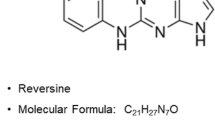
Abstract
Despite the advances of adjuvant chemotherapy and significant improvement of survival, the prognosis for patients with osteosarcoma is generally poor. The search for more effective anti-osteosarcoma agents is necessary and urgent. Here we report that perifosine induces cell apoptosis and growth inhibition in cultured human osteosarcoma cells. Perifosine blocks Akt/mTOR complex 1 (mTORC1) signaling, while promoting caspase-3, c-Jun N-terminal kinases (JNK), and p53 activation. Further, perifosine inhibits survivin expression probably by disrupting its association with heat shock protein-90 (HSP-90). These signaling changes together were responsible for a marked increase of osteosarcoma cell apoptosis and growth inhibition. Finally, we found that a low dose of perifosine enhanced etoposide- or doxorubicin-induced anti-OS cells activity. The results together suggest that perifosine might be used as a novel and effective anti-osteosarcoma agent.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The use of effective adjuvant chemotherapies plays an important role in controlling osteosarcoma (OS) [1, 2]. Despite the latest advances, the prognosis for patients with osteosarcoma is still poor [1]. In order to ensure a cure, it is necessary to develop more effective adjuvant treatments. Perifosine is the first oral available alkylphospholipid which interacts with the cell membrane and affects multiple signal transduction pathways [3–5]. In clinical phase I trials, perifosine administration inhibits multiple tumor cell growth with tolerable side effects [6, 7]. Perifosine blocks Akt activation by disrupting its recruitment to the plasma membrane [3, 8, 9]. A number of recent studies have suggested that the other mechanisms besides Akt inhibition may also be responsible for its cytotoxic effects. For example, perifosine promotes c-Jun NH2-terminal kinase (JNK) activation [4, 10, 11] and p21 accumulation [12]. Further, perifosine inhibits pro-survival extracellular signal-regulated kinase 1/2 (Erk1/2) activation [13]. Here we report that perifosine induces significant anti-osteosarcoma cells ability by modulating multiple signaling pathways.
Materials and Methods
Cell Culture
Human osteosarcoma cell lines U2OS, MG63, and SaOs-2 cells were obtained from Shenyang Jia-qian-sheng Biotech (Shenyang, China). Cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM, Sigma, St. Louis, MO) containing 10 % fetal calf serum (FCS, Sigma, St. Louis, MO), 2 mmol/l l-glutamine, and 100 mg/ml penicillin/streptomycin (Sigma, St. Louis, MO).
Reagents
Perifosine was obtained from Selleck Chemical LLC (Houston, TX); anti-heat shock protein-90 (HSP-90), survivin, and tubulin antibodies were purchased from Santa Cruz Biotechnology (Shanghai, China). SP600125, LY 294002, 17-AAG, 17-DMAG, wortmannin, Akt inhibitor X, z-DEVD-fmk, and z-VAD-fmk were purchased from EMD Bioscience (Shanghai, China). Monoclonal mouse anti-β-actin, MnTBAP, C2 ceramide, and fumonisin B1 (F-B1) were obtained from Sigma (St. Louis, MO). All other antibodies were purchase from Cell Signaling Technology (Boston, MA).
Cell Viability Assay (MTT Assay)
Cell viability was measured by the 3-[4,5-dimethylthylthiazol-2-yl]-2,5 diphenyltetrazolium bromide (MTT) method. In brief, cells were collected and seeded in 96-well plates at a density of 1 × 105 cells/cm2. 10 μl of MTT tetrazolium salt (Sigma, St. Louis, MO) dissolved in PBS at a concentration of 10 mg/ml was added to each well with indicated treatment and incubated in CO2 incubator for 3 h at 37 °C. 200 μl of DMSO (Sigma, St. Louis, MO) was added to dissolve formazan crystals and the absorbance of each well was observed by a plate reader at a test wavelength of 490 nm with a reference wavelength of 630 nm.
Caspase-3 Activity Assay
After indicated treatments, cytosolic proteins from approximately 2 × 106 U2OS cells were extracted in hypotonic cell lysis buffer (25 mm HEPES, pH 7.5, 5 mm MgCl2, 5 mm EDTA, 5 mm dithiothreitol, 0.05 % phenylmethylsulfonyl fluoride). The protein concentration of samples was determined using a Bio-Rad Bradford protein assay kit (Bio-Rad, Shanghai, China). 10 μg of cytosolic extracts were added to caspase assay buffer (312.5 mm HEPES, pH 7.5, 31.25 % sucrose, 0.3125 % CHAPS) with benzyloxycarbonyl-DEVD-7-amido-4-(trifluoromethyl)coumarin as substrates (Calbiochem, Darmstadt, Germany). Release of 7-amido-4-(trifluoromethyl)coumarin (AFC) was quantified, after 2 h of incubation at 37C, using a Fluoroskan system (Thermo-Labsystems, Helsinki, Finland) set to an excitation value of 355 nm and emission value of 525 nm. The results were expressed as relative fluorescence units/μg of protein.
