Abstract
Different immune cell subsets have a relevant role in the pathogenesis of and tissue damage seen in autoimmune thyroid diseases (AITD), including T regulatory (Treg) lymphocytes and T helper (Th) 17 cells. There are several types of CD4+ Treg cells (Foxp3+, CD69+, Tr1), which are able to prevent the appearance of autoimmune diseases, down regulating the immune response and the inflammatory phenomenon. However, despite their presence in peripheral blood and thyroid tissue from patients with AITD, these cells are apparently unable to put down the autoimmune process. Moreover, many reports indicate the involvement of Th17 cells in chronic inflammatory diseases, including AITD. Nevertheless, it is now evident that these lymphocytes show a remarkable plasticity, giving rise to anti-inflammatory (including Treg lymphocytes) and pro-inflammatory cell subtypes. Nowadays, both Treg and Th17 cells must be considered as key elements in the pathogenesis of AITD as well as plausible potential targets for the next generation of therapeutic options of this condition.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It has been widely described that autoimmune thyroid diseases (AITD, mainly Hashimoto’s thyroiditis or HT, and Graves’ disease or GD) appear as consequence of loss of immune tolerance toward organ-specific self-antigens, including thyroglobulin (TG), thyroperoxidase (TPO), and the receptor for TSH [1]. It is also well known that HT is mainly mediated by a cellular autoimmune response, with a heavy inflammatory cell infiltrate, subsequent tissue destruction, and thyroid gland failure [2, 3]. Moreover, GD is chiefly mediated by a humoral autoimmune response, with the synthesis of agonistic auto-antibodies specific for TSH receptor [2]. However, in HT, an evident humoral autoimmune response is also observed (anti-TG and anti-TPO Abs), and in GD, a variable degree of T cell activation is observed, with variable levels of thyroid cell infiltration [2, 3]. Accordingly, in Graves’ ophthalmopathy (GO), the most frequent extrathyroidal manifestation of GD, both, the humoral and cellular immune responses seem to be involved in its pathogenesis [4]. Orbital disease is detectable on clinical grounds in approximately 25 % of Graves’ patients and is most commonly mild. Moderate-to-severe forms account for 5 % of cases [5].
T helper CD4+ lymphocytes are generated in the thymus, emerging to the peripheral lymphoid tissues as mature naïve T cells. Although the majority of auto-reactive T lymphocytes are deleted in the thymus, it is evident that the presence of T cells recognizes self-antigens in the peripheral blood and other tissues from healthy individuals. The activation and proliferation of these auto-reactive cells are prevented (immune tolerance) by different immune regulatory mechanisms, including the suppressive effect of T regulatory (Treg) lymphocytes. However, in certain individuals, the auto-reactive T cells escape from the control of the immune regulatory mechanisms, with their subsequent activation, proliferation, and differentiation (autoimmune response). It has been widely described that the differentiation of CD4+ T cells results in the preferential generation of distinct Th cell subsets, including Th1, Th2, and Th17 lymphocytes [6]. It has been also reported the apparent role of both Treg and Th17 lymphocytes in the pathogenesis of AITD, and significant advances in knowledge of these cells have occurred in the recent years. Thus, nowadays, both Treg and Th17 cells are considered as plausible potential targets for the modern therapy of autoimmune and chronic inflammatory conditions [7, 8].
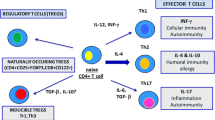
T regulatory (Treg) cells
Although the presence of T cells with immunosuppressive activity was initially reported by Gershon and Kondo more than four decades ago [9], the first proper characterization of a Treg cell subset was made by Sakaguchi, et al. 25 years latter [10]. These lymphocytes were characterized as CD4+ cells expressing the transcription factor Foxp3 and high constitutive levels of the α chain of interleukin (IL)-2 receptor (CD25high or CD25bright). As shown in Fig. 1, these cells were initially denominated as natural Treg (nTreg) lymphocytes, since they emerge from the thymus as fully differentiated cells. The majority of nTreg cells recognize self-antigens, but in contrast with effector or conventional T cells, they show a limited capacity of cell proliferation when their antigen receptor (TCR) is engaged. In addition, nTreg cells show a prominent capability to inhibit the activation, proliferation, and cytokine synthesis by conventional or effector lymphocytes [11]. Since it is now evident that regulatory cells with an identical phenotype of nTreg cells may be generated both in vivo, outside the thymus, and in vitro, nTreg lymphocytes have been renamed as tTreg (thymus-derived Treg), whereas the other two subsets are called as pTreg (peripheral differentiated Treg) and iTreg (in vitro generated Treg) lymphocytes [12]. In all these cases, Treg cells exert their immunosuppressive effect through different mechanisms, including the synthesis of anti-inflammatory cytokines (transforming growth factor-β or TGF-β, IL-10, and IL-35) [13].
Differentiation of the main CD4+ Treg cell subsets. In the thymus and under the combined effect of IL-2 and TGF-β, a subset of CD4+ immature T cells is differentiated into Foxp3+ Treg cells. In the peripheral lymphoid tissues, these cells are converted into memory (CD45RO+) cells by the combined effect of cytokines and antigen recognition (TCR engagement). These lymphocytes are called as thymus or natural Treg (tTreg and nTreg, respectively) lymphocytes, since they emerge from thymus as fully differentiated cells. In contrast, CD4+ Foxp3- conventional naïve cells can be induced to differentiate, in the periphery or in vitro, into Foxp3+ Treg lymphocytes (pTreg and iTreg, respectively), by the combined action of IL-2, TGF-β, and TCR engagement. As indicated, tTreg, pTreg, and iTreg cells exhibit the same phenotype and function. Moreover, through a process that remains to be characterized, another regulatory CD4+ cell subset emerges fully differentiated from the thymus, which is characterized by the constitutive expression of CD69 and the absence of Foxp3 (Treg CD69+). These cells are converted into memory lymphocytes upon antigen recognition and, very likely, IL-2 effect. Finally, when CD4+ conventional naïve cells are exposed in the periphery to IL-10 during TCR engagement, they are differentiated into a regulatory cell subset characterized by the absence of Foxp3, the synthesis of IL-10, and the expression of the LAG-3 and CD49b markers (Type 1 regulatory or Tr1 cells)
Another CD4+ regulatory cell subset that apparently emerges from the thymus fully differentiated corresponds to the CD69+ Treg cells (Fig. 1). These lymphocytes show a constitutive expression of CD69, do not express Foxp3, and mainly exert their suppressive activity through the synthesis of IL-10 and TGF-β [14, 15]. Finally, the type 1 regulatory (Tr1) cells have been mainly characterized by Roncarolo et al. as CD4+ Foxp3− lymphocytes that synthesize IL-10 and that are differentiated under the influence of this cytokine (Fig. 1) [16]. Many different data indicate that all these Treg cell types, mainly those expressing Foxp3, have a relevant role in the prevention and pathogenesis of autoimmune diseases, including AITD [16, 17] (Table 1).
