Abstract
Muscular dystrophy (MD) refers to a group of more than 30 genetically and clinically heterogeneous disorders, characterized by progressive weakness and degeneration of the skeletal muscles that control movement. To date, MD is still incurable but increasing evidence suggests that stem cells might represent a therapeutic option in the future. This review will outline recent progress in this field involving the use of adult and embryonic stem cells. We will discuss in further detail the nature of these cells and their distinct biological properties which lead to their unique advantages and disadvantages in regard to therapeutic application.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Muscular dystrophies are heterogeneous neuromuscular disorders of inherited origin, including X-linked recessive as in Duchenne or Becker muscular dystrophy (MD), autosomal dominant as in Facioscapulohumeral MD, myotonic dystrophy and limb–girdle MD type 1, or autosomal recessive as in limb–girdle MD type 2 (reviewed in [1]). Duchenne muscular dystrophy (DMD) is the most common affecting 1 out of every 3,500 live born boys. It is caused by mutations in the dystrophin gene, resulting in the absence of dystrophin in skeletal muscle and other tissues [2, 3]. In the early phase of the disease, skeletal muscle of DMD patients is characterized by an ongoing process of degeneration and regeneration which is later followed by exhaustion of its regenerative capacity, fibrosis, and eventual disruption of the muscle tissue architecture. Clinically it is characterized by progressive muscle weakness and atrophy, leading patients to be confined to a wheelchair before the age of 12 and eventual death due to respiratory insufficiency [1]. No effective therapy is available at present. Several studies in the last two decades have exploited the potential of cell based therapies to promote muscle regeneration. Initial studies focused on the transplantation of adult myoblasts, which through cell fusion, lead to the development of new or hybrid muscle fibers [4–6]. However, this approach is hindered by the poor survival and limited migratory ability of injected myoblasts [5, 7], and produced discouraging results in early clinical trials [8–11]. Rather than myoblasts, stem cells, which are endowed with self-renewal and differential potential, would be preferable for therapeutic applications.
Embryonic and adult stem cells differ significantly in regard to their differentiation potential and in vitro expansion capability. While adult stem cells constitute a reservoir for tissue regeneration throughout the adult life, they are tissue-specific and possess limited capacity to be expanded ex vivo. Embryonic Stem (ES) cells are derived from the inner cell mass of blastocyst embryos and, by definition, are capable of unlimited in vitro self-renewal and have the ability to differentiate into any cell type of the body. While transplanted adult stem cells are primed to respond immediately to tissue damage by appropriate tissue specific differentiation, transplantation of ES cells in the undifferentiated state produces teratomas.
Muscle Adult Stem Cells
Satellite Cells
The regeneration ability of adult skeletal muscle has conventionally been attributed to the satellite cell: a quiescent population of muscle precursors located beneath the basal lamina of each myofiber [12]. In mature skeletal muscle these cells are normally in a dormant state, being activated in response to muscle damage or disease. Activated satellite cells can proliferate and fuse to existing necrotic muscle fibers or together to form new myotubes with central nuclei. Satellite cells can be defined by their position, beneath the basal lamina [12], and in the last decade phenotypically, by the expression of specific surface markers including M-cadherin [13], c-Met [13], CD34 [14], syndecan-4 [15], and more importantly, Pax7, a paired box homeodomain-containing transcription factor [16, 17]. It has been demonstrated that following the activation and proliferation of satellite cells, a small subset of cells do not undergo the terminal differentiation pathway, but retain the ability to go back to a quiescent state and thus preserve the satellite cell pool [18–20]. Recent experiments by Rudnicki’s group [21] revealed an heterogeneity within the satellite compartment and that, the apical–basal location of a daughter cell determines whether a cell will self-renew (basal position—in contact with basal lamina) or differentiate (apical—pushed away from basal lamina). Based on these characteristics of self-renewal and ability to regenerate injured muscle, satellite cells are quite well accepted as the stem cells of adult skeletal muscle. In terms of engraftment, it has been reported that the transplantation of single intact myofibers, containing in average 22 satellite cells, into injured muscles results not only in efficient muscle regeneration but also in expansion of the satellite cell pool (tenfold) [22]. Similar outcome has been reported with the transplantation of FACS-purified Pax3-GFP+ satellite cells of diaphragm [23] or myofiber-associated CD45−Sca-1−Mac-1−CXCR4+ β1-integrin+ cells [24]. Although to date, improvement in muscle contractility has not been reported after transplantation of satellite cells, they represent a potential cell source for skeletal muscle regeneration. The major challenge in applying these cells for therapeutic purposes is to obtain enough cells since activation and in vitro expansion diminishes their engraftment ability [23]. A similar phenomenon has been observed for murine and human hematopoietic stem cells [25–27].
Muscle Derived Stem Cells (MDSCs)
It has been proposed that satellite cells can move between adjacent muscle fibers and therefore reside for some time in the interstitial space during growth and regeneration. In addition to these satellite cells, a distinct population named muscle derived stem cells (MDSCs) has been identified in the muscle interstitial space [28]. This cell population, isolated by using a pre-plating method which removes adherent cells, can be expanded in vitro for up to 30 passages while maintaining their phenotype in terms of both surface markers (Sca-1+ CD34+ Desminlow) and myogenic potential [7, 29]. Contribution to muscle regeneration in dystrophic mice has been observed following their local [7, 29] or systemic injection [30]. However, there has been no evidence of functional improvement in the muscles from dystrophic mice treated with MDSCs [31].
Muscle Side Population (SP) Cells
Side population (SP) cells were first identified in the bone marrow based on their capacity to uptake the vital dye Hoechst 33342 [32], and BM SP cells are known to be highly enriched for long-term repopulating hematopoetic stem cells [32, 33]. Since then, this population has been identified in several tissues [34–38] including skeletal muscle [39, 40]. Muscle SP cells are Sca-1+ Lin− and although unable to undergo myogenesis in vitro [40], there is evidence for their in vivo participation in skeletal muscle regeneration [39–41]. Recently one group has used SP cells isolated from muscles of mdx mice, a mouse model for DMD that lacks dystrophin, as vehicles to deliver a micro-dystrophin transgene back to mdx recipient mice [41]. Although engraftment was observed with freshly purified as well as expanded SP cells after their intra-arterial transplantation, dystrophin restoration was not obtained at significant levels [41].
