Abstract
Cord blood (CB) is an important alternative source of hematopoietic stem cells (HSCs) for transplantation today. The principal drawbacks of cord blood transplantation are the limited number of hematopoietic stem cells and a long time to engraftment. Several promising approaches for engraftment enhancement are under intensive investigation. Such are transplantation with two cord blood units, co transplantation of cord blood and haploidentical HSCs and different methods for expansion of cord blood hematopoietic stem cells. In addition there are several ways for improving of homing of HSCs such as co- infusion of CB hematopoietic stem cells and mesenchymal stem cells, administration of parathyroid hormone (PTH), intra- bone transplantation and targeting the CXCR4/SDF1 system. These strategies are expected to increase the availability of transplantation to adults, for whom the chance to find a cord blood suitable for a single unit transplant is small. Recent advances in elucidation of the molecular mechanisms responsible for the proliferation and self-renewal of hematopoietic stem cells may bring further improvement of the outcomes of cord blood transplantation. This review summarizes the recent progress in the field of cord blood derived hematopoietic stem cells. It presents the strategies applied and points out directions for the future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
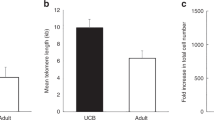
The first umbilical cord blood (CB) transplantation (CBT) was reported by Gluckman in 1989 [1]. Since then nearly 20,000 CB transplants have been performed worldwide in pediatric and adult patients. Hematopoietic stem cells (HSCs) derived from CB are superior compared to their counterparts from mobilized peripheral blood (PB) or bone marrow (BM) in several ways. They are less mature [2, 3] and have greater telomere length [4, 5], which may contribute to their superior proliferation potential [6–8]. They are immediately available and their collection is simple with no risk to mother or newborn (Fig. 1). However, the amount of nucleated cells in the cord blood unit is ten times smaller than in BM or PB grafts. This translates clinically into a higher incidence of engraftment failure and longer time to cell recovery – the major default of CBT. Transplants with CB result in significantly lower rates of acute and chronic graft versus host disease (GVHD) [9, 10], in spite of broader human leukocyte antigen (HLA) disparity between CB unit and recipient. This is possibly due the lower number and relatively naïve repertoire of the cord blood T cells [11, 12]. The role of dendritic cells was not excluded and is intensively studied [13]. It is of great importance that the graft versus leukemia (GVL) effect is preserved, most probably due to the unique natural killer (NK) subsets [14, 15].
Today there are about 600,000 CB units frozen in public cord banks in 25 countries. This enables selection of an optimal CB unit that is crucial for a successful transplantation [16]. For patients with hematological malignancies the order of priority should be a unit with the biggest cell dose, seconded by maximal HLA matching. While in patients with genetic diseases, for which no GVL activity is necessary, maximal HLA matching is of first concern [17]. Cumulative clinical data have demonstrated the growing importance of the cell dose with increasing HLA mismatch. Researchers from the University of Minnesota have defined adequate single units as: >3 × 107 nucleated cells/kg (NC/kg) for 6/6 HLA-matched units,>4 × 107 NC/kg for 5/6 HLA-matched units and >5 × 107 NC/kg for 4/6 HLA-matched units [18]. Since counting total nucleated cells (TNC) may involve also subsets that are not contributing directly to engraftment, there is increasing emphasis on the number and quality of the CD34 positive cells in CB graft. Wagner and coworkers demonstrated correlation between a CD34+ cell dose of 1.7 × 105 cell/kg and a faster neutrophil recovery [19]. The data of Sanz et al. may indicate that the CD34+ cell count is the most important factor dictating and predicting a CBT outcome [20]. Unfortunately, the total CD34+ cell count is still not routinely used in comparative studies due to lack of sufficient common standardization of the CD34 assays among the various laboratories.
CBT - Clinical Practice
In 2004 two major registry reports [21, 22] and one single institution analysis [23] compared outcomes of transplants of unrelated CB and bone marrow to adults with leukemia. The results are shown in Table 1.
The study of Rocha et al. [21] involved two groups and a total of 682 adults with acute leukemia. One group included 98 unrelated CB and the second group 584 unrelated BM (6/6 matched) transplants. The time period was between 1998 and 2002. Both groups behaved similarly in terms of transplant related mortality (TRM), chronic GVHD, relapse rate and leukemia free survival (LFS), but the unrelated CB group had a significantly lower incidence of acute GVHD. Laughlin et al. [22] compared transplantations performed to leukemia patients from 1996 to 2001. Their study involved one group of 150 unrelated CBT with less than 5/6 mismatch, a second group of 367 unrelated BMT (6/6 matched) and a third group of 83 unrelated BMT (5/6 matched). Recipients of the 6/6 matched unrelated BM showed the lowest TRM and overall mortality and no significant difference in acute GVHD. The results for unrelated CBT and 5/6 matched unrelated BMT were similar.
The results of Rocha and Laughlin differ probably due to difference in HLA antigen matching, as the first analyzed transplantations performed only after 1998, when the unit and the recipient selections became better.
In a similar vein Takahashi et al. [23]compared 113 unrelated CB transplants (1/6–4/6 matched) to 45 unrelated BM transplants (88% matched 6/6). The UCB group exhibited improved LFS, significantly less TRM and less acute GVHD. In a pilot report from 2007 the same group pronounced CBT as the method of choice for unrelated donor grafts. They pursued the outcome of unrelated CBT for 100 patients with hematological malignancies [24] and found the results similar to those obtained with matched related BM and PBSC transplantations. These results are encouraging, but do not take into account the ethnical homogeneity of the Japanese population.