Treg cells and autoimmune thyroiditis
Since the earliest reports on the physiological role of Treg cells, it was evident that the absence of these cells was tightly associated with thyroid autoimmunity [1–3, 17, 18]. This point was later confirmed by the presence of autoimmune thyroiditis (in addition to other clinical features) in mice or humans that do not express Foxp3 (IPEX syndrome) and that lack of tTreg and i/pTreg lymphocytes [19, 20]. Accordingly, in subsequent studies in patients with AITD and in animal models of thyroiditis, it was expected to detect a diminished number and/or a defective function of Treg cells. In fact, several reports confirmed a defective function of Foxp3+ Treg cells in patients with HT or GD [21–26]. Likewise, we observed that the immunosuppressive function of CD69+ Treg cells is also defective in the majority of patients with AITD [27]. In contrast, the levels of CD4+ Treg cells are variable in the peripheral blood from patients with AITD, whereas a prominent accumulation of these lymphocytes in the inflamed thyroid tissue has been observed [17, 23, 26, 27]. However, although there is an active recruitment of Treg cells into the inflammatory cell infiltrate of AITD, these lymphocytes are dysfunctional and unable to effectively down regulate the ongoing autoimmune process and inflammatory phenomenon [21–27]. Likewise, although the precise cause(s) of Treg dysfunction in AITD (as well as in other autoimmune diseases) remains to be determined [7, 11, 12], it is of interest the possible transition of these lymphocytes toward pro-inflammatory cells (mainly Th17 and Th1 lymphocytes) [12, 28–30], a phenomenon that may further contribute to the perpetuation of the autoimmune process. In this regard, it is of interest the in vitro transition of CD4+ Foxp3+ Treg cells into IL-17-producing lymphocytes when are exposed to different cytokines, mainly IL-2/IL-15 and IL-21/IL-23 [31, 32]. In addition, different data show the increased frequency of Foxp3+/IL-17+ or Foxp3+/IFN-γ+ lymphocytes, as well as the enhanced differentiation of Treg cells into Treg/Th17 or Treg/Th1 lymphocytes in patients with psoriasis and type 1 diabetes, respectively [33, 34]. This interesting point has been recently reviewed by Pandiyan, et al. and Du, et al. [35, 36]. However, the extent of Treg cell conversion into Th17 and Th1 lymphocytes in AITD remains as an interesting point to be disclosed. Likewise, the levels and function of Tr1 cells are another relevant issue to be addressed in patients with AITD [16]. Finally, the possible role of other regulatory cells in the pathogenesis of AITD is an additional point of interest. In this regard, it has been recently reported the involvement of B regulatory lymphocytes in a mice model of autoimmune thyroiditis induced by iodine [37].
Th17 cells
Since the original description of CD4+ T helper cell subsets, based on their profile of cytokine synthesis and called as Th1 and Th2, almost three decades ago [38], additional subtypes have been characterized [39]. Thus, Th17 cells were originally described in 2003 [40], whereas IL-17 was identified in 1993 [41, 42]. Nowadays, a very significant advance in the knowledge of Th cell subsets, their differentiation pathways, and their role in the defense against pathogens and involvement in the pathogenesis of different conditions have occurred [39]. Thus, it is well known that upon antigen recognition by CD4+ T naïve cells, the presence of IL-12, IFN-γ, and IL-2 (and the expression of the transcription factor T-bet) induces their differentiation into Th1 cells (which mainly synthesize IFN-γ and IL-2), whereas the presence of IL-4 inhibits Th1 differentiation and favors the generation of Th2 lymphocytes, which mainly synthesize IL-4, IL-5, IL-6, and IL-13 (Fig. 2). The latter process requires the expression of the transcription factor GATA-3. Moreover, although early contradictory data on the factors that induce the differentiation of Th17 cells in humans were reported, it is now evident that this process is mainly induced by the combined action of IL-1β, IL-6, and IL-23, and the expression of the transcription factor RORC2 with the activation of the STAT3 intracellular pathway (Fig. 2) [43]. The conventional or classical Th17 cells are mainly characterized by the synthesis of IL-17A, and IL-17F, which may act on epithelial cells, fibroblasts, or macrophages, inducing the release of pro-inflammatory mediators (e.g., chemokines, TNF-α, and IL-1β) [43]. In this regard, the pathogenic role of Th17 lymphocytes was initially demonstrated in 2005, through the passive transfer of IL-17-producing cells induced by the proteolipid protein (PLP) peptide and IL-23 in mice; in these experiments, Th17 but not Th1 cells were able to mediate experimental allergic encephalomyelitis [44]. However, it is now evident that classical or conventional Th17 cells require a further differentiation to exert a pathogenic role under different scenarios (e.g., multiple sclerosis, rheumatoid arthritis, or inflammatory bowel disease) [43–48]. Thus, the sustained exposure of classical Th17 lymphocytes to IL-23 results in their conversion into pro-inflammatory or pathogenic cells, acquiring the capability to synthesize IFN-γ and GM-CSF (Fig. 2) [43–48]. On the other hand, the continuous effect of TGF-β on classical Th17 cells induces their conversion into non-pathogenic lymphocytes, involved in the defense against different extracellular bacteria and fungi and tissue repair [43–48]. Interestingly, these subtypes of Th17 cells can be easily identified by multi-parametric flow cytometry, through the detection of different markers, mainly CXCR3, CD161, MDR1/CD243, and obviously, IFN-γ and IL-17 [48, 49]. The plasticity of Th17 cells is further evidenced by the ability of pathogenic Th17 lymphocytes (which synthesize IL-17 and IFN-γ) to differentiate into a Th1-like (non-classic Th1) cell, which synthesize IFN-γ and GM-CSF (but not IL-17) and that have a very relevant role in the pathogenesis of the tissue damage seen in autoimmune inflammatory conditions [43–50]. Finally, it has been also described that non-pathogenic Th17 cells may evolve into Foxp3+ Treg cells, which may further antagonize the activity of pathogenic Th17 lymphocytes [12, 30]. Interestingly, as stated above, the opposite differentiation pathway (Treg to Th17) as well as an apparently stable mixed phenotype (Th17/Treg) has been also documented [12, 30, 33–36, 51]. All these data indicate the great plasticity of Th17 cells and their complex role in the immune system, in health and disease (Table 2).