Bone Marrow (BM) Stem Cells
Bone marrow contains hematopoietic stem cells (HSCs), which give rise to all blood lineages, and a population of stromal cells, known as mesenchymal stem cells (MSCs), which give rise to fat, cartilage, and bone precursors. A decade ago Ferrari and colleagues demonstrated for the first time that cells present in the BM were able to migrate into areas of skeletal muscle degeneration and participate in the regeneration process [42]. Although adherent and non-adherent cell fractions were tested, it was not clear which cell population within BM was endowed with regenerative potential. This initial study was followed by a flourish of papers [39, 43–45] reporting dystrophin restoration following the transplantation of BM-derived cells into mdx mice, however when enumerated engraftment was not found therapeutically relevant (below 5%). Subsequent lineage tracing studies of single hematopoietic stem cells demonstrated that BM contribution to the muscle compartment does not occur through a myogenic stem cell intermediate but through stochastic cell fusion of circulating myeloid cells with mature myofibers [46]. However this study does not exclude the possible involvement of MSCs in muscle repair. In vivo myogenic potential was first attributed to MSCs by Saito and colleagues more than a decade ago [47], and has extended to MSC preparations isolated from human adipose tissue [48] and adult human synovial membrane [49]. Recent work by Dezawa and colleagues with human MSCs show that activated Notch along with specific cytokines [50] induce efficient generation of skeletal muscle progenitors. Interestingly, the Notch-induced myogenic population contained Pax7+ cells that contributed to subsequent regeneration of muscle upon repetitive damage without additional cell transplantation [50]. Because functional studies were not performed, it is unknown whether engrafted cells improve muscle contractility. We have recently demonstrated that Pax3, the master regulator of the embryonic myogenic program, is able to induce the in vitro differentiation of a murine BM mesenchymal stem cell line toward the muscle lineage [51]. However, when we applied this approach to adult primary human MSCs, despite activation of the myogenic program and reasonable engraftment, Pax3-modified MSCs failed to promote functional recovery (Gang et al, unpublished data). These results put in question the validity of MSCs for therapeutic purposes in neuromuscular disorders.
Peripheral Blood AC133+ Cells
AC133 (also known as prominin-1 or CD133) is a human antigen associated with the hematopoietic [52, 53], endothelial [54, 55] and neural [56] lineages. It has recently being demonstrated that AC133 also identifies a population of cells within human peripheral blood that can undergo myogenic differentiation under specific in vitro culture conditions. When co-cultured with C2C12 mouse myoblasts or exposed to Wnt-producing fibroblasts, they were induced to express M-cadherin, Pax7, CD34 and Myf5 [57]. Following intra-muscular or intra-arterial transplantation into scid/mdx mice, these cells gave rise to donor-derived (human) dystrophin+ myofibers. Although the levels of engraftment were somewhat limited, there was functional improvement in regenerated single myofibers of treated mice when compared to controls [57]. Nevertheless, the true identity of these cells is still unclear. It is difficult to concur that AC133+ cells might represent circulating myogenic progenitors since the majority (>97%) are positive for CD45 (a pan-hematopoietic marker). It is reasonable to hypothesize that these cells are equivalent to the fusogenic circulating myeloid cells described by Camargo and colleagues for bone marrow, as discussed above [46]. Although debatably premature, CD133+ cells have already been submitted to a double-blind phase I clinical trial involving eight boys with DMD, which apparently resulted in no side effects [58]. More recently the same authors have reported encouraging results in which DMD donor-derived AC133+ cells, manipulated to promote exon skipping to bypass DMD frameshift mutation, lead to significant recovery of transplanted scid/mdx mice [59].
Vessel Derived Stem Cells (Meso-angioblasts)
Nearly a decade ago, Cossu’s group reported the identification of a mesodermal stem cell population in the wall of the mouse embryonic dorsal aorta endowed with endothelial and myogenic potential [60]. Expression of myogenic markers including CD34, M-cadherin, Myf5, MyoD, and desmin was observed when dorsal aorta explant cultures were subjected to in vitro differentiation [60]. Further studies with these dorsal aorta-derived cells revealed their extensive in vitro proliferative and multilineage differentiation potential which led the authors to name them as meso-angioblasts [61]. Since then, meso-angioblasts have become a major focus of interest by this group, which has recently identified cells with similar properties in post-natal tissues of dogs [62] and humans [63]. Based on their perivascular location, the “meso-angioblast” of adult vasculature has been designated as a type of pericyte. Intra-arterial transplantation of adult meso-angioblasts into dystrophic mice [63, 64] or GRMD dogs (a canine model of DMD) [62], results in extensive and widespread engraftment, which is accompanied by functional improvement. These findings lay the groundwork for clinical studies using meso-angioblasts/pericytes to treat patients with muscular dystrophy.
ES Cells: Pluripotency
The tremendous self-renewal and multilineage differentiation potential of ES cells endow them with unique advantages for cell therapies and regenerative medicine. Until the 1980s, experiments were limited to the use of tumor-derived mouse embryonal carcinoma (EC) cells, which could participate in the development of completely normal adult mice, creating mosaic mice when transplanted into blastocyst embryos. The tumor-derived cells contributed not only to somatic cells, but also to the germline [65]. ES cells were first obtained from blastocyst embryos in 1981 [66, 67] and have since been used extensively in mouse transgenic research. When injected into syngeneic or immunocompromised adult mice, it was observed that ES cells generate teratomas containing all three germ layers—ectoderm, mesoderm and endoderm, indicating their pluripotency [67]. This strategy was applied again 17 years later by Thomson and colleagues to demonstrate the pluripotency of human ES cell lines [68]. It was further demonstrated that mouse ES cells, like their malignant counterparts (EC cells), have the ability to contribute to somatic tissues as well as germ cells when reintroduced into blastocysts, even after being cultured and manipulated in vitro [69].