Very recently, Rocha et al. [25] examined the outcomes of 1240 adults with acute leukemia who received unrelated CB (148), or unrelated BM (matched- 243; mismatched- 111), or unrelated PBSC (matched -518; mismatched -210). The time span was 4 years (2002–2006) and the median follow up was 2 years. In contrast to the previous reports, all BM and PBSC grafts were matched at allele levels for HLA-A, -B, -C and –DRB1. In this analysis the CB recipients had delayed neutrophil recovery and the lowest levels of acute and chronic GVHD. They showed a TRM higher in CB recipients than in matched BM and matched PBSC recipients (41% versus 26 and 27%, respectively), but similar to that in mismatched BM and PBSC recipients. LFS was similar for CB and matched PBSC and BM recipients. 74% of the CB grafts were 4/6 matched and the median cell dose was 2.5 × 107 NC/kg. Today 2.5 × 107 NC/kg is considered a rather low TNC threshold for a CB unit with a high number of HLA mismatches. It is therefore possible that 5–6/6 matched CB may provide even better results.
Interestingly, in a similar analysis, that included 619 children with acute leukemia, the same group demonstrated, that LFS after 6/6 matched CB was higher, (60%) than after the current “gold standard” 8/8 matched unrelated BM, (38%) [26]. This indicates that 6/6 matched CB with an adequate cell dose may be the superior HSC source for children with hematological malignancies (the probability to find such a graft is only 10%).
This finding is of vast importance taking into account that in the US only 30% of the patients for whom a search for unrelated matched BM was initiated ultimately received a transplant. This is mainly due to deterioration of the patients’ condition or death during the search period. In view of this statistics, for patients with high risk hematological malignancies, such as acute leukemia, a search for unrelated CB and BM donors should be started simultaneously. CB, matched or with up to 2 HLA mismatched antigens, should be preferred if a matched BM donor cannot be found within 2–3 months.
Strategies for Engraftment Enhancement
Engraftment failure and prolonged time to cell recovery remain major challenges for CBT. There are two principal approaches for engraftment hastening. One is to augment the cell dose; the other is to improve the stem cells homing. Different strategies aiming at these goals are detailed below.
How to Increase the Cell Dose?
Transplantations with Two Cord Blood Units
Transplantation with two CB units gained much publicity during the last 4 years and has the potential to become a major leader in the field. It was pioneered by the group of the University of Minnesota when in 2005 Barker et al. [27] published the pilot study of a double CBT to 23 adults with leukemia after myeloablative conditioning (Cytoxan and total body irradiation). The median TNC dose infused was 3.5 × 107/kg. Most of the patients received CB units mismatched in 1 or 2 out of 6 HLA antigens. The median time to neutrophil recovery was 23 days. The incidences of grade II–IV and III–IV acute GVHD were 65% and 13%, respectively. The rate of chronic GVHD was 23% and the TRM was 22%. After 10 months of follow up the predicted disease free survival (DFS) was 57%. Twenty one of 23 consecutive patients (all evaluable patients) achieved donor engraftment. Such an elevated engraftment rate for adults was unprecedented and exciting. The time to neutrophil recovery was similar to that after single CBT (23 days). A potential rise in GVHD was anticipated and indeed, the rate of moderate acute GVHD seems higher, but the frequency of severe acute and chronic GVHD is comparable for double and single CBT. There is an opinion, that the choice of a CB unit for transplant should include consideration of the partial inter-unit match [28, 29] in addition to the unit-recipient match. This recommendation is yet to be validated by multicenter studies.
Subsequently, Veneris et al. [30], also from the Minnesota group, published the results of single (n = 67) and double (n = 29) CBTs for patients with acute lymphatic and myeloid leukemia, who underwent a myeloablative conditioning with Cytoxan, Fludarabine and total body irradiation. The most fascinating discovery was the low -11% risk of relapse for patients in complete remission (CR) 1/2, who were transplanted with two CB units. For single unit recipients the risk of relapse is (54%, p < 0.01). Extension of this study to 177 patients [31] showed similar results.
These reports suggest that reduced risk of relapse of acute leukemia is associated with double CBT. A putative explanation is that in the double CB the GVL effect is more prominent, due to the higher HLA disparity and reduces relapse. A different explanation is a non-HLA disparity, such as the Killer Inhibitory Receptors (KIR) mismatch, between the CB units and the recipient, or between the 2 CB units themselves [32]. Altogether these data presents DCBT as a feasible curative procedure that will extent application of CBT to adults.
In accordance, two randomized phase II clinical trials in adults with acute leukemia and one phase III prospective pediatric experiment comparing double versus single CBT have been recently initiated by the Minnesota group.
A curious observation well documented, but not understood is that about 1 month after myeloablative conditioning and by day 100 after nonmyeloablative double CBT, only one of the administered CB units sustains the hematopoiesis. Even when multiple (up to 7) CB units were infused at the same time, only one single CB unit is responsible for the long-term engraftment [33]. In the settings of DCBT, the unit which disappears, possibly contributes to hematopoiesis during the first weeks, hastens neutrophil recovery and contributes to the higher rate of engraftment. It is not yet possible to predict which of the multiple CBT units will be the one to prevail and sustain hematopoiesis.
Variables that seem likely to be involved have been investigated. Such are the cell dose, the viability of the cells, the CD3 cell number, the HLA match, the ABO type, the gender and the order of administration. None of them has been found to enable prediction of the prevailing unit.
