Abstract
Cisplatin-induced generation of reactive oxygen species leads to acute nephrotoxicity limiting its use in the treatment of various cancers. Gelsemine, an alkaloid isolated from Gelsemium elegans, is known to possess anti-inflammatory and anti-cancer activities. This study was aimed to investigate as to whether gelsemine can serve as a protective agent against cisplatin-induced nephrotoxicity. Male Wistar rats were divided into 6 groups, each with 6 rats. Group 1 served as control and received the vehicles (peanut oil for 14 days and 0.9 % saline on day 14 for gelsemine and cisplatin respectively). Group 2 received a single intraperitoneal injection of cisplatin on day 14. Group 3 and 4 were pretreated with two different doses of gelsemine in addition to cisplatin, and group 5 and 6 received only gelsemine. The effects of gelsemine on cisplatin-induced nephrotoxicity were examined by measuring anti-oxidant enzymes activities, lipid peroxidation, and DNA damage in the kidneys, a well-established model of oxidative damage. Pretreatment of rats with gelsemine caused a significant attenuation of cisplatin-induced DNA and oxidative damages. The blockade of lipid peroxidation and xanthine oxidase activity was accompanied by increased production and/or activity of anti-oxidants, both enzymatic (catalase, glutathione peroxidase, glutathione reductase, and glutathione-S-transferase) and non-enzymatic (GSH). The biomarkers of kidney malfunctioning, creatinine, and blood urea nitrogen were ameliorated. The results of the present study suggest that gelsemine effectively suppressed cisplatin-induced renal injury by improving redox status.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cisplatin is the most potent anti-tumor agent used for the treatment of a wide variety of cancers including, testicular, ovarian, bladder, endometrial, cervical, and lung [1–5]. However, the dose-limiting toxicities caused by cisplatin constraint its use to a full therapeutic potential [6]. The mitochondrion is considered to be the primary target of cisplatin-induced oxidative damage resulting in the loss of sulphydryl proteins, inhibition of calcium uptake, and a reduction in the mitochondrial membrane potential [1, 6, 7]. Cisplatin accumulated in the renal cortex (proximal tubular epithelium) mitochondria of the kidney, the major route of its excretion [1, 8–10], generates reactive oxygen species (ROS). The ROS are known to directly bind with the cellular macromolecules, including lipids, proteins, and DNA, and impair their normal functioning [1, 11–14]. In an aqueous environment, the chloride ligands of cisplatin are replaced by water molecules, generating the positively charged electrophiles. These electrophiles react with nucleophilic sites of DNA, RNA, and proteins and impair their normal functioning. As a result of lipid peroxidation, the integrity of the renal cell membrane is compromised, the proteins are denatured. The adduct formation with DNA results in interstrand and intra-strand crosslinks causing the arrest of DNA synthesis and replication in rapidly proliferating cells. The endogenous anti-oxidant defense system is also depleted which further aggravates oxidative damage leading to impaired kidney functioning and inflammation [1, 15–18].
In recent years, a growing interest in the use of phytochemicals as free radical scavengers and inhibitors of oxidative stress has been considerably exploited. For instance, anti-oxidant rich foods have been suggested to play an essential role in the prevention of cardiovascular diseases and cancer [17, 18]. Gelsemine, an alkaloid has been isolated from plants of genus Gelsemium of the family Loganiaceae. The family comprises three popularly known species: Gelsemium elegans Benth. (G. elegans), Gelsemium sempervirens Ait. (G. sempervirens), and Gelsemium rankinii Small (G. rankinii). Whereas, G. elegans is native to China and Southeast Asia, G. sempervirens Ait. and G. rankinii are native to North America. The use of G. sempervirens has for long been known to alleviate pain and reduce anxiety in the traditional folk medicine. Also, very low doses of G. sempervirens extracts have been shown to potently reduce anxiety in animal models [19, 20]. In fact, a single administration of gelsemine at a dose far below its LD50 in mice exerted a potent anxiolytic effect that could be blocked by strychnine, a glycine receptor antagonist [21]. Therefore, the G. elegans alkaloids have been considered to be promising in treating anxiety-related psychiatric disorders; however, they showed no significant anti-depressant effect [19, 20].
The pharmacological effects of gelsemine on cisplatin-induced nephrotoxicity have not been studied so far. The present study was aimed to investigate the effects of gelsemine on cisplatin-induced oxidative and DNA damages.