Quantification of Apoptosis by ELISA
The Cell Apoptosis ELISA Detection Kit (Roche, Palo Alto, CA) was used to quantify U2OS cell apoptosis according to manufacturer’s protocol. In brief, after indicated treatments, the cytoplasmic histone/DNA fragments from cells were extracted and bound to immobilized anti-histone antibody. Subsequently, the peroxidase-conjugated anti-DNA antibody was used for the detection of immobilized histone/DNA fragments. After addition of substrate for peroxidase, the spectrophotometric absorbance of the samples was determined using a plate reader at 405 nM.
Hoechst Nuclear Staining Assessing the Percentage of Apoptotic Cells
Similar to previously reported [11], to detect apoptotic cells, U2OS cells with indicated treatment were fixed with 4 % formaldehyde in phosphate-buffered saline (PBS) for 15 min at 4 °C. Cells were incubated for 15 min with 5 μg/ml of Hoechst 33342 (Sigma, St. Louis, MO) to stain the nuclei. After washing with PBS, the apoptotic cells were observed under a Confocal Fluorescence microscope (magnification 1:100). Cells exhibiting condensed chromatin and/or fragmented nuclei (Hoechst 33342 stain, Blue) were scored as apoptotic cells. For each Hoechst experiment, at least 400 cells in 20 random scope fields were counted for apoptotic percentage.
Western Blotting
Cells were washed with ice-cold PBS, scraped into PBS, and collected by centrifugation. Pellets were re-suspended in a lysis buffer containing 50 mmol/l HEPES, 150 mmol/l NaCl, 1 mmol/l EDTA, 1 mmol/l EGTA, 10 % glycerol, 0.5 % NP-40, 0.5 % Tween 20, 1 mmol/l dithiothreitol, and protease inhibitor cocktail (Roche Diagnostics, Indianapolis, IN) and vortexed for 20 min at 4 °C; insoluble material was removed by centrifugation. Proteins (30 μg) were resolved by SDS-PAGE and transferred to nitrocellulose membranes. Membranes were incubated sequentially in TBS containing 0.05 % Tween-20 and 5 % non-fat dry milk as follows: no addition, 1 h at room temperature (blocking); primary antibody, overnight at 4 °C; and secondary antibody diluted 1/4,000, 2 h at room temperature. Bound secondary antibody was detected by West Pico and West Femto chemiluminescent substrates (Pierce, Rockford, IL). Western blot results were quantified by Image J software from NIH website.
Measurement of Ceramide Generation
After overnight starvation in 1 % FBS containing media, cells was treated with indicated treatments. The lipids were then extracted with chloroform/methanol. Cells were scraped into 1 ml of ice-cold methanol. One milliliter of chloroform and 0.9 ml of 2 mol/l KCl + 0.2 mol/l H3PO4 was added to each aliquot and the chloroform phases were dried under nitrogen. Ceramide was separated by thin layer chromatography (TLC) utilizing Silica Gel 60-coated glass plates (Fisher). Fifty percent of the lengths of these TLC plates were developed in chloroform/methanol/acetic acid (9:1:1) and then dried. The plates were re-developed in petroleum ether/diethylether/acetic acid (60:40:1) and then dried and stained with iodide vapor. The identity of the ceramide was standardized by the addition of authentic ceramide standards. Radioactive ceramide was then quantified by scraping from the TLC plates followed by liquid scintillation counting.
Immunoprecipitation
U2OS with indicated treatments were lysed with lysis buffer, 150 mM NaCl (pH 7.4), 1 % Triton X-100, 10 % glycerol, 0.3 mM EDTA, 0.2 mM Na3VO4, and protease inhibitor cocktails (Roche Diagnostics, Indianapolis, IN). Precleared samples (600 μg of protein each) were incubated with primary antibodies in lysis buffer overnight at 4 °C. To this, 35 μl of protein A/G beads was added and the samples were incubated for 2 h at 4 °C. The beads were washed five times with phosphate-buffered saline (PBS) and twice with lysis buffer, boiled, followed by Western blotting analysis.
Statistical Analysis
The values in the figures are expressed as the mean ± standard deviation (SD). Values of p < 0.05 were considered as statistically significant with a student t test. CalcuSyn software was utilized to calculate Combination Index (CI), CI < 1 was considered as synergism.
Results
Perifosine Induces Growth Inhibition in Cultured Osteosarcoma Cells
Perifosine has been effective in killing multiple cancer cells both in vivo and in vitro [3–7], we first tested its effect in osteosarcoma cells. “MTT” cell viability assay results in Fig. 1a demonstrated that perifosine dose-dependently reduced cell viability in three osteosarcoma cell lines (SaOs-2, U2OS, and MG-63). U2OS is the most sensitive line. U2OS cell viability reduced to 25, 43, 32, and 17 % of control level after 1, 5, 10, and 25 μM of perifosine treatment, respectively (Fig. 1a). Live cell count data in Fig. 1b further confirmed perifosine’s ability against osteosarcoma cells.