Co-transplantation of CB and Haploidentical HSCs
Magro et al. [34] reported another novel approach that includes co-transplantation of cord blood together with a limited amount of HSC from a haploidentical donor. Transient hematopoiesis from the haploidentical cells resulted in a faster neutrophil recovery (10 days). Later on, the HSC from the CB provided the long-term stable engraftment while the haploidentical cells disappeared completely. Transient contribution of haploidentical HSC or even non HLA sharing cells provided the necessary protection from infections in the early stage of engraftment and substantially decreased the day 100 mortality.
Ex Vivo Expansion of CB Derived Stem Cells
Preclinical studies demonstrated the superior proliferative potential of CB derived HSC. Cytokine stimulated proliferation of CB stem cells was found to be 80- fold better than that of BM progenitors [35]. Vormoor et al. [36] demonstrated that CB, but not adult BM stem cells, can repopulate non diabetic severe combined immune deficient (NOD/SCID) mice without aid by exogenous cytokines.
In 2002 Shpall et al. [37] reported the first safe transplantation of ex- vivo expanded umbilical stem cells. CB was frozen in two fractions. CD34+ cells isolated from one of the fractions were co-cultured with cytokine cocktail (SCF, TPO and G-CSF). After 10 days in culture the TNC dose increased 56-fold and CD34+ cells increased four- fold. Thirty seven patients were co-administered with one expanded and one unmanipulated CB fractions. No engraftment hastening was detected.
Later on, McNiece et al. [38] applied a two-step, 14-days expansion protocol which yields and increase in TNC as great as 400-fold and a 20- fold increase in the CD34+ cells. The efficacy of this technique was tested in the prospective trial in MD Anderson. Seventy one patients with hematologic malignancies after myeloablative or reduced intensity conditioning were randomized for transplantation with two unmanipulated CB units or one unmanipulated and one expanded CB unit using a two-step strategy [39]. Both units were infused on day 0. The engraftment enhancement was observed in the patients who received one expanded and one unmanipulated CB units They showed a significantly faster neutrophil recovery median of 7 days (range 4–15 days, n = 14) compared to 14 days (range 5–32 days, n = 12) for those administered with two unmanipulated CB units.
Interestingly, several months after transplantation of the combination expanded/unmanipulated, chimerism analysis showed only the unmanipulated CB unit. It has been suggested that ex vivo expansion is associated with cell cycle abnormalities [40], acquired homing defects [41, 42] and induction of apoptosis [43–45]. This may explain the failure to speed engraftment by CB expansion. The predominance of the unmanipulated unit raised concerns, that during ex- vivo expansion one may be selectively expanding a specific subset of stem cells, which are short- lived, more mature and of lower “quality”. This predominance could result in initial earlier hematopoietic recovery followed by later graft failure [46]. Yet, it could be utilized when ex- vivo expanded and “unmanipulated” CB fractions are combined for transplantation,. In this case, short- term progenitors from the expanded portion would provide faster transient engraftment, reducing early morbidity and mortality, while the unmanipulated CB would provide sustained hematopoiesis [47].
Methods to improve the current expansion protocols exist. Thus, selection of HSCs for expansion based on the CD133 rather than the CD34 expression may isolate more immature stem cells with greater proliferation potential [48, 49]. Transplantation of CD133+ progenitors has already shown excellent engraftment in murine models [50].
Another approach to improve the expansion outcome is development of new agents to stimulate proliferation of HSCs. For example, the copper chelator tetraethylenepentamine (TEPA) and the histone deacetylase inhibitor (HDAC) - valproic acid have been shown to transiently block differentiation of CB stem cells thus enhancing their proliferation [51]. TEPA and HDAC are now at the stage of phase II clinical trials [52, 53].
With the recent advances in understanding the molecular mechanisms involved in proliferation and maintenance of HSCs, additional candidates for further manipulation of HSCs have been introduced. Such are Notch-1, Wnt/B-catenin, the receptor tyrosine kinase Tie2, transcriptional factor Bmi-1 and homebox genes HoxB. Development of synthetic compounds in order to target these sites is of particular interest [54]. Thus, Delaney et al. [55] reported on five patients with high risk leukemia transplanted with a combination of one unmanipulated and one expanded CB units using engineered Notch delta- 1 ligand and cytokines cocktail. Expansion resulted in the average increase in CD34+ population of 160-fold and in TNC number of 660 fold. The time to neutrophil recovery was 14 days (range 7–34, n = 5) in expansion arm compared to 25 days (range 16–48, n = 17) in recipients of two unmanipulated CB units. These encouraging results suggest that using Notch delta-1 ligand, may promote expansion of less mature HSCs with greater proliferation potential.Future clinical trials will establish the role of the novel techniques of ex vivo expansion in clinical practice.
How to Improve the Stem Cells Homing?
Principal modalities to enhance a homing of CB HSCs include application of mesenchymal stem cells (MSCs), direct intra- bone transplantation and administration of parathyroid hormone (PTH), as well as modulation of the CXCR4/SDF1 system.
Mesenchymal Stem Cells (MSCs) to Facilitate Engraftment
Schofild’s “niche” hypothesis suggests that HSCs reside in the micro environmental niches [56] which consist of MSC- derived cells such as osteoblasts, chondrocytes and adipocytes. These cells provide a rich variety of signals (cytokines, extracellular matrix proteins and adhesion molecules) regulating proliferation and differentiation of HSCs [57]. Currently BM is a main source of MSCs for therapeutic use. However, recently MSC–like cells have been isolated from Wharton’s jelly of umbilical cords and named umbilical cord matrix (UCM) stem cells [58, 59].