T helper cell subsets and Th17 lymphocyte plasticity. Resting T naïve CD4+ lymphocytes localized in the periphery (e.g., lymph nodes) are activated when antigenic peptides are recognized on the surface of antigen presenting cells (APC); these recently activated lymphocytes may differentiate into several subsets, characterized by their profiles of cytokine synthesis. In the presence of IL-12 (mainly derived from APC) and IFN-γ (derived from innate lymphoid cells), Th0 differentiates into classic Th1 cells, which mostly produce IL-2 and IFN-γ. When Th0 lymphocytes are exposed to IL-4 during their interaction with APC, they are differentiated into Th2 cells, which mainly synthesize cytokines involved in the generation of the humoral immune response (IL-4, IL-5, IL-6, IL-13). In the presence IL-1β, IL-6, and IL-23, Th0 lymphocytes give rise to Th17 lymphocytes, characterized by the expression of the RORC2 transcription factor and the production of IL-17A, IL-17F, and IL-21. The sustained effect of IL-23 (synthesized by APC and macrophages) on Th17 cells induces their conversion into pro-inflammatory pathogenic lymphocytes, which are able to release IL-17A, IFN-γ, and GM-CSF; When these pathogenic cells remain in an inflammatory milieu, under high concentrations of pro-inflammatory cytokines (IL-12, TNF-α, IL-1β), they evolve into non-classic Th1 lymphocytes, which synthesize IFN-γ and GM-CSF, but not IL-17. Moreover, the sustained effect of TGF-β on Th17 lymphocytes induces their transition toward non-pathogenic cells, characterized by the synthesis of IL-17A and IL-10, which may evolve into induced (i) or peripheral (p) Treg cells. Finally, other cytokines or cytokine combinations may induce the differentiation of Th0 lymphocytes into additional Th cell subsets (Th5, Th6, Th9, T follicular helper cells, or Tfh). The specific transcription factors necessary for the differentiation of Th1, Th2, Th17, and Th22 cells are indicated (T-bet, GATA-3, RORC2, and AhR, respectively) as well as the main cytokines involved in these processes are indicated in red. The main cytokines synthesized by each cell subset appear in blue squares
As shown in Fig. 2, Th naïve cells may differentiate into other lymphocyte subsets, including Th3 (regulatory cells, through the release of TGF-β), Th9 (involved in allergy and synthesizing IL-9 and IL-10), Tfh (which provide help to B lymphocytes via IL-4 and IL-21, among others), and Th22. The latter cells mainly synthesize IL-22 (and TNF-α, but not IL-17) and have a relevant role in the pathogenesis of psoriasis and, likely, other autoimmune conditions [52]. IL-22 is also synthesized, at variable degrees, by the different subtypes of Th17 cells.
Th17 cells and autoimmune thyroiditis
Since the original description of Th17 cells (in 2003) and their pro-inflammatory activity [40, 43] it was evident their involvement in the pathogenesis of different autoimmune diseases. In regard of AITD, early reports on the possible role of Th17 cells (in 2009) indicated that they are necessary for the induction of an animal model of autoimmune thyroiditis [53] and that patients with GD unresponsive to conventional therapy show increased levels of IL-17+ T cells [54, 55]. We subsequently detected that AITD patients have increased levels of Th17 cells in both peripheral blood and thyroid tissue as well as an enhanced in vitro differentiation of their T cells into Th17 lymphocytes, induced by IL-6/IL-23 [56]. In an additional study, Qin et al. observed increased expression of IL-17 gene in patients with HT [57]. Further studies confirmed the increased levels of IL-17 and/or Th17 cells in the peripheral blood and/or thyroid tissue from patients with HT or GD [57–59]. In addition, Kristensen et al. showed that the in vitro stimulation of T cells from patients with HT with TPO induces a higher differentiation of Th17 lymphocytes compared to healthy controls [60]. Additional studies corroborated the increased levels of IL-17+ T cells with a diminished Treg/Th17 cell ratio in HT patients [61, 62]. Moreover, the possible role of different immune regulatory molecules involved in the induction (e.g., leptin, with a direct effect on T naïve cells, and GITRL, inhibiting Treg cell function) [63, 64] or inhibition (e.g., galectin-9) [65] of Th17 differentiation has been also reported. Finally, the possible functional expression of the IL-17A receptor by thyrocytes has been also documented [66].
Other cytokines and cell subsets related to Th17 lymphocytes have been studied in AITD. Thus, increased plasma levels of IL-21, IL-22, and IL-23 have been reported in AITD, mainly in HT [56, 61, 67–71]. In addition, increased levels and differentiation of Th22 cells have been observed in patients with HT or GD or both [61, 67, 68].
In the case of GO, there are also evidences on the participation of Th17 cells and their cytokines in the pathogenesis of the inflammatory process of the orbital connective tissue and extraocular muscles seen in this condition [4, 72–74]. Accordingly, increased serum levels of IL-17 have been detected in these patients [73, 74]. In addition, it has been described a very significant association between different single nucleotide polymorphisms of the IL-23R gene and GO [75]. However, the presence of Th17 cells and their characteristics into the orbital tissue have not studied yet.