ES Cells: Tissues in the Petri Dish
An important observation that was originally made with EC cells was the capacity of these cells to differentiate into multilayered cystic structures under suspension conditions. These cellular aggregates known as embryoid bodies (EBs) mimic many temporal events of early embryogenesis [70, 71]. This process is also recapitulated by differentiating ES cells, which initially develop extra-embryonic endoderm, followed by primitive ectoderm, and subsequent specification of mesoderm [72, 73]. Using this system, several groups have been able to differentiate ES cells into a variety of specialized cell types including, hematopoietic precursors [74–78], pancreatic β-like cells [79–81], hepatocytes [82, 83], neural cells [84, 85], germ cells [86–88], cardiomyocytes [75], [89–91], smooth muscle [92], and skeletal muscle [93–96], which we will discuss in further detail.
ES Cells and Skeletal Muscle Regeneration
The first evidence for the presence of skeletal myogenic progenitors within EBs was reported by Rohwedel and colleagues more than a decade ago [93]. This study as well as a report involving EC cells [97] showed that activation of the myogenic program during EB development recapitulates mouse embryogenesis as indicated by the sequential expression of myogenic regulatory factors (MRFs). However the readout in both these reports was confined to gene expression analyses of EBs and their outgrowths, or morphological assessment of rare differentiated cells [93, 97]. Despite these initial studies, there has been very limited information concerning the in vivo regenerative capability of ES-derived cells. One group has reported the presence of sporadic donor-derived (dystrophin+) myofibers following the transplantation of differentiating mouse ES cells into two mdx mice [95]. In order to obtain this result, it was necessary to co-culture day 7 EBs on a preparation of primary mouse muscle precursor cells for 4 days. A more recent study has reported the engraftment of human ES-derived myoblasts in SCID/Beige mice, as evidenced by the presence of human nuclear antigen and laminin. In these experiments, ES cells were subjected to mesenchymal differentiation under selective culture conditions and FACS purification [94]. Despite the potential benefits of using ES cells to treat muscular dystrophy, these few reports do not provide a feasible method to generate large quantities of myogenic progenitors for skeletal muscle regeneration.
While early events of pre-implantation embryogenesis are recapitulated reasonably well in EBs, later events, including the generation of somites, neural tube or notochord, all required for inducing the proper patterning of paraxial mesoderm to induce myogenesis does not occur in a recognizable way. We have recently reported that it is possible to bypass this defective environment and directly pattern paraxial mesoderm by over-expressing Pax3, the master regulator of the myogenic program [96]. For an optimal control of Pax3 over-expression, we developed an ES cell line in which expression of Pax3 can be induced by doxycycline (dox). Our results showed that expression of Pax3 during EB differentiation enhanced paraxial mesoderm, and cells with phenotypic and gene expression profile equivalent to myogenic progenitors within this population.
Transplantation of bulk Pax3-induced cells led to teratoma formation, indicating the presence of residual undifferentiated cells [96]. We therefore developed a cell purification strategy based on surface markers present on paraxial or lateral plate mesoderm. By sorting for PDGFαR, a paraxial mesoderm marker, and absence of Flk-1, a lateral plate mesoderm marker, we were able to purify a teratoma-free cell population enriched for early muscle precursors with significant potential for muscle regeneration. Following purification and in vitro expansion, these cells generated a homogenous monolayer of myogenic progenitors expressing Pax3 and Myf5 as well as CD44, CD29, M-cadherin, CXCR4 and syndecam-4, surface markers associated with satellite cells [13–15], or with the migration and differentiation of myogenic progenitors [98]. When this population was injected locally or systemically into mdx mice, there was significant restoration of dystrophin levels, which was accompanied by a boost in the contractile strength of engrafted muscles [96].
The ability to generate cells with in vivo muscle regenerative potential in culture and systemically transfer them to recipients is an important step towards the therapeutic application of ES cell-derived cells.
Deriving Pluripotent Stem Cells from Somatic Cells
In order to generate patient and disease specific stem cells, many groups have been working on methods of reprogramming somatic cells to an undifferentiated ES-like pluripotent state. Reprogrammed cells from MD patients eliminate the risks of immune rejection of the grafted tissue, a common complication of allogeneic transplantations, and open up new possibilities for drug development [99]. Replicating pioneering work using amphibian embryos [100, 101], cloning in mammals was first achieved through nuclear transfer of embryonic blastomeres of sheep [102] and cattle [103] in the late 1980s. A decade later, Ian Wilmut and colleagues applied somatic cell nuclear transfer (SCNT) to clone a mammal from differentiated adult cells—the famous sheep “Dolly”—[104], proving that a differentiated mammalian somatic cell nucleus could be reprogrammed to an embryonic state. Since then, many other laboratories have claimed to produce live offspring resultant of fetal and adult somatic cells in other species, such as mice [105], cattle [106], pigs [107], goats [108], rabbits [109], and cats [110]. In addition, interesting experiments have been able to differentiate mouse nuclear transfer ES (ntES) cells into several tissues, including HSCs [111], neurons and gametes [112]. Despite the advances, this methodology has been overtaken by a recent flow of papers using genetic manipulation to transiently overexpress pluripotency factors in somatic cells—instead of NT—to achieve pluripotent stem cells from somatic cells. This method was pioneered by Shinya Yamanaka’s team, using retroviral expression of four transcription factors, Oct4, Sox2, Klf4 and c-Myc [113]. iPS cells share many similarities to ES cells, including the formation of teratomas following their subcutaneous transplantation into nude mice. These cells also contributed to many different cell types in mid-gestation embryos however, these embryos failed to complete development, indicating that their pluripotency was slightly hampered when compared to ES cells [113]. In 2007, this same group demonstrated that the reprogramming potential retained by these four factors could be translated from mouse to humans [114]. Following Yamanaka’s discoveries, many other groups have further studied the potential of iPS cells, contributing to a fast advance of this new and promising field of investigation [113–119]. Therapeutic application of mouse iPS cells has already been demonstrated in mouse models of sickle cell anemia [120] and Parkinson’s disease [121], and no doubt are being attempted in many other mouse models, including MD.