MSCs may accelerate engraftment in two ways. First, MSCs are able to enhance ex vivo expansion of HSCs [60, 61]. Co-culture of bone marrow derived MSCs and cord blood HSCs resulted in a 16- to 37- fold increase in CD34+ cells [62]. A clinical trial using this strategy is underway at MD Anderson with CB HSCs expanded on related donor MSCs combined with an unmanipulated CB unit. Two units were co- infused to first 6 patients following myeloablative conditioning. The median time to neutrophil recovery was 14.5 days (range 12–23) and for platelet engraftment was 30 days (range 25–51). Two of six patients developed grade II GVHD that resolved with steroids. One patient died of pneumonia at day 150. Five of six patients are alive and in complete remission after 1 year follow up [39].
The second approach to facilitate an engraftment is a co- transplantation of HSCs and MSCs. The intriguing observation is that MSCs have very low immunogenity and therefore could be transplanted over the HLA barrier. Co-infusion of ex-vivo expanded “third party” (non donor, not- recipient) BM derived MSCs and HSCs from PB and BM was tested in pilot studies [63, 64]. Increased relapse rate in patients administered with MSCs was reported by Ning et al. [63] and should be reevaluated in future larger trials.
Regarding cord blood, McMillan et al. [65] co-administered haploidentical parental MSCs and single CB unit to 8 children. Three patients received the second dose of MSCs on day +21. All 8 patients achieved neutrophil engraftment at a median of 19 days. With a median follow- up of 6.8 years 5 patients were alive and disease free. In another study, the “third party” expanded MSCs and cord blood HSCs were co-administered to 9 patients [66]. The outcomes were compared these of 46 patients who did not received MSCs. No significant differences in engraftment or incidence of acute GVHD were observed. Two patients in the MSCs group developed steroid refractory grade II acute GVHD. It is important to note that, repeated infusion of MSCs caused a complete resolution of GVHD.
The ability of MSCs to control acute GVHD originates in their ability to modulate the immune response through interaction with T – lymphocytes, B- lymphocytes, NK-cells and dendritic cells [67, 68]. The role of MSCs in prevention and treatment of acute GVHD is extensively studied today [69–71].
In conclusion, expanded “third party” MSCs may become a universal donor that will significantly change the field of cord blood transplantation. However, many questions remain open, like what are the optimal source, dose and mode of administration. The concern regarding a possible increase in relapse rate and possible tumorogenicity also need to be addressed
Parathyroid Hormone Treatment (PTH)
Osteoblasts are important supporting components of the hematopoetic niche. Calvi et al. [72] demonstrated that stimulation with PTH results in increasing number of osteoblasts and HSC in a murine model. In view of the promising results of the phase I trial, the team of Ballen initiated lately a phase II clinical trial testing the kinetics of engraftment during PTH treatment following CBT [73].
Intra-bone Transplantation
Frassoni et al. reported recently the results of a phase I/II study performed on 32 patients with acute leukemia who received a single intra-bone marrow transplantation of CB following myeloablative conditioning. The overall survival was 50% compared to 39% for intravenous CB transplantation. The median transplanted cell dose was relatively low, 2.6 × 107/kg (range 1.4–4.2 × 107/kg). The median time to neutrophil recovery was 23 days with remarkably fast platelet engraftment at 36 days, in comparison to about 3 months for intravenous transplantation. Extension of the analysis to 62 patients [74] demonstrated similar results suggesting that intra-bone transplantation of CB cells may overcome the problem of delayed platelet recovery following CBT. Another intriguing and promising discovery was that the intra-bone approach diminished acute GVHD grade III-IV to 0% while with the conventional intravenous method it was 11%. In accordance, Phase I/II clinical trials in Minnesota have evaluated the efficacy of a double CB grafts intra bone administration to 10 recipients [75]. One of the units was randomly assigned to intravenous and the second - to intra-bone marrow infusion. Nine of ten patients engrafted, 5 with the intravenous unit and 4 with the intra-bone unit. In contrast to the single CBT model, and the Frassoni data, intra-bone administration of a single one of two CB units did not shorten the time to neutrophil and platelet recovery, it rather caused a high TRM of 50%. This discrepancy may indicate that this interesting though technically cumbersome approach may not be the ultimate solution.
Targeting CXCR4/CDF1 system
Stem cell derived factor 1 (SDF1) through its receptor CXCR4 promote homing and retention of HSCs in bone marrow [76].After transplantation, HSCs traffic to the BM following SDF-1 gradient, which is increased by the conditioning regimen (TBI, myeloablative chemotherapy) [77–79]. Thus, optimal expression of CXCR4 by HSCs and the effective level of SDF-1 in BM are two important factors regulating an engraftment of HSCs in BM. Several molecules, such as complement protein C3a, hyaluronic acid, fibronectin and fibrinogen, have been shown to up regulate the expression of CXCR4 on the HSCs [80–82].The role of pretreatment of a CB graft with a-C3a in engraftment enhancement is currently being investigated in preclinical models [83]. CD26/dipeptidylpeptidase IV (DPPIV) is an enzyme which is able to cleave SDF-1 and as a results to alter the migration of HSCs to bone marrow. Based on the preclinical data CD26/DPPIV is expressed on the subset of cord blood HSCs. Inhibition of the CD26/DPPIV peptidase activity may therefore represent an additional potential approach for improving of homing and engraftment during cord blood transplantation [84].