Materials and Methods
Chemicals
Reduced glutathione, glutathione reductase, oxidized glutathione, bovine serum albumin, oxidized and reduced nicotinamide adenine dinucleotide phosphate, reduced nicotinamide adeninedinucleotide phosphate, gelsemine, flavine adenine dinucleotide, glucose-6-phosphate, 2,6-dichlorophenolindophenol, thiobarbituric acid, xanthine, and dinitrophenyl hydrazine were obtained from Abcam. Cisplatin was purchased from Sigma; ferric nitrate, ammoniumthiocyanate, 1,2-dichloroethane, hydrogen peroxide, magnesium chloride, di-sodium hydrogen phosphate, sodium di-hydrogen phosphate, and sodium hydroxide were purchased from Aladdin (China). All of the other reagents used were of highest purity and commercially available.
Animals
Male Wistar rats, 150–250 g, 6–8 weeks old, were obtained from Shanghai Laboratory Animal Center, Chinese Academy of Sciences. Rats were housed in polypropylene cages in groups of four rats per cage in the animal care facility under room temperature (25 + 1 °C) with a 12-h light–dark cycle. The rats were allowed free access to standard laboratory animal feed and water. Before treatment, the rats were allowed to acclimatize for a week. All of the used experimental procedures were checked and approved by the institutional animal ethics committee.
Experimental Procedure
Rats were randomly divided into six groups, each consisting of eight animals. Group 1 served as control and received peanut oil (vehicle of gelsemine) for 14 days and 0.9 % saline (vehicle of cisplatin) on day 14 only. Group 2 served as negative control and received an oral dose of peanut oil daily for 14 days and a single intraperitoneal injection of cisplatin (7.5 mg/kg) on the 14th day. Cisplatin was dissolved in 0.9 % saline at 25 °C at a concentration of 1 mg/ml. Groups 3 and 4 were pretreated with an oral dose of gelsemine 5 mg/kg (G1) and 25 mg/kg (G2), respectively, for 14 days and a single intraperitoneal injection of cisplatin (7.5 mg/kg) was administered on the 14th day in both the groups. Group 5 and 6 received only G1 and G2 of gelsemine for 14 days. On the 15th day, the rats were subjected to mild ether anesthesia and then sacrificed by cervical dislocation. The blood was drawn from the retro-orbital sinus, and serum was obtained. The kidneys were excised after perfusion washed with ice-cold saline, and homogenates were prepared in chilled phosphate buffer.
Preparation of Post-Mitochondrial Supernatant
Tissue processing and preparation of post-mitochondrial supernatant (PMS) were carried out as described before [1]. Kidneys were removed quickly, cleaned free of extraneous material, and immediately perfused with ice-cold saline (0.85 % sodium chloride). The homogenates prepared in chilled phosphate buffer (0.1 m, pH 7.4) containing KCl (1.17 %) were filtered through muslin cloth, and centrifuged at 800×g for 5 min at 4 °C. The nuclear supernatants were separated from the nuclear pellets and centrifuged at 10 500×g for 20 min at 4 °C to obtain PMS, which was used for measuring activity of various enzymes.
Biochemical Parameters and Toxicity Markers
Measurement of malondialdehyde (MDA), glutathione-S-transferase (GST) activity, glutathione peroxidase (GPx) activity, glutathione reductase (GR) activity, reduced glutathione (GSH), catalase (CAT) activity, xanthine oxidase (XO) activity, creatinine, and estimation of blood urea nitrogen (BUN) was carried out by the method described before [1, 22–28].
Statistical Analysis
Differences between groups were analyzed using analysis of variance followed by Dunnet’s multiple comparisons test. All data points represent the mean with standard deviation of the treatment groups.
Results
Effect of Gelsemine Pretreatment on Cisplatin-Induced Increase in the Levels of Malondialdehyde
Compared with the untreated (control) group, cisplatin induced a significant amplification (P < 0.01) in the formation of the malondialdehyde (MDA), the biomarker of lipid peroxidation (Fig. 1a). Pretreatment with gelsemine at both doses G1 and G2 led to a significant (P < 0.01) restoration of membrane lipids integrity when compared with the group treated with cisplatin only. Gelsemine alone did not show any significant effect compared with control.