Overall, these data strongly support that Th17 cells have a very relevant role in the pathogenesis of the chronic inflammatory phenomenon and tissue damage seen in AITD [56, 76]. However, there are several key issues to be solved, including the precise phenotype of the IL-17+ cells detected in the peripheral blood and thyroid gland from patients with AITD. Although it is very likely that these cells correspond to pathogenic or pro-inflammatory Th17 lymphocytes (CD4+CXCR3+CD161+MDR1/CD243+ in addition to IFN-γ+IL-17+ and IL-10−), with few non-pathogenic cells (CD4+CXCR3+CD161−MDR1/CD243− as well as IL-17+ and IL-10+) [47, 48], this remains as an interesting point to be studied. Moreover, the possible effect of thyroid hormones on Th17 and Treg cells is another issue of potential interest.
Treg and Th17 cells as potential therapeutic targets in AITD
Although the current therapy of hyper of hypothyroidism associated with AITD (including anti-thyroid drugs, radioiodine ablation, and levothyroxine substitutive therapy) is reasonably effective in most cases, it is evident that an additional approach could significantly improve the treatment of patients with HT, GD, and GO. In this regard, although it is current practice the use of corticosteroid or radiotherapy in GO, our current knowledge on its pathogenesis [4, 72] along with the novel biological agents including mAbs against cell surface antigens as CD3 or CD20 or anti-cytokine or cytokine receptors such as anti-TNF or anti-IL6R, and blockers of intracellular signaling pathways (mainly kinase inhibitors), currently available and in development, increases the possibility of using selective immunosuppressive agents for this condition [77]. In addition, for the therapy of AITD, the antagonists of Th17 cells (e.g., anti-IL-17, anti-IL-23 or anti-IL-6R mAb, STAT3 inhibitors, chemokine blockers) along with the administration of in vitro generated autologous Treg cells (or their in vivo induction) remain as an interesting possibility to be tested in the near future.
Abbreviations
- Abs:
-
Antibodies
- mAb:
-
Monoclonal antibodies
- Tfh:
-
T follicular helper
- Treg:
-
T regulatory
- Th:
-
T helper
- AITD:
-
Autoimmune thyroid diseases
- IL-:
-
Interleukin
- IFN:
-
Interferon
- TG:
-
Thyroglobulin
- TPO:
-
Thyroperoxidase
- TGF-β:
-
Transforming growth factor-β
- GO:
-
Graves’ ophthalmopathy
References
A. Antonelli, S.M. Ferrari, A. Corrado, A. Di Domenicantonio, P. Fallahi, Autoimmune thyroid disorders. Autoimmun. Rev. 14, 174–180 (2015)
S.A. Morshed, R. Latif, T.F. Davies, Delineating the autoimmune mechanisms in Graves’ disease. Immunol. Res. 54, 191–203 (2012)
H. Li, T. Wang, The autoimmunity in Graves’ disease. Front. Biosci. 18, 782–787 (2013)
Y. Wang, T.J. Smith, Current concepts in the molecular pathogenesis of thyroid-associated ophthalmopathy. Invest. Ophthalmol. Vis. Sci. 55, 1735–1748 (2014)
L. Bartalena, V. Fatourechi, Extrathyroidal manifestations of Graves’ disease: a 2014 update. J. Endocrinol. Invest. 37, 691–700 (2014)
L.A. Zúñiga, R. Jain, C. Haines, D.J. Cua, Th17 cell development: from the cradle to the grave. Immunol. Rev. 252, 78–88 (2013)
C.R. Grant, R. Liberla, G. Mieli-Vergani, D. Vergani, M.S. Longhi, Regulatory T-cells in autoinmune diseases: challenges, controversies and-yet-unanswered questions. Autoimmun. Rev. 14, 105–116 (2015)
A.L. Croxford, P. Kulig, B. Becher, IL-12 and IL-23 in health and disease. Cytokine Growth Factor Rev. 25, 415–421 (2014)
R.K. Gershon, K. Kondo, Cell interactions in the induction of tolerance: the role of thymic lymphocytes. Immunology 18, 723–737 (1970)
S. Hori, T. Takahashi, S. Sakaguchi, Control of autoimmunity by naturally arising regulatory CD4+ T cells. Adv. Immunol. 81, 331–371 (2003)
M. Miyara, Y. Ito, S. Sakaguchi, Treg-cell therapies for autoimmune rheumatic diseases. Nat. Rev. Rheumatol. 10, 543–551 (2014)
S. Sakaguchi, D.A. Vignali, A.Y. Rudensky, R.E. Niec, H. Waldmann, The plasticity and stability of regulatory T cells. Nat. Rev. Immunol. 13, 461–467 (2013)
S. Sakaguchi, K. Wing, Y. Onishi, P. Prieto-Martin, T. Yamaguchi, Regulatory T cells: how do they suppress immune responses? Int. Immunol. 21, 1105–1111 (2009)
Y. Han, Q. Guo, M. Zhang, Z. Chen, X. Cao, CD69+ CD4+ CD25− T cells, a new subset of regulatory T cells, suppress T cell proliferation through membrane-bound TGF-β1. J. Immunol. 182, 111–120 (2009)
M. Vitales-Noyola, L. Doníz-Padilla, C. Álvarez-Quiroga, A. Monsiváis-Urenda, H. Portillo-Salazar, R. González-Amaro, Quantitative and functional analysis of CD69(+) NKG2D(+) T regulatory cells in healthy subjects. Hum. Immunol. 76, 511–518 (2015)