Conclusions
Cord blood derived stem cells, as compared to their counterparts from bone marrow and peripheral blood, have a bigger telomerase length and demonstrate a higher proliferation potential. One of their main drawbacks for transplantation, the low cell dose is on the way to being resolved. Transplantation with two cord blood units is the most established strategy and has already considerably increased the availability of CBT to adults with hematological malignancies. Another approach, the feasibility of which was confirmed in the clinic, is expansion of HSCs using cytokines or differentiation blocking agents, such as TEPA.
Double CBT and transplantation of expanded stem cells have significantly reduced the high incidence of graft failure traditionally being related to CBT. The recent data that double CBT may be associated with a reduced risk of relapse in patients with acute leukemia is especially encouraging. However, the prolonged time to cell recovery and the early mortality associated with CB transplants remain to be improved. Improvement may come from the development of a new compounds capable to target key cites of the molecular network responsible for the HSCs renewal and maintenance. Encouraging results from the first trial using one of such a compounds, Notch delta-1 ligand, raise the hope that this novel agent may promote expansion of HSCs with greater proliferation potential.
Application of MSCs is an additional new approach to enhance engraftment and control GVHD, which is now being tested in clinical trials. Hopefully, at least some of aforementioned original and novel strategies will enter our daily clinical practice soon, changing favorably this extremely interesting and challenging field – transplantation of cord blood stem cells.
References
Gluckman, E., Broxmeyer, H. A., Auerbach, A. D., et al. (1989). Hematopoietic reconstitution in a patient with Fanconi’s anemia by means of umbilical-cord blood from an HLA-identical sibling. The New England Journal of Medicine, 321, 1174–1178.
Holyoake, T. L., Nicolini, F. E., & Eaves, C. J. (1999). Functional differences between transplantable human hematopoietic stem cells from fetal liver, cord blood, and adult marrow. Experimental Hematology, 27, 1418–1427.
Hows, J. M., Bradley, B. A., Marsh, J. C., et al. (1992). Growth of human umbilical-cord blood in longterm haemopoietic cultures. Lancet, 340, 73–76.
Schuller, C. E., Jankowski, K., & Mackenzie, K. L. (2007). Telomere length of cord blood-derived CD34(+) progenitors predicts erythroid proliferative potential. Leukemia, 21, 983–991.
Gammaitoni, L., Weisel, K. C., Gunetti, M., et al. (2004). Elevated telomerase activity and minimal telomere loss in cord blood long-term cultures with extensive stem cell replication. Blood, 103, 4440–4448.
Lu, L., Xiao, M., Shen, R. N., Grigsby, S., & Broxmeyer, H. E. (1993). Enrichment, characterization, and responsiveness of single primitive CD34 human umbilical cord blood hematopoietic progenitors with high proliferative and replating potential. Blood, 81, 41–48.
Tanavde, V. M., Malehorn, M. T., Lumkul, R., et al. (2002). Human stem-progenitor cells from neonatal cord blood have greater hematopoietic expansion capacity than those from mobilized adult blood. Experimental Hematology, 30, 816–823.
Piacibello, W., Sanavio, F., Severino, A., et al. (1999). Engraftment in nonobese diabetic severe combined immunodeficient mice of human CD34(+) cord blood cells after ex vivo expansion: evidence for the amplification and self-renewal of repopulating stem cells. Blood, 93, 3736–3749.
Rubinstein, P., Carrier, C., Scaradavou, A., et al. (1998). Outcomes among 562 recipients of placental-blood transplants from unrelated donors. The New England Journal of Medicine, 339, 1565–1577.
Gluckman, E., Rocha, V., Boyer-Chammard, A., et al. (1997). Outcome of cord-blood transplantation from related and unrelated donors. Eurocord transplant group and the European blood and marrow transplantation group. The New England Journal of Medicine, 337, 373–381.
Nitsche, A., Zhang, M., Clauss, T., Siegert, W., Brune, K., & Pahl, A. (2007). Cytokine profiles of cord and adult blood leukocytes: differences in expression are due to differences in expression and activation of transcription factors. BMC Immunology, 8, 18.
Garderet, L., Dulphy, N., Douay, C., et al. (1998). The umbilical cord blood alphabeta T-cell repertoire: characteristics of a polyclonal and naive but completely formed repertoire. Blood, 91, 340–346.
Mohty, M., & Gaugler, B. (2008). Inflammatory cytokines and dendritic cells in acute graft-versus-host disease after allogeneic stem cell transplantation. Cytokine & Growth Factor Reviews, 19, 53–63.
Dalle, J. H., Menezes, J., Wagner, E., et al. (2005). Characterization of cord blood natural killer cells: implications for transplantation and neonatal infections. Pediatric Research, 57, 649–655.
Verneris, M. R., & Miller, J. S. (2009). The phenotypic and functional characteristics of umbilical cord blood and peripheral blood natural killer cells. British Journal Haematology, 147, 185–191.
Sullivan, M. J. (2008). Banking on cord blood stem cells. Nature Reviews. Cancer, 8, 555–563.
Rocha, V., & Gluckman, E. (2009). Improving outcomes of cord blood transplantation: HLA matching, cell dose and other graft- and transplantation-related factors. British Journal Haematology, 147, 262–274.
Smith, A. R., & Wagner, J. E. (2009). Alternative haematopoietic stem cell sources for transplantation: place of umbilical cord blood. British Journal Haematology, 147, 246–261.