Conclusions and Future Prospects
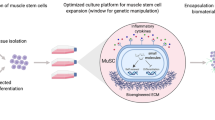
Even though there has been great advance in the generation of cell populations endowed with in vivo myogenic potential, several questions remain to be answered before any of these cell preparations can be moved into clinical trials. Based on their unique characteristics and in vivo skeletal muscle regeneration potential, several stem cell populations discussed in this review (Fig. 1) are excellent clinical candidates, including mesoangioblasts, PB AC133+ cells, satellite cells, and ES-derived myogenic progenitors. Since mesoangioblasts have been extensively tested in mouse models of disease, and more recently even in dystrophic dogs, it is reasonable to assume that they are on top of the list. However, the fact that the derivation and use of these cells has not yet been replicated by other labs brings caution. Because of their easy accessibility, PB AC133+ cells, although not so extensively studied as mesoangioblasts, would come in second in the list, especially as vehicles for gene therapy. Again, independent groups must obtain similar results using animal models before moving to clinical trials. At this point, despite their potential, satellite cell preparations are not feasible for therapeutic applications. Approaches to expand these cells ex vivo while retaining their regenerative potential are required. Last but not least, ES-derived progenitors which possess tremendous self-renewal and regenerative potential, but are still in their infancy days. One could assume that the combination of iPS technology with the lineage-specific reprogramming using Pax3 would be ideal for the generation of patient-specific myogenic progenitors for musculoskeletal diseases. However before this can be accomplished, many milestones have to be overcome, including (1) optimization of engraftment such that the satellite cell compartment is significantly repopulated, (2) development of a protocol to obtain similar populations of muscle precursors from human ES cells, and (3) activation of the Pax3 regulatory cascade without genetic manipulation, possibly through the use of protein transduction.
References
Emery, A. E. (2002). The muscular dystrophies. Lancet, 359, 687–695.
Koenig, M., Hoffman, E. P., Bertelson, C. J., Monaco, A. P., Feener, C., & Kunkel, L. M. (1987). Complete cloning of the Duchenne muscular dystrophy (DMD) cDNA and preliminary genomic organization of the DMD gene in normal and affected individuals. Cell, 50, 509–517.
Hoffman, E. P., Brown, R. H. J., & Kunkel, L. M. (1987). Dystrophin: The protein product of the Duchenne muscular dystrophy locus. Cell, 51, 919–928.
Brussee, V., Tardif, F., Roy, B., Goulet, M., Sebille, A., & Tremblay, J. P. (1999). Successful myoblast transplantation in fibrotic muscles: no increased impairment by the connective tissue. Transplantation, 67, 1618–1622.
Gussoni, E., Blau, H. M., & Kunkel, L. M. (1997). The fate of individual myoblasts after transplantation into muscles of DMD patients. Nature Medicine, 3, 970–977.
Partridge, T. A., Morgan, J. E., Coulton, G. R., Hoffman, E. P., & Kunkel, L. M. (1989). Conversion of mdx myofibers from dystrophin-negative to positive by injection of normal myoblasts. Nature, 337, 176–179.
Qu, Z., Balkir, L., van Deutekom, J. C., Robbins, P. D., Pruchnic, R., & Huard, J. (1998). Development of approaches to improve cell survival in myoblast transfer therapy. Journal of Cell Biology, 142, 1267.
Mendell, J. R., Kissel, J. T., Amato, A. A., King, W., Signore, L., Prior, T. W., et al. (1995). Myoblast transfer in the treatment of Duchenne’s muscular dystrophy. New England Journal of Medicine, 333, 832–838.
Partridge, T., Lu, Q. L., Morris, G., & Hoffman, E. (1998). Is myoblast transplantation effective? Nature Medicine, 4, 1208–1209.
Tremblay, J. P., Malouin, F., Roy, R., Huard, J., Bouchard, J. P., Satoh, A., et al. (1993). Results of a triple blind clinical study of myoblast transplantations without immunosuppressive treatment in young boys with Duchenne muscular dystrophy. Cell Transplantation, 2, 99–112.
Vilquin, J. T. (2005). Myoblast transplantation: Clinical trials and perspectives. Acta Myologica, 24, 119–27.
Mauro, A. (1961). Satellite cells of skeletal muscle fibres. Journal of Biophysical and Biochemical Cytology, 9, 493–496.
Cornelison, D. D., & Wold, B. J. (1997). Single-cell analysis of regulatory gene expression in quiescent and activated mouse skeletal muscle satellite cells. Developments in Biologicals, 191, 270–283.
Sherwood I, R., Christensen, J. L., Weissman, I. L., & Wagers, A. J. (2004). Determinants of skeletal muscle contributions from circulating cells, bone marrow cells, and hematopoietic stem cells. Stem Cells, 22, 1292–304.
Cornelison, D. D., Filla, M. S., Stanley, H. M., Rapraeger, A. C., & Olwin, B. B. (2001). Syndecan-3 and syndecan-4 specifically mark skeletal muscle satellite cells and are implicated in satellite cell maintenance and muscle regeneration. Developments in Biologicals, 239, 79–94.
Oustanina, S., Hause, G., & Braun, T. (2004). Pax7 directs postnal renewal and propagation of myogenic satellite cells but not their specification. EMBO Journal, 23, 3430–3439.
Seale, P., Sabourin, L. A., Girgis-Gabardo, A., Mansouri, A., Gruss, P., & Rudnicki, M. A. (2000). Pax7 is required for the specification of myogenic satellite cells. Cell, 102, 777–786.
Zammit, P. S., Golding, J. P., Nagata, Y., Hudon, V., Partridge, T. A., & Beauchamp, J. R. (2004). Muscle satellite cells adopt divergent fates: a mechanism for self-renewal? Journal of Cell Biology, 166, 347–357.
Halevy, O., Piestun, Y., Allouh, M. Z., Rosser, B. W., Rinkevich, Y., Reshef, R., et al. (2004). Pattern of Pax7 expression during myogenesis in the posthatch chicken establishes a model for satellite cell differentiation and renewal. Developmental Dynamics, 231, 489–502.
Olguin, H. C., & Olwin, B. B. (2004). Pax-7 up-regulation inhibits myogenesis and cell cycle progression in satellite cells: a potential mechanism for self-renewal. Developments in Biologicals, 275, 375–388.
Kuang, S., Kuroda, K., Le Grand, F., & Rudnicki, M. A. (2007). Asymmetric self-renewal and commitment of satellite stem cells in muscle. Cell, 129, 999–1010.
Collins, C. A., Olsen, I., Zammit, P. S., Heslop, L., Petrie, A., Partridge, T. A., et al. (2005). Stem cell function, self-renewal, and behavioral heterogeneity of cells from the adult muscle satellite cell niche. Cell, 122, 289–301.