M.G. Roncarolo, S. Gregori, R. Bacchetta, M. Battaglia, Tr1 cells and the counter-regulation of immunity: natural mechanisms and therapeutic applications. Curr. Top. Microbiol. Immunol. 380, 39–68 (2014)
M. Marazuela, M.A. García-López, N. Figueroa-Vega, H. de la Fuente, B. Alvarado-Sánchez, A. Monsiváis-Urenda, F. Sánchez-Madrid, R. González-Amaro, Regulatory T cells in human autoimmune thyroid disease. J. Clin. Endocrinol. Metab. 91, 3639–3646 (2006)
P. Verginis, H.S. Li, G. Carayanniotis, Tolerogenic semimature dendritic cells suppress experimental autoimmune thyroiditis by activation of thyroglobulin-specific CD4+ CD25+ T cells. J. Immunol. 174, 7433–7439 (2005)
E. Gambineri, T.R. Torgerson, H.D. Ochs, Immune dysregulation, polyendocrinopathy, enteropathy, and X-linked inheritance (IPEX), a syndrome of systemic autoimmunity caused by mutations of Foxp3, a critical regulator of T-cell homeostasis. Curr. Opin. Rheumatol. 15, 430–435 (2003)
D.J. Kasprowicz, P.S. Smallwood, A.J. Tyznik, S.F. Ziegler, Scurfin (FoxP3) controls T-dependent immune responses in vivo through regulation of CD4+ T cell effector function. J. Immunol. 171, 1216–1223 (2003)
C. Mao, S. Wang, Y. Xiao, J. Xu, Q. Jiang, M. Jin, X. Jiang, H. Guo, G. Ning, Y. Zhang, Impairment of regulatory capacity of CD4+ CD25+ regulatory T cells mediated by dendritic cell polarization and hyperthyroidism in Graves´ disease. J. Immunol. 186, 4734–4743 (2011)
M. Nakahara, Y. Nagayama, T. Ichikawa, L. Yu, G.S. Eisenbarth, N. Abiru, The effect of regulatory T-cell depletion on the spectrum of organ-specific autoimmune diseases in nonobese diabetic mice at different ages. Autoimmunity 44, 504–510 (2011)
D. Pan, Y.H. Shin, G. Gopalakrishnan, J. Hennessey, L.J. De Groot, Regulatory T cells in Graves’ disease. Clin. Endocrinol. 71, 587–593 (2009)
P. Verginis, H.S. Li, G. Carayanniotis, Tolerogenic semimature dendritic cells suppress experimental autoimmune thyroiditis by activation of thyroglobulin-specific CD4+ CD25+ T cells. J. Immunol. 174, 7433–7439 (2005)
A.B. Glick, A. Wodzinski, P. Fu, A.D. Levine, D.N. Wald, Impairment of regulatory T-cell function in autoimmune thyroid disease. Thyroid 23, 871–878 (2013)
A. Bossowski, M. Moniuszko, M. Dabrowska, B. Sawicka, M. Rusak, M. Jeznach, J. Wójtowicz, A. Bodzenta-Lukaszyk, A. Bossowska, Lower proportions of CD4+ CD25high and CD4+ Foxp3+, but not CD4+ CD25+ CD127lowFoxp3+ T cell levels in children with autoimmune thyroid diseases. Autoimmune 46, 222–230 (2013)
A. Rodríguez-Muñoz, M. Vitales-Noyola, A. Ramos-Levi, A. Serrano-Somavilla, R. González-Amaro, M. Marazuela, Levels of regulatory T cells CD69+ NKG2D+ IL-10+ are increased in patients with autoimmune thyroid disorders. Endocrine (2015). doi:10.1007/s12020-015-0662-2
R. Basu, R.D. Hatton, C.T. Weaver, The Th17 family: flexibility follows function. Immunol. Rev. 252, 89–203 (2013)
F. Annunziato, L. Cosmi, F. Liotta, E. Maggi, S. Romagnani, Human T helper type 1 dichotomy: origin, phenotype and biological activities. Immunology 144, 343–351 (2014)
Y.K. Lee, R. Mukasa, R.D. Hatton, C.T. Weaver, Developmental plasticity of Th17 and Treg cells. Curr. Opin. Immunol. 21, 274–280 (2009)
G. Beriou, C.M. Costantino, C.W. Ashley, L. Yang, V.K. Kuchroo, C. Baecher-Allan, D.A. Hafler, IL-17-producing human peripheral regulatory T cells retain suppressive function. Blood 113, 4240–4249 (2009)
H.J. Koenen, R.L. Smeets, P.M. Vink, E. van Rijssen, A.M. Boots, I. Joosten, Human CD25highFoxp3pos regulatory T cells differentiate into IL-17-producing cells. Blood 112, 2340–2352 (2008)
H.J. Bovenschen, P.C. van de Kerkhof, P.E. van Erp, R. Woestenenk, I. Joosten, H.J. Koenen, Foxp3+ regulatory T cells of psoriasis patients easily differentiate into IL-17A-producing cells and are found in lesional skin. J. Invest. Dermatol. 131, 1853–1860 (2011)
S.A. McClymont, A.L. Putnam, M.R. Lee, J.H. Esensten, W. Liu, M.A. Hulme, U. Hoffmüller, U. Baron, S. Olek, J.A. Bluestone, T.M. Brusko, Plasticity of human regulatory T cells in healthy subjects and patients with type 1 diabetes. J. Immunol. 186, 3918–3926 (2011)
P. Pandiyan, J. Zhu, Origin and functions of pro-inflammatory cytokine producing Foxp3+ regulatory T cells. Cytokine (2015). doi:10.1016/j.cyto.2015.07.005
R. Du, H. Zhao, F. Yan, H. Li, IL-17+ Foxp3+ T cells: an intermediate differentiation stage between Th17 cells and regulatory T cells. J. Leukoc. Biol. 96, 39–48 (2014)
L. Shi, M. Bi, R. Yang, J. Zhou, S. Zhao, C. Fan, Z. Shan, Y. Li, W. Teng, Defective expression of regulatory B cells in iodine-induced autoimmune thyroiditis in non-obese diabetic H-2(h4) mice. J. Endocrinol. Invest. 37, 43–50 (2014)