Wagner, J. E., Barker, J. N., DeFor, T. E., et al. (2002). Transplantation of unrelated donor umbilical cord blood in 102 patients with malignant and nonmalignant diseases: influence of CD34 cell dose and HLA disparity on treatment-related mortality and survival. Blood, 100, 1611–1618.
Sanz, Jm, Saavedra, S., Montesinos, P., et al. (2008). Unrelated Donor Umbilical Cord Blood Transplantation (UD-UCBT) for adult aatients with high-risk Acute Myeloid Leukemia (AML). ASH Annual Meeting Abstracts, 112, 4401.
Rocha, V., Labopin, M., Sanz, G., et al. (2004). Transplants of umbilical-cord blood or bone marrow from unrelated donors in adults with acute leukemia. The New England Journal of Medicine, 351, 2276–2285.
Laughlin, M. J., Eapen, M., Rubinstein, P., et al. (2004). Outcomes after transplantation of cord blood or bone marrow from unrelated donors in adults with leukemia. The New England Journal of Medicine, 351, 2265–2275.
Takahashi, S., Iseki, T., Ooi, J., et al. (2004). Single-institute comparative analysis of unrelated bone marrow transplantation and cord blood transplantation for adult patients with hematologic malignancies. Blood, 104, 3813–3820.
Takahashi, S., Ooi, J., Tomonari, A., et al. (2007). Comparative single-institute analysis of cord blood transplantation from unrelated donors with bone marrow or peripheral blood stem-cell transplants from related donors in adult patients with hematologic malignancies after myeloablative conditioning regimen. Blood, 109, 1322–1330.
Rocha, V., Eapen, M., Scaradavou, E., Gluckman, E., Laughlin, M., Stevens, M. M., et al. (2009). Effect of stem cell source on transplant outcome in adults with acute leukaemia: a comparison between unrelated bone marrow, peripheral blood and cord bloodblood. Bone Marrow Transplantation, 43, S5.
Eapen, M., Rubinstein, P., Zhang, M. J., et al. (2007). Outcomes of transplantation of unrelated donor umbilical cord blood and bone marrow in children with acute leukaemia: a comparison study. Lancet, 369, 1947–1954.
Barker, J. N., Weisdorf, D. J., DeFor, T. E., et al. (2005). Transplantation of 2 partially HLA-matched umbilical cord blood units to enhance engraftment in adults with hematologic malignancy. Blood, 105, 1343–1347.
Tse, W., Bunting, K. D., & Laughlin, M. J. (2008). New insights into cord blood stem cell transplantation. Current Opinion in Hematology, 15, 279–284.
Delaney, C., Gutman, J. A., & Appelbaum, F. R. (2009). Cord blood transplantation for haematological malignancies: conditioning regimens, double cord transplant and infectious complications. British Journal Haematology, 147, 207–216.
Verneris, M. R., Brunstein, C., DeFor, T. E., et al. (2005). Risk of Relapse (REL) after Umbilical Cord Blood Transplantation (UCBT) in Patients with acute leukemia: marked reduction in recipients of two units. ASH Annual Meeting Abstracts, 106, 305.
Verneris, M. R., Brunstein, C. G., Barker, J., et al. (2009). Relapse risk after umbilical cord blood transplantation: enhanced graft-versus-leukemia effect in recipients of 2 units. Blood, 114, 4293–4299.
Willemze, R., Rodrigues, C. A., Labopin, M., et al. (2009). KIR-ligand incompatibility in the graft-versus-host direction improves outcomes after umbilical cord blood transplantation for acute leukemia. Leukemia, 23, 492–500.
Lister, J., Gryn, J. F., McQueen, K. L., Harris, D. T., Rossetti, J. M., & Shadduck, R. K. (2007). Multiple unit HLA-unmatched sex-mismatched umbilical cord blood transplantation for advanced hematological malignancy. Stem Cells and Development, 16, 177–186.
Magro, E., Regidor, C., Cabrera, R., et al. (2006). Early hematopoietic recovery after single unit unrelated cord blood transplantation in adults supported by co-infusion of mobilized stem cells from a third party donor. Haematologica, 91, 640–648.
van de Ven, C., Ishizawa, L., Law, P., & Cairo, M. S. (1995). IL-11 in combination with SLF and G-CSF or GM-CSF significantly increases expansion of isolated CD34+ cell population from cord blood vs. adult bone marrow. Experimental Hematology, 23, 1289–1295.
Vormoor, J., Lapidot, T., Pflumio, F., et al. (1994). Immature human cord blood progenitors engraft and proliferate to high levels in severe combined immunodeficient mice. Blood, 83, 2489–2497.
Shpall, E. J., Quinones, R., Giller, R., et al. (2002). Transplantation of ex vivo expanded cord blood. Biology of Blood and Marrow Transplantation, 8, 368–376.
McNiece, I., Jones, R., Bearman, S. I., et al. (2000). Ex vivo expanded peripheral blood progenitor cells provide rapid neutrophil recovery after high-dose chemotherapy in patients with breast cancer. Blood, 96, 3001–3007.
De Lima, M., McMannis, J. D., Saliba, R., et al. (2008). Double Cord Blood Transplantation (CBT) with and without Ex-Vivo Expansion (EXP): a randomized. Controlled study. ASH Annual Meeting Abstracts, 112, 154.
Glimm, H., Oh, I. H., & Eaves, C. J. (2000). Human hematopoietic stem cells stimulated to proliferate in vitro lose engraftment potential during their S/G(2)/M transit and do not reenter G(0). Blood, 96, 4185–4193.