Montarras, D., Morgan, J., Collins, C., Relaix, F., Zaffran, S., Cumano, A., et al. (2005). Direct isolation of satellite cells for skeletal muscle regeneration. Science, 309, 2064–2067.
Sherwood, R. I., Christensen, J. L., Conboy, I. M., Conboy, M. J., Rando, T. A., Weissman, I. L., et al. (2004). Isolation of adult mouse myogenic progenitors: functional heterogeneity of cells within and engrafting skeletal muscle. Cell, 119, 543–54.
van der Loo, J. C., & Ploemacher, R. E. (1995). Marrow- and spleen-seeding efficiencies of all murine hematopoietic stem cell subsets are decreased by preincubation with hematopoietic growth factors. Blood, 85, 2598–2606.
Traycoff, C. M., Cornetta, K., Yoder, M. C., Davidson, A., & Srour, E. F. (1996). Ex vivo expansion of murine hematopoietic progenitor cells generates classes of expanded cells possessing different levels of bone marrow repopulating potential. Experimental Hematology, 24, 299–306.
Guenechea, G., Segovia, J. C., Albella, B., Lamana, M., Ramírez, M., Regidor, C., Fernández, M. N., Bueren, J. A., et al. (1999). Delayed engraftment of nonobese diabetic/severe combined immunodeficient mice transplanted with ex vivo-expanded human CD34(+) cord blood cells. Blood, 93, 1097–1105.
Qu-Petersen, Z., Deasy, B., Jankowski, R., Ikezawa, M., Cummins, J., Pruchnic, R., et al. (2002). Identification of a novel population of muscle stem cells in mice: Potential for muscle regeneration. Journal of Cell Biology, 157, 851–864.
Deasy, B. M., Jankowski, R. J., & Huard, J. (2001). Muscle-derived stem cells: Characterization and potential for cell-mediated therapy. Blood Cells, Molecules and Disease, 27, 924–933.
Torrente, Y., Tremblay, J. P., Pisati, F., Belicchi, M., Rossi, B., Sironi, M., et al. (2001). Intraarterial injection of muscle-derived CD34(+)Sca-1(+) stem cells restores dystrophin in mdx mice. Journal of Cell Biology, 152, 335–348.
Mueller, G. M., O’Day, T., Watchko, J. F., & Ontell, M. (2002). Effect of injecting primary myoblasts versus putative muscle-derived stem cells on mass and force generation in mdx mice. Human Gene Therapy, 13, 1081–1090.
Goodell, M. A., Brose, K., Paradis, G., Conner, A. S., Mulligan, R. C., et al. (1996). Isolation and functional properties of murine hematopoietic stem cells that are replicating in vivo. Journal of Experimental Medicine, 183, 1797–806.
Goodell, M. A., Rosenzweig, M., Kim, H., Marks, D. F., DeMaria, M., & Paradis, G. (1997). Dye efflux studies suggest that hematopoietic stem cells expressing low or undetectable levels of CD34 antigen exist in multiple species. Nature Medicine, 3, 1337–1345.
Hierlihy, A. M., Seale, P., Lobe, C. G., Rudnicki, M. A., & Megeney, L. A. (2002). The post-natal heart contains a myocardial stem cell population. FEBS Letters, 530, 239–243.
Inowa, T., Hishikawa, K., Takeuchi, T., Kitamura, T., & Fujita, T. (2008). Isolation and potential existence of side population cells in adult human kidney. International Journal of Urology, 15, 272–274.
Welm, B. E., Tepera, S. B., Venezia, T., Graubert, T. A., Rosen, J. M., & Goodell, M. A. (2002). Sca-1(pos) cells in the mouse mammary gland represent an enriched progenitor cell population. Developments in Biologicals, 245, 42–56.
Martin, C. M., Meeson, A. P., Robertson, S. M., Hawke, T. J., Richardson, J. A., Bates, S., et al. (2004). Persistent expression of the ATP-binding cassette transporter, Abcg2, identifies cardiac SP cells in the developing and adult heart. Developments in Biologicals, 265, 262–75.
Tsuchiya, A., Heike, T., Baba, S., Fujino, H., Umeda, K., Matsuda, Y., et al. (2007). Long-term culture of postnatal mouse hepatic stem/progenitor cells and their relative developmental hierarchy. Stem Cells, 25, 895–902.
Gussoni, E., Soneoka, Y., Strickland, C. D., Buzney, E. A., Khan, M. K., Flint, A. F., et al. (1999). Dystrophin expression in the mdx mouse restored by stem cell transplantation. Nature, 401, 390–394.
Asakura, A., Seale, P., Girgis-Gabardo, A., & Rudnicki, M. A. (2002). Myogenic specification of side population cells in skeletal muscle. Journal of Cell Biology, 159, 123–134.
Bachrach, E., Perez, A. L., Choi, Y. H., Illigens, B. M., Jun, S. J., del Nido, P., et al. (2006). Muscle engraftment of myogenic progenitor cells following intraarterial transplantation. Muscle Nerve, 34, 44–52.
Ferrari, G., Cusella-De Angelis, G., Coletta, M., Paolucci, E., Stornaiuolo, A., Cossu, G., et al. (1998). Muscle regeneration by bone marrow-derived myogenic progenitors. Science, 279, 1528–1530.
Bittner, R. E., Schofer, C., Weipoltshammer, K., Ivanova, S., Streubel, B., Hauser, E., et al. (1999). Recruitment of bone-marrow-derived cells by skeletal and cardiac muscle in adult dystrophic mdx mice. Anatomy and Embryology (Berl), 199, 391–396.
Fukada, S., Miyagoe-Suzuki, Y., Tsukihara, H., Yuasa, K., Higuchi, S., Ono, S., et al. (2002). Muscle regeneration by reconstitution with bone marrow or fetal liver cells from green fluorescent protein-gene transgenic mice. Journal of Cell Science, 115, 1285–1293.
Ferrari, G., Stornaiuolo, A., & Mavilio, F. (2001). Failure to correct murine muscular dystrophy. Nature, 411, 1014–1015.
Camargo, F. D., Green, R., Capetanaki, Y., Jackson, K. A., & Goodell, M. A. (2003). Single hematopoietic stem cells generate skeletal muscle through myeloid intermediates. Nature Medicine, 9, 1520–1527.