T.R. Mosmann, H. Cherwinski, M.W. Bond, M.A. Giedlin, R.L. Coffman, Two types of murine helper T cell clone. I. Definition according to profiles of lymphokines activities and secreted proteins. J. Immunol. 136, 2348–2357 (1986)
N. Schmitt, H. Ueno, Regulation of human helper T cell subset differentiation by cytokines. Curr. Opin. Immunol. 34, 130–136 (2015)
S. Aggarwal, N. Ghilardi, M.H. Xie, F.J. de Sauvage, A.L. Gurney, Interleukin-23 promotes a distinct CD4 T cell activation state characterized by the production of interleukin-17. J. Biol. Chem. 278, 1910–1914 (2003)
E. Rouvier, M.F. Luciani, M.G. Mattei, F. Denizot, P. Golstein, CTLA-8, cloned from an activated T cell, bearing AU-rich messenger RNA instability sequences, and homologous to a herpesvirus saimiri gene. J. Immunol. 150, 5445–5456 (1993)
Z. Yao, S.L. Painter, W.C. Fanslow, D. Ulrich, B.M. Macduff, M.K. Spriggs, R.J. Armitage, Human IL-17: a novel cytokine derived from T cells. J. Immunol. 155, 5483–5486 (1995)
X. Song, H. Gao, Y. Qian, Th17 differentiation and their pro-inflammation function. Adv. Exp. Med. Biol. 841, 99–151 (2014)
C.L. Langrish, Y. Chen, W.M. Blumenschein, J. Mattson, B. Basham, J.D. Sedgwick, T. McClanahan, R.A. Kastelein, D.J. Cua, IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J. Exp. Med. 201, 233–240 (2005)
K. Ghoreschi, A. Laurence, X.P. Yang, C.M. Tato, M.J. McGeachy, J. Konkel, H.L. Ramos, L. Wei, T. Davidson, N. Bouladoux, J. Grainger, Q. Chen, Y. Kanno, W.T. Watford, H.W. Sun, G. Eberl, E. Schevach, Y. Belkaid, D.J. Cua, W. Chen, O´Shea, J.J.: Generation of pathogenic Th17 cells in the absence of TGF-β signaling. Nature 467, 967–971 (2010)
K. Ghoreschi, A. Laurence, X.P. Yang, K. Hirahara, J.J. O’Shea, T helper 17 cell heterogeneity and pathogenicity in autoinmune disease. Trends Immunol. 32, 395–401 (2011)
Y. Lee, A. Awasthi, N. Yosef, F.J. Quintana, S. Xiao, A. Peters, C. Wu, M. Kleinewietfeld, S. Kunder, D. Hafler, R.A. Sobel, A. Regev, V.K. Kuchroo, Induction and molecular signature of pathogenic Th17 cells. Nat. Immunol. 13, 991–999 (2012)
S.A. Basdeo, B. Moran, D. Cluxton, M. Canavan, J. McCormick, M. Connolly, C. Orr, K.H.G. Mills, D.J. Veale, U. Fearon, J.M. Fletcher, Polyfunctional, pathogenic CD161+ Th17 cells lineage cells are resistant to regulatory T cell-mediated suppression in the context of autoimmunity. J. Immunol. 195, 528–540 (2015)
R. Ramesh, L. Kozhaya, K. McKevitt, I.M. Djuretic, T.J. Carlson, M.A. Quintero, J.L. McCauley, M.T. Abreu, D. Unutmaz, M.S. Sundrud, Pro-inflammatory human Th17 cells selectively express P-glycoprotein and are refractory to glucocorticoids. J. Exp. Med. 211, 89–114 (2014)
T. Feng, A.T. Cao, C.T. Weaver, C.O. Elson, Y. Cong, Interleukin-12 converts Foxp3+ regulatory T cells to interferon-γ-producing Foxp3+ T cells that inhibit colitis. Gastroenterology 140, 2031–2043 (2011)
M.A. Kluger, M.C. Meyer, A. Nosko, B. Goerke, M. Luig, C. Wegscheid, G. Tiegs, R.A. Stahl, U. Panzer, O.M. Steinmetz, RORγt+ Foxp3+ cells are an independent bifunctional regulatory T cell lineage and mediate crescentic GN. J. Am. Soc. Nephrol. (2015). doi:10.1681/ASN.2014090880
K. Eyerich, S. Eyerich, Th22 cells in allergic disease. Allergo. J. Int. 24, 1–7 (2015)
I. Horie, N. Abiru, Y. Nagayama, G. Kuriya, O. Saitoh, T. Ichikawa, Y. Iwakura, K. Eguchi, T helper type 17 immune response plays an indispensable role for development of iodine-induced autoimmune thyroiditis in nonobese diabetic-H2h4 mice. Endocrinology 150, 5135–5142 (2009)
F. Hayashi, M. Watanabe, T. Nanba, N. Inoue, T. Akamizu, Y. Iwatani, Association of the -31C/T functional polymorphism in the interleukine-1β gene with the intractability of Graves’ disease and the proportion of T helper type 17 cells. Clin. Exp. Immunol. 158, 281–286 (2009)
T. Nanba, M. Watanabe, N. Inoue, Y. Iwatani, Increases of the Th1/Th2 cell ratio in severe Hashimoto’s disease and in the proportion of Th17 cells in intractable Graves’ disease. Thyroid 19, 495–501 (2009)
N. Figueroa-Vega, M.A. Pérez, I. Benedicto, F. Sánchez-Madrid, R. González-Amaro, M. Marazuela, Increased circulating pro-inflammatory cytokine and Th17 lymphocytes in Hashimoto´s thyroiditis. J. Clin. Endocrinol. Metab. 95, 953–962 (2010)
Q. Qin, P. Liu, L. Liu, R. Wang, N. Yan, J. Yang, X. Wang, M. Pandey, J. Zhang, The increased but non-predominant expression of Th17- and Th1-specific cytokines in Hashimoto´s thyroiditis but not in Graves´ disease. Braz. J. Med. Biol. Res. 45, 1202–1208 (2012)