Ramirez, M., Segovia, J. C., Benet, I., et al. (2001). Ex vivo expansion of umbilical cord blood (UCB) CD34(+) cells alters the expression and function of alpha 4 beta 1 and alpha 5 beta 1 integrins. British Journal Haematology, 115, 213–221.
Zhai, Q. L., Qiu, L. G., Li, Q., et al. (2004). Short-term ex vivo expansion sustains the homing-related properties of umbilical cord blood hematopoietic stem and progenitor cells. Haematologica, 89, 265–273.
Domen, J., Cheshier, S. H., & Weissman, I. L. (2000). The role of apoptosis in the regulation of hematopoietic stem cells: overexpression of Bcl-2 increases both their number and repopulation potential. The Journal of Experimental Medicine, 191, 253–264.
Liu, B., Buckley, S. M., Lewis, I. D., Goldman, A. I., Wagner, J. E., & van der Loo, J. C. (2003). Homing defect of cultured human hematopoietic cells in the NOD/SCID mouse is mediated by Fas/CD95. Experimental Hematology, 31, 824–832.
Wang, L. S., Liu, H. J., Xia, Z. B., Broxmeyer, H. E., & Lu, L. (2000). Expression and activation of caspase-3/CPP32 in CD34(+) cord blood cells is linked to apoptosis after growth factor withdrawal. Experimental Hematology, 28, 907–915.
Holyoake, T. L., Alcorn, M. J., Richmond, L., et al. (1997). CD34 positive PBPC expanded ex vivo may not provide durable engraftment following myeloablative chemoradiotherapy regimens. Bone Marrow Transplantation, 19, 1095–1101.
Pecora, A. L., Stiff, P., Jennis, A., et al. (2000). Prompt and durable engraftment in two older adult patients with high risk chronic myelogenous leukemia (CML) using ex vivo expanded and unmanipulated unrelated umbilical cord blood. Bone Marrow Transplantation, 25, 797–799.
Gallacher, L., Murdoch, B., Wu, D. M., Karanu, F. N., Keeney, M., & Bhatia, M. (2000). Isolation and characterization of human CD34(-)Lin(-) and CD34(+)Lin(-) hematopoietic stem cells using cell surface markers AC133 and CD7. Blood, 95, 2813–2820.
Peichev, M., Naiyer, A. J., Pereira, D., et al. (2000). Expression of VEGFR-2 and AC133 by circulating human CD34(+) cells identifies a population of functional endothelial precursors. Blood, 95, 952–958.
Bonanno, G., Perillo, A., Rutella, S., et al. (2004). Clinical isolation and functional characterization of cord blood CD133+ hematopoietic progenitor cells. Transfusion, 44, 1087–1097.
Peled, T., Landau, E., Mandel, J., et al. (2004). Linear polyamine copper chelator tetraethylenepentamine augments long-term ex vivo expansion of cord blood-derived CD34+ cells and increases their engraftment potential in NOD/SCID mice. Experimental Hematology, 32, 547–555.
de Lima, M., McMannis, J., Gee, A., et al. (2008). Transplantation of ex vivo expanded cord blood cells using the copper chelator tetraethylenepentamine: a phase I/II clinical trial. Bone Marrow Transplantation, 41, 771–778.
Bug, G., Gul, H., Schwarz, K., et al. (2005). Valproic acid stimulates proliferation and self-renewal of hematopoietic stem cells. Cancer Research, 65, 2537–2541.
Hofmeister, C. C., Zhang, J., Knight, K. L., Le, P., & Stiff, P. J. (2007). Ex vivo expansion of umbilical cord blood stem cells for transplantation: growing knowledge from the hematopoietic niche. Bone Marrow Transplantation, 39, 11–23.
Delaney, C., Heimfeld, S., Brashem-Stein, C., Voorhies, H. Manger, RL., Bernstein, ID. Notch-mediated expansion of human cord blood progenitor cells capable of rapid myeloid reconstitution. Nat Med, 16:232-6.
Schofield, R. (1978). The relationship between the spleen colony-forming cell and the haemopoietic stem cell. Blood Cells, 4, 7–25.
Verfaillie, C. M. (2002). Hematopoietic stem cells for transplantation. Nature Immunology, 3, 314–317.
Fu, Y. S., Cheng, Y. C., Lin, M. Y., et al. (2006). Conversion of human umbilical cord mesenchymal stem cells in Wharton’s jelly to dopaminergic neurons in vitro: potential therapeutic application for Parkinsonism. Stem Cells, 24, 115–124.
Sarugaser, R., Lickorish, D., Baksh, D., Hosseini, M. M., & Davies, J. E. (2005). Human umbilical cord perivascular (HUCPV) cells: a source of mesenchymal progenitors. Stem Cells, 23, 220–229.
Bakhshi, T., Zabriskie, R. C., Bodie, S., et al. (2008). Mesenchymal stem cells from the Wharton’s jelly of umbilical cord segments provide stromal support for the maintenance of cord blood hematopoietic stem cells during long-term ex vivo culture. Transfusion, 48, 2638–2644.
Robinson, S. N., Ng, J., Niu, T., et al. (2006). Superior ex vivo cord blood expansion following co-culture with bone marrow-derived mesenchymal stem cells. Bone Marrow Transplantation, 37, 359–366.
McNiece, I., Harrington, J., Turney, J., Kellner, J., & Shpall, E. J. (2004). Ex vivo expansion of cord blood mononuclear cells on mesenchymal stem cells. Cytotherapy, 6, 311–317.