Saito, T., Dennis, J. E., Lennon, D. P., Young, R. G., & Caplan, A. I. (1995). Myogenic expression of mesenchymal stem cells within myotubes of mdx mice in vitro and in vivo. Tissue Engineering, 1, 327–243.
Rodriguez, A. M., Pisani, D., Dechesne, C. A., Turc-Carel, C., Kurzenne, J. Y., Wdziekonski, B., et al. (2005). Transplantation of a multipotent cell population from human adipose tissue induces dystrophin expression in the immunocompetent mdx mouse. Journal of Experimental Medicine, 201, 1397–1405.
De Bari, C., Dell’Accio, F., Vandenabeele, F., Vermeesch, J. R., Raymackers, J. M., & Luyten, F. P. (2003). Skeletal muscle repair by adult human mesenchymal stem cells from synovial membrane. Journal of Cell Biology, 60, 909–918.
Dezawa, M., Ishikawa, H., Itokazu, Y., Yoshihara, T., Hoshino, M., Takeda, S., et al. (2005). Bone marrow stromal cells generate muscle cells and repair muscle degeneration. Science, 309, 314–317.
Gang, E. J., Bosnakovski, D., Simsek, T., To, K., Perlingeiro, R. C. R. (2008). Pax3 activation promotes the differentiation of mesenchymal stem cells toward the myogenic lineage. Experimental Cell Research, doi:10.1016/j.yexcr.2008.02.016.
Yin, A. H., Miraglia, S., Zanjani, E. D., Almeida-Porada, G., Ogawa, M., Leary, A. G., et al. (1997). AC133, a novel marker for human hematopoietic stem and progenitor cells. Blood, 90(12), 5002–5012, Dec 15.
de Wynter, E. A., Buck, D., Hart, C., Heywood, R., Coutinho, L. H., & Clayton, A. (1998). CD34+AC133+cells isolated from cord blood are highly enriched in long-term culture-initiating cells, NOD/SCID-repopulating cells and dendritic cell progenitors. Stem Cells, 16, 387–396.
Peichev, M. N. A., Pereira, D., Zhu, Z., Lane, W. J., Williams, M., Oz, M. C., et al. (2000). Expression of VEGFR-2 and AC133 by circulating human CD34(+) cells identifies a population of functional endothelial precursors. Blood, 95, 952–958.
Salven, P., Mustjoki, S., Alitalo, R., Alitalo, K., & Rafii, S. (2003). VEGFR-3 and CD133 identify a population of CD34+lymphatic/vascular endothelial precursor cells. Blood, 101, 168–172.
Sawamoto, K., Nakao, N., Kakishita, K., Ogawa, Y., Toyama, Y., Yamamoto, A., et al. (2001). Generation of dopaminergic neurons in the adult brain from mesencephalic precursor cells labeled with a nestin-GFP transgene. Journal of Neuroscience, 21, 3895–3903.
Torrente, Y., Belicchi, M., Sampaolesi, M., Pisati, F., Meregalli, M., D’Antona, G., et al. (2004). Human circulating AC133(+) stem cells restore dystrophin expression and ameliorate function in dystrophic skeletal muscle. Journal of Clinical Investigation, 114, 182–195.
Torrente, Y., Belicchi, M., Marchesi, C., Dantona, G., Cogiamanian, F., Pisati, F., et al. (2007). Autologous transplantation of muscle-derived CD133 + stem cells in Duchenne muscle patients. Cell Transplant, 16, 563–577.
Benchaouir, R., Meregalli, M., Farini, A., D’Antona, G., Belicchi, M., Goyenvalle, A., et al. (2007). Restoration of human dystrophin following transplantation of exon-skipping-engineered DMD patient stem cells into dystrophic mice. Cell Stem Cell, 1, 646–657.
De Angelis, L., Berghella, L., Coletta, M., Lattanzi, L., Zanchi, M., Cusella-De Angelis, M. G., et al. (1999). Skeletal myogenic progenitors originating from embryonic dorsal aorta coexpress endothelial and myogenic markers and contribute to postnatal muscle growth and regeneration. Journal of Cell Biology, 147, 869–878.
Minasi, M. G., Riminucci, M., De Angelis, L., Borello, U., Berarducci, B., Innocenzi, A., et al. (2002). The meso-angioblast: a multipotent, self-renewing cell that originates from the dorsal aorta and differentiates into most mesodermal tissues. Development, 129, 2773–2783.
Sampaolesi, M., Blot, S., D’Antona, G., Granger, N., Tonlorenzi, R., Innocenzi, A., et al. (2006). Mesoangioblast stem cells ameliorate muscle function in dystrophic dogs. Nature, 444, 574–579.
Dellavalle, A., Sampaolesi, M., Tonlorenzi, R., Tagliafico, E., Sacchetti, B., Perani, L., et al. (2007). Pericytes of human skeletal muscle are myogenic precursors distinct from satellite cells. Nature of Cell Biology, 9, 255–267.
Sampaolesi, M., Torrente, Y., Innocenzi, A., Tonlorenzi, R., D’Antona, G., Pellegrino, M. A., et al. (2003). Cell therapy of alpha-sarcoglycan null dystrophic mice through intra-arterial delivery of mesoangioblasts. Science, 301, 487–492.
Mintz, B., & Illmensee, K. (1975). Normal genetically mosaic mice produced from malignant teratocarcinoma cells. Proceedings of the National Academy of Sciences of the United States of America, 72, 3585–35859.
Evans, M. J., & Kaufman, M. H. (1981). Establishment in culture of pluripotential cells from mouse embryos. Nature, 292, 154–156.
Martin, G. R. (1981). Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proceedings of the National Academy of Sciences of the United States of America, 78, 7634–7638.
Thomson, J. A., Itskovitz-Eldor, J., Shapiro, S. S., Waknitz, M. A., Swiergiel, J. J., Marshall, V. S., et al. (1998). Embryonic stem cell lines derived from human blastocysts. Science, 282, 1145–1147.
Bradley, A., Evans, M., Kaufman, M. H., & Robertson, E. (1984). Formation of germ-line chimaeras from embryo-derived teratocarcinoma cell lines. Nature, 309, 255–256.