Effect of gelsemine on cisplatin-induced increase in lipid peroxidation (a), reduction of catalase (CAT) activity (b) and elevation of xanthine oxidase (XO) activity (c) in rat kidney. CIS cisplatin, G1 gelsemine 5 mg/kg, G2 gelsemine 25 mg/kg. Each value indicates mean ± SEM of six observations. # P < 0.05, ## P < 0.01, compared with corresponding value of control. *P < 0.05, **P < 0.01, compared with the corresponding value for cisplatin-treated group
Effect of Gelsemine Pretreatment on Reduced Activity of Catalase Caused by Cisplatin
A significant reduction in the CAT activity (P < 0.01) was observed in the kidneys of cisplatin-treated rats compared with the untreated control (Fig. 1b). Pretreatment with gelsemine caused a significant and dose-dependent elevation of the CAT activity (P < 0.01). Compared with the control, gelsemine alone did not produce a significant effect on the catalase activity.
Effect of Gelsemine Pretreatment on the Cisplatin-Induced Activity of Xanthine Oxidase
A significant increase (P < 0.01) in the activity of XO was detected in the renal tissue of cisplatin-treated rats when compared with controls (Fig. 1c). Pretreatment with both G1 and G2 doses of gelsemine caused a significant restoration of XO activity (P < 0.01). A treatment with gelsemine alone (G1 and G2) showed no significant change when compared with the control group.
Effect of Gelsemine Pretreatment on the Cisplatin-Induced Decrease in the Levels of Reduced Glutathione
The level of GSH was depressed significantly (P < 0.01) in the cisplatin-treated group compared with the untreated control group (Fig. 2a). Gelsemine pretreatment caused a significant elevation in the GSH levels (P < 0.01) compared with the cisplatin-treated group. The GSH levels in the group treated with gelsemine alone were not significantly different from those of the control group.
Effect of gelsemine on cisplatin-induced reduction of reduced glutathione (GSH) level (a), and activities of glutathione-S-transferase (GST) (b), glutathione peroxidase (GPx) (c) glutathione reductase (GR) (d) in rat kidney. CIS cisplatin, G1 gelsemine 5 mg/kg, G2 gelsemine 25 mg/kg. Each value indicates mean ± SEM of six observations. # P < 0.05, ## P < 0.01, compared with corresponding value of control. *P < 0.05, **P < 0.01, compared with the corresponding value for cisplatin-treated group
Effect of Gelsemine Pretreatment on the Cisplatin-Induced Decrease in the Activity of Glutathione-S-Transferase
A significant decrease in GST activity (P < 0.01) was observed in cisplatin-treated rats compared with the control (Fig. 2b). Two weeks pretreatment with gelsemine dose-dependently and significantly elevated the GST activity (P < 0.01).
Effect of Gelsemine Pretreatment on the Cisplatin-Induced Reduction of the Glutathione Peroxidase Activity
Rats treated with cisplatin showed a significant reduction in the activity of GPx (P < 0.01) when compared with control (Fig. 2c). However, rats pretreated with either G1 or G2 doses of gelsemine caused a significant restoration of GPx activity (P < 0.01). Compared to the control group, Gelsemine alone (G1 or G2) produced no significant effect.
Effect of Cisplatin and Gelsemine Pretreatment on the Activity of Glutathione Reductase
Compared with the controls, the cisplatin-treated rats showed a significant decrease in GR activity (P < 0.001) (Fig. 2d). The activity was found to be restored and significantly elevated in the rats pretreated with gelsemine G1 and G2 (P < 0.01). Gelsemine alone did not show any significant effect compared with control.
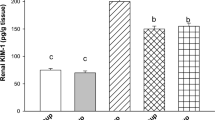
Effect of Gelsemine Pretreatment on the Levels of Cisplatin-Caused Nephrotoxicity Markers
Cisplatin administration resulted in a significantly increased (P < 0.001) creatinine concentration compared with the untreated control group (Fig. 3a). The administration of gelsemine at G1 and G2 doses prevented the elevation of creatinine levels, and the effect was dose-dependent and significant compared with the untreated group (P < 0.01). A significant increase (P < 0.01) in the BUN levels of cisplatin-treated rats was observed when compared with controls (Fig. 3b). Pretreatment with gelsemine G1 and G2 doses caused a marked reduction of BUN (P < 0.01). Gelsemine alone was not found to produce a significant effect compared with control.