Ning, H., Yang, F., Jiang, M., et al. (2008). The correlation between cotransplantation of mesenchymal stem cells and higher recurrence rate in hematologic malignancy patients: outcome of a pilot clinical study. Leukemia, 22, 593–599.
Ball, L. M., Bernardo, M. E., Roelofs, H., et al. (2007). Cotransplantation of ex vivo expanded mesenchymal stem cells accelerates lymphocyte recovery and may reduce the risk of graft failure in haploidentical hematopoietic stem-cell transplantation. Blood, 110, 2764–2767.
Macmillan, M. L., Blazar, B. R., DeFor, T. E., & Wagner, J. E. (2009). Transplantation of ex-vivo culture-expanded parental haploidentical mesenchymal stem cells to promote engraftment in pediatric recipients of unrelated donor umbilical cord blood: results of a phase I-II clinical trial. Bone Marrow Transplantation, 43, 447–454.
Gonzalo-Daganzo, R., Regidor, C., Martin-Donaire, T., et al. (2009). Results of a pilot study on the use of third-party donor mesenchymal stromal cells in cord blood transplantation in adults. Cytotherapy, 11, 278–288.
Nauta, A. J., & Fibbe, W. E. (2007). Immunomodulatory properties of mesenchymal stromal cells. Blood, 110, 3499–3506.
Noel, D., Djouad, F., Bouffi, C., Mrugala, D., & Jorgensen, C. (2007). Multipotent mesenchymal stromal cells and immune tolerance. Leukaemia & Lymphoma, 48, 1283–1289.
Le Blanc, K., Frassoni, F., Ball, L., et al. (2008). Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet, 371, 1579–1586.
von Bonin, M., Stolzel, F., Goedecke, A., et al. (2009). Treatment of refractory acute GVHD with third-party MSC expanded in platelet lysate-containing medium. Bone Marrow Transplantation, 43, 245–251.
Resnick, I. B., Jr., Stepensky, P., Shapira, M. Y., et al. (2009). Treatment of severe acute graft-versus-host disease with mesenchymal stromal cells. ASH Annual Meeting Abstracts, 114, 2249.
Calvi, L. M., Adams, G. B., Weibrecht, K. W., et al. (2003). Osteoblastic cells regulate the haematopoietic stem cell niche. Nature, 425, 841–846.
Ballen, K. K., Shpall, E. J., Avigan, D., et al. (2007). Phase I trial of parathyroid hormone to facilitate stem cell mobilization. Biology of Blood and Marrow Transplantation, 13, 838–843.
Frassoni, F. Intra-bone route of administration offers new perspectives for safer transplantation of hematopoietic stem cells. Cytotherapy, 12:5-6.
Brunstein, C. G., Barker, J. N., Weisdorf, D. J., et al. (2009). Intra-BM injection to enhance engraftment after myeloablative umbilical cord blood transplantation with two partially HLA-matched units. Bone Marrow Transplantation, 43, 935–940.
Kollet, O., Dar, A., & Lapidot, T. (2007). The multiple roles of osteoclasts in host defense: bone remodeling and hematopoietic stem cell mobilization. Annual Review of Immunology, 25, 51–69.
Miyazaki, O., Nishimura, G., Okamoto, R., et al. (2009). Induction of systemic bone changes by preconditioning total body irradiation for bone marrow transplantation. Pediatric Radiology, 39, 23–29.
Ceradini, D. J., Kulkarni, A. R., Callaghan, M. J., et al. (2004). Progenitor cell trafficking is regulated by hypoxic gradients through HIF-1 induction of SDF-1. Natural Medicines, 10, 858–864.
De Falco, E., Porcelli, D., Torella, A. R., et al. (2004). SDF-1 involvement in endothelial phenotype and ischemia-induced recruitment of bone marrow progenitor cells. Blood, 104, 3472–3482.
Avigdor, A., Goichberg, P., Shivtiel, S., et al. (2004). CD44 and hyaluronic acid cooperate with SDF-1 in the trafficking of human CD34+ stem/progenitor cells to bone marrow. Blood, 103, 2981–2989.
Reca, R., Mastellos, D., Majka, M., et al. (2003). Functional receptor for C3a anaphylatoxin is expressed by normal hematopoietic stem/progenitor cells, and C3a enhances their homing-related responses to SDF-1. Blood, 101, 3784–3793.
Wysoczynski, M., Reca, R., Ratajczak, J., et al. (2005). Incorporation of CXCR4 into membrane lipid rafts primes homing-related responses of hematopoietic stem/progenitor cells to an SDF-1 gradient. Blood, 105, 40–48.
Ratajczak, M. Z., Reca, R., Wysoczynski, M., et al. (2004). Transplantation studies in C3-deficient animals reveal a novel role of the third complement component (C3) in engraftment of bone marrow cells. Leukemia, 18, 1482–1490.
Christopherson, K. W., Hangoc, G., & Broxmeyer, H. E. (2002). Cell Surface Peptidase CD26/Dipeptidylpeptidase IV Regulates CXCL12/Stromal Cell-Derived Factor-1α-Mediated Chemotaxis of Human Cord Blood CD34+ Progenitor Cells. Journal of Immunology, 169, 7000–7008.
Acknowledgement
We would like to thank Prof. Essie Kariv for her generous help in preparing this manuscript.
Conflict of Interests
The authors declare no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stanevsky, A., Shimoni, A., Yerushalmi, R. et al. Cord Blood Stem Cells for Hematopoietic Transplantation. Stem Cell Rev and Rep 7, 425–433 (2011). https://doi.org/10.1007/s12015-010-9183-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-010-9183-9