Martin, G. R., & Evans, M. J. (1975). Differentiation of clonal lines of teratocarcinoma cells: formation of embryoid bodies in vitro. Proceedings of the National Academy of Sciences of the United States of America, 72, 1441–1445.
Martin, G. R., Wiley, L. M., & Damjanov, I. (1977). The development of cystic embryoid bodies in vitro from clonal teratocarcinoma stem cells. Developments in Biologicals, 61, 230–244.
Leahy, A., Xiong, J. W., Kuhnert, F., & Stuhlmann, H. (1999). Use of developmental marker genes to define temporal and spatial patterns of differentiation during embryoid body formation. Journal of Experimental Zoology, 284, 67–81.
Rathjen, J., & Rathjen, P. D. (2001). Mouse ES cells: experimental exploitation of pluripotent differentiation potential. Current Opinion Genetics and Develeopmet, 11, 587–594.
Perlingeiro, R. C., Kyba, M., & Daley, G. Q. (2001). Clonal analysis of differentiating embryonic stem cells reveals a hematopoietic progenitor with primitive erythroid and adult lymphoid-myeloid potential. Development, 128, 4597–4604.
Doetschman, T. C., Eistetter, H., Katz, M., Schmidt, W., & Kemler, R. (1985). The in vitro development of blastocyst-derived embryonic stem cell lines: formation of visceral yolk sac, blood islands and myocardium. Journal of Embryology and Experimental Morphology, 87, 27–45.
Schmitt, R. M., Bruyns, E., & Snodgrass, H. R. (1991). Hematopoietic development of embryonic stem cells in vitro: cytokine and receptor gene expression. Genes and Development, 5, 728–740.
Wiles, M. V., & Keller, G. (1991). Multiple hematopoietic lineages develop from embryonic stem (ES) cells in culture. Development, 111, 259–267.
Kyba, M., Perlingeiro, R. C., & Daley, G. Q. (2002). HoxB4 confers definitive lymphoid-myeloid engraftment potential on embryonic stem cell and yolk sac hematopoietic progenitors. Cell, 109, 29–37.
Soria, B., Roche, E., Berná, G., León-Quinto, T., Reig, J. A., & Martín, F. (2000). Insulin-secreting cells derived from embryonic stem cells normalize glycemia in streptozotocin-induced diabetic mice. Diabetes, 49, 157–162.
Lumelsky, N., Blondel, O., Laeng, P., Velasco, I., Ravin, R., & McKay, R. (2001). Differentiation of embryonic stem cells to insulin-secreting structures similar to pancreatic islets. Science, 292, 1389–1394.
Assady, S., Maor, G., Amit, M., Itskovitz-Eldor, J., Skorecki, K. L., & Tzukerman, M. (2001). Insulin production by human embryonic stem cells. Diabetes, 50, 1691–1697.
Hamazaki, T., Iiboshi, Y., Oka, M., Papst, P. J., Meacham, A. M., Zon, L. I., Terada, N., et al. (2001). Hepatic maturation in differentiating embryonic stem cells in vitro. FEBS Letter, 497, 15–19.
Rambhatla, L., Chiu, C. P., Kundu, P., Peng, Y., & Carpenter, M. K. (2003). Generation of hepatocyte-like cells from human embryonic stem cells. Cell Transplant, 12, 1–11.
Zhang, S. C., Wernig, M., Duncan, I. D., Brüstle, O., & Thomson, J. A. (2001). In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nature Biotechnology, 19, 1129–1133.
Bain, G., Kitchens, D., Yao, M., Huettner, J. E., & Gottlieb, D. I. (1996). Embryonic stem cells express neuronal properties in vitro. Developments in Biologicals, 168, 342–357.
Hübner, K., Fuhrmann, G., Christenson, L. K., Kehler, J., Reinbold, R., De La Fuente, R., Wood, J., Strauss, J. Fr., et al. (2003). Derivation of oocytes from mouse embryonic stem cells. Science, 300, 1251–1256.
Geijsen, N., Horoschak, M., Kim, K., Gribnau, J., Eggan, K., & Daley, G. Q. (2004). Derivation of embryonic germ cells and male gametes from embryonic stem cells. Nature, 427, 148–154.
Toyooka, Y. T. N., Akasu, R., & Noce, T. (2003). Embryonic stem cells can form germ cells in vitro. Proceedings of the National Academy of Sciences of the United States of America, 100, 11457–11462.
Klug, M. G., Soonpaa, M. H., Koh, G. Y., & Field, L. J. (1996). Genetically selected cardiomyocytes from differentiating embryonic stem cells form stable intracardiac grafts. Journal of Clinical Invest, 98, 216–224.
Müller, M., Fleischmann, B. K., Selbert, S., Ji, G. J., Endl, E., Middeler, G., et al. (2000). Selection of ventricular-like cardiomyocytes from ES cells in vitro. FASEB Journal, 14, 2540–2548.
He, J. Q., Ma, Y., Lee, Y., Thomson, J. A., & Kamp, T. J. (2003). Human embryonic stem cells develop into multiple types of cardiac myocytes: action potential characterization. Circulation Research, 93, 32–39.
Drab, M., Haller, H., Bychkov, R., Erdmann, B., Lindschau, C., Haase, H., et al. (1997). From totipotent embryonic stem cells to spontaneously contracting smooth muscle cells: a retinoic acid and db-cAMP in vitro differentiation model. FASEB Journal, 11, 905–915.
Rohwedel, J., Maltsev, V., Bober, E., Arnold, H. H., Hescheler, J., & Wobus, A. M. (1994). Muscle cell differentiation of embryonic stem cells reflects myogenesis in vivo: developmentally regulated expression of myogenic determination genes and functional expression of ionic currents. Developments in Biologicals, 164, 87–101.
Barberi, T., Bradbury, M., Dincer, Z., Panagiotakos, G., Socci, N. D., & Studer, L. (2007). Derivation of engraftable skeletal myoblasts from human embryonic stem cells. Nature Medicine, 13, 642–648.
Bhagavati, S., & Xu, W. (2005). Generation of skeletal muscle from transplanted embryonic stem cells in dystrophic mice. Biochemical and Biophysical Research Communicaitons, 333, 644–649.
