral blood flow autoregulation and reactivity to carbon dioxide in acute liver failure by moderate hypothermia. Hepatology. 2001;34(1):50–4. doi:10.1053/jhep.2001.25386.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nissen, P., Pacino, H., Frederiksen, H.J. et al. Near-Infrared Spectroscopy for Evaluation of Cerebral Autoregulation During Orthotopic Liver Transplantation. Neurocrit Care 11, 235–241 (2009). https://doi.org/10.1007/s12028-009-9226-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-009-9226-8