Effect of gelsemine on cisplatin-induced increase in serum creatinine (a) and blood urea nitrogen levels (b). CIS cisplatin, G1 gelsemine 5 mg/kg, G2 gelsemine 25 mg/kg. Each value indicates mean ± SEM of six observations. # P < 0.05, ## P < 0.01, compared with corresponding value of control. *P < 0.05, **P < 0.01, compared with the corresponding value for cisplatin-treated group
Effect of Gelsemine on Cisplatin-Induced DNA Damage
Figure 4 shows analysis of isolated DNA in the different groups. Group 2, treated with cisplatin, showed more smearing pattern compared with group 1, thus validating cisplatin-induced DNA damage. Groups 3 and 4 showed the protective effects of gelsemine against cisplatin- induced DNA damage. The DNA was not found to be damaged in group1 and group 5, which were treated only with peanut oil and gelsemine, respectively.
Discussion
Cisplatin, one of the most effective and widely used cytotoxic agents in the treatment of various cancers, is also associated with nephrotoxicity [1, 17, 27–31]. The cisplatin-induced acute renal toxicity is known to involve mitochondrial oxidative stress. Cellular macromolecules, including lipids, proteins and DNA, are directly affected by the cisplatin-generated ROS. These reactive oxygen species cause peroxidation of the membrane lipids, denaturing of the proteins, and inactivation of the enzymes [6, 7]. Previous studies have also indicated that cisplatin potently inhibited the cellular anti-oxidant armory and induced GSH depletion, involved in nephrotoxicity [1, 8, 17]. A number of anti-oxidants including natural products isolated from plants have been reported to suppress the cytotoxic effects of cisplatin [1, 8–14, 16, 18, 30–34]. Gelsemine, an alkaloid isolated from G. elegans, has earlier been found to exhibit anti-nociceptive and anti-tumor activities [19, 21, 34]. We studied for the first time the protective effects of gelsemine against cisplatin-induced nephrotoxicity.
Our results demonstrated that cisplatin-induced increase in the activity of oxidative enzyme, xanthine oxidase, was substantially suppressed by gelsemine. Also, the lipid peroxidation reflected in the enhanced production of malondialdehyde was reversed dose-dependently by gelsemine pretreatment. The prevention of lipid peroxidation protected the membrane integrity thereby led to reduction in the leakage of creatinine and BUN into the blood. Our results clearly showed a decline of serum content of creatinine and BUN in gelsemine-treated rats. We also found that gelsemine effectively blocked the cisplatin-induced impairment of anti-oxidant defense system including enzymes (GPx, GST, GR, and CAT) and reduced glutathione. The reversal of cisplatin-mediated nephrotoxicity is evident from the prevention of DNA damage in the gelsemine-treated animals. Clearly, gelsemine appears to play a dual role in protecting the cells from oxidative damage, which is blockade of ROS generation and enhancement of the anti-oxidant system.
Since the toxic actions of the anti-cancer drugs are the basis of their effectiveness in combating with the growing tumors, the important question to ask is if the prevention of cytotoxicity would impair the therapeutic efficacy of the drug. Further work is needed to investigate the debilitating effect of gelsemine on therapeutic efficacy of cisplatin. However, the anti-cancer activity of gelsemine [34] may actually prove to be an added advantage and impart this compound with multiple beneficial effects. Thus, clinical investigations are needed to be conducted to study the use of gelsemine as adjuvant in cisplatin-based chemotherapy.
In conclusion, this study demonstrated that mechanism of cisplatin toxicity is related to free radical generation and depletion of the anti-oxidant defense system. A potent inhibition of cisplatin-induced oxidative stress by gelsemine involved suppression of ROS and a concomitant enhancement of the anti-oxidant statue of the cells. The protective effect of gelsemine against cisplatin-mediated nephrotoxicity suggests the possibility of its use as an adjuvant with antineoplastic drugs. Moreover, the anti-tumor activity of the compound as reported previously might prove to be of additive benefit.
References
Sultana, S., Verma, K., & Khan, R. (2012). Nephroprotective efficacy of chrysin against cisplatin-induced toxicity via attenuation of oxidative stress. Journal of Pharmacy and Pharmacology, 64, 872–881.
Sanatani, M. S., Lazo-Langner, A., & Al-Rasheedy, I. M. (2013). Cisplatin and short-term 5-Fluorouracil infusion for paraneoplastic microangiopathic hemolytic anemia in gastric cancer: a case report and review of the literature. Case Reports in Oncological Medicine, 2013, 594787.
Petrelli, F., Zaniboni, A., Coinu, A., et al. (2013). Cisplatin or not in advanced gastric cancer: a systematic review and meta-analysis. PLoS ONE, 8, e83022.