Darabi, R., Gehlbach, K., Bachoo, R. M., Kamath, S., Osawa, M., Kamm, K. E., et al. (2008). Functional skeletal muscle regeneration from differentiating embryonic stem cells. Nature Medicine, 14, 134–143.
Ridgeway, A. G., & Skerjanc, I. S. (2001). Pax3 is essential for skeletal myogenesis and the expression of Six1 and Eya2. Journal of Biology Chemicals, 276, 19033–19039.
Mylona, E., Jones, K. A., Mills, S. T., & Pavlath, G. K. (2006). CD44 regulates myoblast migration and differentiation. Journal of Cell Physiology, 209, 314–321.
Prentice, D. A. (2006). Current science of regenerative medicine with stem cells. Journal of Investigative Medicine, 54, 33–37.
Briggs, R., & King, T. J. (1952). Transplantation of Living Nuclei From Blastula Cells into Enucleated Frogs’ Eggs. Proceedings of the National Academy of Sciences of the United States of America, 38, 455–463.
Gurdon, J. B., Elsdale, T. R., & Fischberg, M. (1958). Sexually mature individuals of Xenopus laevis from the transplantation of single somatic nuclei. Nature, 182, 64–65.
Willadsen, S. M. (1986). Nuclear transplantation in sheep embryos. Nature, 320, 63–65.
Prather, R. S., Barnes, F. L., Sims, M. M., Robl, J. M., Eyestone, W. H., & First, N. L. (1987). Nuclear transplantation in the bovine embryo: assessment of donor nuclei and recipient oocyte. Biology of Reproduction, 37, 859–866.
Wilmut, I., Schnieke, A. E., McWhir, J., Kind, A. J., & Campbell, K. H. (1997). Viable offspring derived from fetal and adult mammalian cells. Nature, 385, 810–813.
Wakayama, T., Perry, A. C., Zuccotti, M., Johnson, K. R., & Yanagimachi, R. (1998). Full-term development of mice from enucleated oocytes injected with cumulus cell nuclei. Nature, 394, 369–374.
Cibelli, J. B., Stice, S. L., Golueke, P. J., Kane, J. J., Jerry, J., Blackwell, C., et al. (1998). Cloned transgenic calves produced from nonquiescent fetal fibroblasts. Science, 280, 1256–1258.
Betthauser, J., Forsberg, E., Augenstein, M., Childs, L., Eilertsen, K., Enos, J., et al. (2000). Production of cloned pigs from in vitro systems. Nature Biotechnology, 18, 1055–1059.
Reggio, B. C., James, A. N., Green, H. L., Gavin, W. G., Behboodi, E., Echelard, Y., & Godke, R. A. (2001). Cloned transgenic offspring resulting from somatic cell nuclear transfer in the goat: oocytes derived from both follicle-stimulating hormone-stimulated and nonstimulated abattoir-derived ovaries. Biology of Reproduction, 65, 1528–1533.
Chesné, P., Adenot, P. G., Viglietta, C., Baratte, M., Boulanger, L., & Renard, J. P. (2002). Cloned rabbits produced by nuclear transfer from adult somatic cells. Nature Biotechnology, 20, 366–369.
Shin, T., Kraemer, D., Pryor, J., Liu, L., Rugila, J., & Howe, L. (2002). A cat cloned by nuclear transplantation. Nature, 415, 859.
Rideout, W. Mr., Hochedlinger, K., Kyba, M., Daley, G. Q., & Jaenisch, R. (2002). Correction of a genetic defect by nuclear transplantation and combined cell and gene therapy. Cell, 109, 17–27.
Wakayama, T., Tabar, V., Rodriguez, I., Perry, A. C., Studer, L., & Mombaerts, P. (2001). Differentiation of embryonic stem cell lines generated from adult somatic cells by nuclear transfer. Science, 292, 740–743.
Takahashi, K., & Yamanaka, S. (2006). Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell, 126, 663–676.
Takahashi, K., Tanabe, K., Ohnuki, M., Narita, M., Ichisaka, T., Tomoda, K., et al. (2007). Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell, 131, 861–872.
Yu, J., Vodyanik, M. A., Smuga-Otto, K., Antosiewicz-Bourget, J., Frane, J. L., Tian, S., et al. (2007). Induced pluripotent stem cell lines derived from human somatic cells. Science, 318(5858), 1917–1920.
Blelloch, R., Venere, M., Yen, J., & Ramalho-Santos, M. (2007). Generation of induced pluripotent stem cells in the absence of drug selection. Cell Stem Cell, 1, 254–247.
Wernig, M., Meissner, A., Foreman, R., Brambrink, T., Ku, M., Hochedlinger, K., et al. (2007). In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature, 448, 318–324.
Park, I. H., Zhao, R., West, J. A., Yabuuchi, A., Huo, H., Ince, T. A., et al. (2008). Reprogramming of human somatic cells to pluripotency with defined factors. Nature, 451, 141–146.
Byrne, J. A., Pedersen, D. A., Clepper, L. L., Nelson, M., Sanger, W. G., Gokhale, S., et al. (2007). Producing primate embryonic stem cells by somatic cell nuclear transfer. Nature, 450, 497–502.
Hanna, J. W. M., Markoulaki, S., Sun, C. W., Meissner, A., Cassady, J. P., Beard, C., et al. (2007). Treatment of sickle cell anemia mouse model with iPS cells generated from autologous skin. Science, 318, 1920–1923.
Wernig, M., Zhao, J. P., Pruszak, J., Hedlund, E., Fu, D., Soldner, F., et al. (2008). Neurons derived from reprogrammed fibroblasts functionally integrate into the fetal brain and improve symptoms of rats with Parkinson’s disease. Proceedings of the National Academy of Sciences of the United States of America, 105, 5856–5861.
Acknowledgments
This work was supported by the Dr. Bob and Jean Smith Foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Radbod Darabi and Filipe N. C. Santos contributed equally to this manuscript.
Rights and permissions
About this article
Cite this article
Darabi, R., Santos, F.N.C. & Perlingeiro, R.C.R. The Therapeutic Potential of Embryonic and Adult Stem Cells for Skeletal Muscle Regeneration. Stem Cell Rev 4, 217–225 (2008). https://doi.org/10.1007/s12015-008-9023-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-008-9023-3