D. Peng, B. Xu, Y. Wang, H. Guo, Y. Jiang, A high frequency of circulating Th22 and Th17 cells in patients with new onset Graves’ disease. PloS One 8, e68446 (2013)
D. Li, W. Cai, R. Gu, Y. Zhang, H. Zhang, K. Tang, P. Xu, F. Katirai, W. Shi, L. Wang, T. Huang, B. Huang, Th17 plays a role in the pathogenesis of Hashimoto’s thyroiditis in patients. Clin. Immunol. 149, 411–420 (2013)
B. Kristensen, L. Hegedüs, H.O. Madsen, T.J. Smith, C.H. Nielsen, Altered balance between self-reactive T helper (Th)17 cells and Th10 cells and between full-length forkhead box protein 3 (FoxP3) and FoxP3 splice variants in Hashimoto´s thyroiditis. Clin. Exp. Immunol. 180, 58–69 (2015)
H. Guo, D. Peng, Y. Wang, B.C. Xu, J.S. Ni, W. Meng, Y.F. Jiang, A higher frequency of circulating IL-22+ CD4+ T cells in chinese patients with newly diagnosed Hashimoto’s thyroiditis. PloS One 9, e84545 (2014)
H. Xue, X. Yu, L. Ma, S. Song, Y. Li, L. Zhang, T. Yang, H. Liu, The possible role of CD4+ CD25highFoxp3+/CD4+ IL-17A+ cell imbalance in the autoimmunity of patients with Hashimoto thyroiditis. Endocrine (2015). doi:10.1007/s12020-015-0569-y
S. Wang, S.E. Baidoo, Y. Liu, C. Zhu, J. Tian, J. Ma, J. Tong, J. Chen, X. Tang, H. Xu, L. Lu, T cell-derived leptin contributes to increased frequency oh T helper type 17 cells in female patients with Hashimoto’s thyroiditis. Clin. Exp. Immunol. 171, 63–68 (2012)
Y. Liu, X. Tang, J. Tian, C. Zhu, H. Peng, K. Rui, Y. Wang, C. Mao, J. Ma, L. Lu, H. Xu, S. Wang, Th17/Treg cells imbalance and GITRL profile in patients with Hashimoto´s thyroiditis. Int. J. Mol. Sci. 15, 21674–21686 (2014)
S. Leskela, A. Serrano, H. de la Fuente, A. Rodríguez-Muñoz, A. Ramos-Levi, M. Sampedro-Núñez, F. Sánchez-Madrid, R. González-Amaro, M. Marazuela, Graves’ disease is associated with defective expression of the immune regulatory molecule galectin-9 in antigen-presenting dendritic cells. PLoS One 16, e0123938 (2015)
J.R. Li, F.Y. Hong, J.Y. Zeng, G.L. Huang, Functional interleukin-17 receptor A are present in the thyroid gland in intractable Graves’ disease. Cell. Immunol. 281, 85–90 (2013)
R.H. Song, Z.Y. Yu, Q. Qin, X. Wang, F.S. Muhali, L.F. Shi, W.J. Jiang, L. Xiao, D.F. Li, J.A. Zhang, Different levels of circulating Th22 cell and its related molecules in Graves’ disease and Hashimoto’s thyroiditis. Int. J. Clin. Exp. Pathol. 7, 4024–4031 (2014)
X. Bai, J. Sun, W. Wang, Z. Shan, H. Zheng, Y. Zhao, M. Gong, W. Teng, Increased differentiation of Th22 cells in Hashimoto’s thyroiditis. Endocr. J. 61, 1181–1190 (2014)
R.M. Ruggeri, P. Minciullo, S. Saitta, S. Giovanazzo, R. Certo, A. Campennì, F. Trimarchi, S. Gangemi, S. Benvenga, Serum interleukin-22 (IL-22) is increased in the early stage of Hashimoto’s thyroiditis compared to non-autoimmune thyroid disease and healthy controls. Hormones 13, 338–344 (2014)
R.M. Ruggeri, S. Saitta, M. Cristani, S. Giovanazzo, V. Tigano, F. Trimarchi, S. Benvenga, S. Gangemi, Serum interleukin-23 (IL-23) is increased in Hashimoto´s thyroiditis. Endocr. J. 61, 359–363 (2014)
L. Guan, X. Wang, S. Meng, L. Shi, W. Jiang, L. Xiao, X. Shi, J. Xu, J. Zhang, Increased IL-21/IL-22R expression and its proinflammatory effects in autoimmune thyroid disease. Cytokine 72, 160–165 (2015)
S.J. Shan, R.S. Douglas, The pathophysiology of thyroid eye disease. J. Neuroophthalmol. 34, 177–185 (2014)
H. Wei, M. Guan, Y. Qin, C. Xie, X. Fu, F. Gao, Y. Xue, Circulating levels of miR-146a and IL-17 are significantly correlated with the clinical activity of Graves’ ophthalmopathy. Endocr. J. 61, 1087–1089 (2014)
S.E. Kim, J.S. Yoon, K.H. Kim, S.Y. Lee, Increased serum interleukin-17 in Graves’ ophthalmopathy. Graefes Arch. Clin. Exp. Ophthalmol. 250, 1521–1526 (2012)
A.K. Huber, E.M. Jacobson, K. Jazdzewski, E.S. Concepcion, Y. Tomer, Interleukin (IL)-23 receptor is a major susceptibility gene for Graves’ ophthalmopathy: the IL-23/T-helper 17 axis extends to thyroid autoimmunity. J. Clin. Endocrinol. Metab. 93, 1077–1081 (2008)
L. Zheng, P. Ye, C. Liu, The role of the IL-23/IL-17 axis in the pathogenesis of Graves’ disease. Endocr. J. 60, 591–597 (2013)
F. Rajaii, A.N. McCoy, T.J. Smith, Cytokines are both villains and potential therapeutic targets in thyroid-associated ophthalmopathy: From bench to bedside. Expert. Rev. Ophthalmol. 9, 227–234 (2014)
D. Cao, R. van Vollenhoven, L. Klareskog, C. Trollmo, V. Malmstrom, CD25brightCD4+ regulatory T cells are enriched in inflamed joints of patients with chronic rheumatic disease. Arthritis Res. Ther. 6, R335–R346 (2004)
M.R. Ehrenstein, J.G. Evans, A. Singh, S. Moore, G. Warnes, D.A. Isenberg, C. Mauri, Compromised function of regulatory T cells in rheumatoid arthritis and reversal by anti-TNFalpha therapy. J. Exp. Med. 200, 277–285 (2004)