Zhang, G., Fu, C., Zhang, Y., et al. (2012). Extended-field intensity-modulated radiotherapy and concurrent cisplatin-based chemotherapy for postoperative cervical cancer with common iliac or para-aortic lymph node metastases: a retrospective review in a single institution. International Journal of Gynecological Cancer, 22, 1220–1225.
Jiang, L., Yang, K. H., Guan, Q. L., et al. (2012). Cisplatin plus etoposide versus other platin-based regimens for patients with extensive small-cell lung cancer: a systematic review and meta-analysis of randomised, controlled trials. The Internal Medicine Journal, 42, 1297–1309.
Marullo, R., Werner, E., Degtyareva, N., Moore, B., Altavilla, G., Ramalingam, S. S., et al. (2013). Cisplatin induces mitochondrial-ROS response that contributes to cytotoxicity depending on mitochondrial redo status and bioenergetic functions. PLoS ONE, 8(11), e81162.
Yang, Y., Liu, H., Liu, F., & Dong, Z. (2014). Mitochondrial dysregulation and protection in cisplatin nephrotoxicity. Archives of Toxicology, 88, 1249–1256.
Waly, M. I., Ali, B. H., & Nemmar, A. (2013). Acute effects of diesel exhaust particles and cisplatin on oxidative stress in cultured human kidney (HEK 293) cells, and the influence of curcumin thereon. Toxicology in Vitro, 27, 2299–2304.
Domitrovic, R., Cvijanovic, O., Pernjak-Pugel, E., et al. (2013). Berberine exerts nephroprotective effect against cisplatin-induced kidney damage through inhibition of oxidative/nitrosative stress, inflammation, autophagy and apoptosis. Food and Chemical Toxicology, 62, 397–406.
Waly, M. I., Ali, B. H., Al-Lawati, I., et al. (2013). Protective effects of emodin against cisplatin-induced oxidative stress in cultured human kidney (HEK 293) cells. Journal of Applied Toxicology, 33, 626–630.
Waly, M. I., Al Moundhri, M. S., & Ali, B. H. (2011). Effect of curcumin on cisplatin- and oxaliplatin-induced oxidative stress in human embryonic kidney (HEK) 293 cells. Renal Failure, 33, 518–523.
Rodrigues, M. A., Rodrigues, J. L., Martins, N. M., et al. (2011). Carvedilol protects against cisplatin-induced oxidative stress, redox state unbalance and apoptosis in rat kidney mitochondria. Chemico-Biological Interactions, 189, 45–51.
Rjiba-Touati, K., Boussema, I. A., Belarbia, A., et al. (2011). Protective effect of recombinant human erythropoietin against cisplatin-induced oxidative stress and nephrotoxicity in rat kidney. International Journal of Toxicology, 30, 510–517.
Dogukan, A., Tuzcu, M., Agca, C. A., et al. (2011). A tomato lycopene complex protects the kidney from cisplatin-induced injury via affecting oxidative stress as well as Bax, Bcl-2, and HSPs expression. Nutrition and Cancer, 63, 427–434.
Liao, Y. J., Jin, Y. P., Lin, L., et al. (2010). Determination of oxidative damage on DNA in brain and kidney of mice induced by anti-tumor agent of cisplatin. Zhongguo Ying Yong Sheng Li Xue Za Zhi, 26, 180–181.
Santos, N. A., Bezerra, C. S., Martins, N. M., et al. (2008). Hydroxyl radical scavenger ameliorates cisplatin-induced nephrotoxicity by preventing oxidative stress, redox state unbalance, impairment of energetic metabolism and apoptosis in rat kidney mitochondria. Cancer Chemotherapy and Pharmacology, 61, 145–155.
Santos, N. A., Catao, C. S., Martins, N. M., et al. (2007). Cisplatin-induced nephrotoxicity is associated with oxidative stress, redox state unbalance, impairment of energetic metabolism and apoptosis in rat kidney mitochondria. Archives of Toxicology, 81, 495–504.
Dillioglugil, M. O., Maral Kir, H., Gulkac, M. D., et al. (2005). Protective effects of increasing vitamin E and a doses on cisplatin-induced oxidative damage to kidney tissue in rats. Urologia Internationalis, 75, 340–344.
Liu, M., Shen, J., Liu, H., et al. (2011). Gelsenicine from Gelsemium elegans attenuates neuropathic and inflammatory pain in mice. Biological and Pharmaceutical Bulletin, 34, 1877–1880.
Liu, M., Huang, H. H., Yang, J., et al. (2013). The active alkaloids of Gelsemium elegans Benth. are potent anxiolytics. Psychopharmacology (Berl), 225, 839–851.
Zhang, J.-Y., Gong, N., Huang, J.-L., Guo, L.-C., & Wan, Y.-X. (2013). Gelsemine, a principal alkaloid from Gelsemium sempervirens Ait., exhibits potent and specific antinociception in chronic pain by acting at spinal a3 glycine receptors. Pain, 154, 2452–2462.
Small, D. M., Bennett, N. C., Roy, S., et al. (2012). Oxidative stress and cell senescence combine to cause maximal renal tubular epithelial cell dysfunction and loss in an in vitro model of kidney disease. Nephron Experimental Nephrology, 122, 123–130.
de Munck, E., Munoz-Saez, E., Antonio, M. T., et al. (2013). Effect of beta-N-methylamino-l-alanine on oxidative stress of liver and kidney in rat. Environmental Toxicology and Pharmacology, 35, 193–199.
Annavarajula, S. K., Dakshinamurty, K. V., Naidu, M. U., et al. (2012). The effect of l-arginine on arterial stiffness and oxidative stress in chronic kidney disease. The Indian Journal of Nephrology, 22, 340–346.
Balat, A., Resic, H., Bellinghieri, G., et al. (2012). Devil’s Triangle in Kidney Diseases: Oxidative Stress, Mediators, and Inflammation. The International Journal of Nephrology, 2012, 156286.
Bhattacharya, S., Manna, P., Gachhui, R., et al. (2013). d-saccharic acid 1,4-lactone protects diabetic rat kidney by ameliorating hyperglycemia-mediated oxidative stress and renal inflammatory cytokines via NF-kappaB and PKC signaling. Toxicology and Applied Pharmacology, 267, 16–29.
Park, C. H., Lee, S. L., Okamoto, T., et al. (2012). Rokumi-jio-gan-Containing Prescriptions Attenuate Oxidative Stress, Inflammation, and Apoptosis in the Remnant Kidney. Evidence-Based Complementary and Alternative Medicine, 2012, 587902.
Chargui, I., Grissa, I., Bensassi, F., et al. (2012). Oxidative stress, biochemical and histopathological alterations in the liver and kidney of female rats exposed to low doses of deltamethrin (DM): A molecular assessment. Biomedical and Environmental Sciences, 25, 672–683.
Ghosh, P., Roy, S. S., Chakraborty, P., et al. (2013). Effects of organoselenium compound 2-(5-selenocyanato-pentyl)-benzo[de]isoquinoline 1,3-dione on cisplatin induced nephrotoxicity and genotoxicity: An investigation of the influence of the compound on oxidative stress and antioxidant enzyme system. BioMetals, 26, 61–73.
Kimoto, Y., Nishinohara, M., Sugiyama, A., et al. (2013). Protective effect of lactoferrin on Cisplatin-induced nephrotoxicity in rats. Journal of Veterinary Medical Science, 75, 159–164.
Mahmoud, M. F., & El Shazly, S. M. (2013). Pioglitazone protects against cisplatin induced nephrotoxicity in rats and potentiates its anticancer activity against human renal adenocarcinoma cell lines. Food and Chemical Toxicology, 51, 114–122.
Kong, D., Zhuo, L., Gao, C., et al. (2013). Erythropoietin protects against cisplatin-induced nephrotoxicity by attenuating endoplasmic reticulum stress-induced apoptosis. Journal of Nephrology, 26, 219–227.
Ali, B. H., Al-Salam, S., Al Husseini, I. S., et al. (2013). Abrogation of cisplatin-induced nephrotoxicity by emodin in rats. Fundamental and Clinical Pharmacology, 27, 192–200.
Zhao, Q.-C., Hua, W., Zhang, L., Guo, T., Zhao, M.-H., Yan, M., et al. (2010). Antitumor activity of two gelsemine metabolites in rat liver microsomes. Journal of Asian Natural Products Research, 12(9), 731–739.
Author information
Authors and Affiliations
Corresponding author
Additional information
Lin Lin and Jing Zheng have contributed equally to this study.
Rights and permissions
About this article
Cite this article
Lin, L., Zheng, J., Zhu, W. et al. Nephroprotective Effect of Gelsemine Against Cisplatin-Induced Toxicity is Mediated Via Attenuation of Oxidative Stress. Cell Biochem Biophys 71, 535–541 (2015). https://doi.org/10.1007/s12013-014-0231-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0231-y