J. Haas, A. Hug, A. Viehover, B. Fritzsching, C.S. Falk, A. Filser, T. Vetter, L. Milkova, M. Korporal, B. Fritz, B. Storch-Hagenlocher, P.H. Krammer, E. Suri-Payer, B. Wildemann, Reduced suppressive effect of CD4+ CD25 high regulatory T cells on the T cell immune response against myelin oligodendrocyte glycoprotein in patients with multiple sclerosis. Eur. J. Immunol. 35, 3343–3352 (2005)
U. Feger, C. Luther, S. Poeschel, A. Melms, E. Tolosa, H. Wiendl, Increased frequency of CD4+ CD25+ regulatory T cells in the cerebrospinal fluid but not in the blood of multiple sclerosis patients. Clin. Exp. Immunol. 147, 412–418 (2007)
J. Maul, C. Loddenkemper, P. Mundt, E. Berg, T. Giese, A. Stallmach, M. Zeitz, R. Duchmann, Peripheral and intestinal regulatory CD4+ CD25(high) T cells in inflammatory bowel disease. Gastroenterology 128, 1868–1878 (2005)
H. Sugiyama, R. Gyulai, E. Toichi, E. Garaczi, S. Shimada, S.R. Stevens, T.S. McCormick, K.D. Cooper, Dysfunctional blood and target tissue CD4+ CD25high regulatory T cells in psoriasis: mechanism underlying unrestrained pathogenic effector T cell proliferation. J. Immunol. 174, 164–173 (2005)
H.J. Bovenschen, P.C. van de Kerkhof, P.E. van Erp, R. Woestenenk, I. Joosten, H.J. Koenen, Foxp3+ regulatory T cells of psoriasis patients easily differentiate into IL-17Aproducing cells and are found in lesional skin. J. Invest. Dermatol. 131, 1853–1860 (2011)
T.M. Brusko, C.H. Wasserfall, M.J. Clare-Salzler, D.A. Schatz, M.A. Atkinson, Functional defects and the influence of age on the frequency of CD4+ CD25+ T-cells in type 1 diabetes. Diabetes 54, 1407–1414 (2005)
J.M. Lawson, J. Tremble, C. Dayan, H. Beyan, R.D. Leslie, M. Peakman, T.I. Tree, Increased resistance to CD4+ CD25hi regulatory T cell-mediated suppression in patients with type 1 diabetes. Clin. Exp. Immunol. 154, 353–359 (2008)
B. Alvarado-Sánchez, B. Hernández-Castro, D. Portales-Pérez, L. Baranda, E. Layseca-Espinosa, C. Abud-Mendoza, A.C. Cubillas-Tejeda, R. González-Amaro, Regulatory T cells in patients with systemic lupus erythematosus. J. Autoimmun. 27, 110–118 (2006)
J.C. Crispin, A. Martínez, J. Alcocer-Varela, Quantification of regulatory T cells in patients with systemic lupus erythematosus. J. Autoimmun. 21, 273–276 (2003)
A. Alunno, M. Manetti, S. Caterbi, L. Ibba-Manneschi, O. Bistoni, E. Bartoloni, V. Valentini, R. Terenzi, R. Gerli, Altered immunoregulation in rheumatoid arthritis: the role of regulatory T cells and proinflammatory th17 cells and therapeutic implications. Mediat. Inflamm. (2015). doi:10.1155/2015/751793
S. Alvermann, C. Hennig, O. Stüve, H. Wiendl, M. Stangel, Immunophenotyping of cerebrospinal fluid cells in multiple sclerosis: in search of biomarkers. JAMA Neurol. 71, 905–912 (2014)
J.S. Tzartos, M.A. Friese, M.J. Craner, J. Palace, J. Newcombe, M.M. Esiri, L. Fugger, Interleukin-17 production in central nervous system-infiltrating T cells and glial cells is associated with active disease in multiple sclerosis. Am. J. Pathol. 172, 146–155 (2008)
C.S. Catana, I. Berindan Neagoe, V. Cozma, C. Magdas, F. Tabaran, D.L. Dumitrascu, Contribution of the IL-17/IL-23 axis to the pathogenesis of inflammatory bowel disease. World J. Gastroenterol. 21, 5823–5830 (2015)
J.L. Harden, J.G. Krueger, A.M. Bowcock, The immunogenetics of psoriasis: a comprehensive review. J. Autoimmun. (2015). doi:10.1016/j.jaut.2015.07.008
L. Reinert-Hartwall, J. Honkanen, H.M. Salo, J.K. Nieminen, K. Luopajärvi, T. Härkönen, R. Veijola, O. Simell, J. Ilonen, A. Peet, V. Tillmann, M. Knip, O. Vaarala, DIABIMMUNE Study Group, Th1/Th17 plasticity is a marker of advanced β cell autoimmunity and impaired glucose tolerance in humans. J. Immunol. 194, 68–75 (2015)
M.S. Maddur, P. Miossec, S.V. Kaveri, J. Bayry, Th17 cells: biology, pathogenesis of autoimmune and inflammatory diseases, and therapeutic strategies. Am. J. Pathol. 181, 8–18 (2012)
Acknowledgments
This work was partially supported by the following grants: 95395 from the Fondo de Cooperación Internacional en Ciencia y Tecnología (FONCICYT-European Union, México, to R.G.-A.); PI13-01414 and PIE-0041 BIOIMID from the Fondo de Investigación Sanitaria—Instituto de Salud Carlos III (to M.M.), and S2011/BMD-2328 TIRONET from the Comunidad de Madrid (to M.M.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
González-Amaro, R., Marazuela, M. T regulatory (Treg) and T helper 17 (Th17) lymphocytes in thyroid autoimmunity. Endocrine 52, 30–38 (2016). https://doi.org/10.1007/s12020-015-0759-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-015-0759-7