Perifosine induces growth inhibition in cultured osteosarcoma cells. Three different osteosarcoma cell lines SaOs-2, U2OS and MG-63 were either left untreated (Ctrl) or treated with different concentration of perifosine (1, 5, 10 and 25 μM) for 48 h, cell viability was analyzed by MTT assay (a), after treatment, dead cells were washed away and left viable cells were counted using a haemocytometer as shown in (b). The data in this figure were representatives of more than three different experiments, and similar results were obtained. The values in the figures are expressed as the means ± standard deviation (SD). * Values of p < 0.05 vs. Ctrl group for U2OS cells, ** Values of p < 0.05 vs. Ctrl group for SaOs-2 cells, *** Values of p < 0.05 vs. Ctrl group for MG-63 cells
Perifosine Promotes Apoptosis in Cultured Osteosarcoma Cells
We used multiple apoptosis detecting assays including Hoechst staining apoptotic nuclear (Fig. 2a), caspase-3 activity assay (Fig. 2b), Histone DNA-ELISA assay (Fig. 2c), and Western blots detecting apoptosis associated proteins (Fig. 2d), our results confirmed that perifosine dose-dependently promoted U2OS cell apoptosis (Fig. 2a–d). Perifosine increased caspase-3 cleavage and activity (Fig. 2b, d); meanwhile, perifosine decreased the expression of anti-apoptosis proteins including Bcl-2 and Bcl-xl (Fig. 2d). Two caspase inhibitors Z-VAD-fmk (mainly against caspase-3) and Z-DEVD-fmk (a general caspase inhibitor) significantly reduced perifosine-induced U2OS cell viability loss (Fig. 2e) and apoptosis (Fig. 2f).
Perifosine promotes apoptosis in cultured osteosarcoma cells. U2OS osteosarcoma cells were either left untreated (Ctrl) or administrated with indicated concentration of perifosine (1, 5, 10 and 25 μM) for 30 or 36 h, cell apoptosis was analyzed by three different apoptosis assays including Hoechst nuclear staining (a), caspase-3 activity assay (b) and Histone-DNA ELISA assay (c). Apoptosis associated proteins including cleaved caspase-3, cleaved caspase-9, Bax, Bcl-2 and Bcl-xl were also measured by Western blot, with tubulin as loading control (d). Effects of two different caspase inhibitors Z-VAD-fmk (50 μM) and Z-DEVD-fmk (50 μM) on perifosine-induced U2OS viable cell lose (e) and apoptosis (f) were also shown. Blot intensity was quantified using Image J software, and the number was normalized to Ctrl group. The data in this figure were representatives of more than 3 different experiments, similar results were obtained. The values in the figures are expressed as the means ± standard deviation (SD). * Values of p < 0.05 compared with Ctrl group. # p < 0.05. For each Hoechst experiment, at least 400 cells in 10 random scope fields were counted for apoptotic rate (Magnification 1: 100) (a)
Perifosine Blocks Akt/mTOR Activation in Cultured Osteosarcoma Cells
Activation of Akt and downstream mTOR signal pathway is important for osteosarcoma cell survival, growth, and apoptosis resistance [14–17]. Perifosine is known to block Akt activation [3, 18]; next, we tested the effect of perifosine on Akt and downstream mTORC1 (mTOR complex 1) activation in cultured osteosarcoma cells. In all three osteosarcoma cell lines (SaOs-2, U2OS, and MG-63), perifosine dose-dependently inhibited Akt phosphorylation (activation) (Fig. S1). Western blot results in Fig. 3a demonstrated that perifosine almost blocked Akt, GSK3α/β, and S6 (mTORC1 activation indicator) phosphorylation in U2OS osteosarcoma cells. As expected, three different PI3K (Phosphoinositide 3-kinase)/Akt inhibitors LY 294002 (LY), wortmannin (WT), and Akt inhibitor X (Akt X) also blocked activation of Akt/mTORC1 pathway. However, compared to perifosine, Akt inhibitors caused a less cell viability loss and apoptosis in U2OS cells (Fig. 3c–d), which suggests that other mechanisms besides Akt inactivation should also contribute to the cytotoxic effects of perifosine.
Perifosine blocks Akt/mTOR activation in cultured osteosarcoma cells. U2OS osteosarcoma cells were either left untreated (Ctrl) or treated with indicated concentration of perifosine or one of the three different PI3K/Akt inhibitors LY 294002 (LY, 10 μM), wortmannin (WT, 10 μM) and Akt inhibitor X (Akt X, 10 μM) for 12 h, activation of Akt/mTOR pathways were measured by Western blots detecting indicated phospho-proteins. Loading controls including tubulin, Akt1, GSK3, S6 and β-actin were also measured. Effects of three different Akt inhibitors and perifosine (10 μM) on U2OS cell viability loss (c, “MTT” assay, 48 h) and apoptosis (d, Histone DNA-ELISA assay, 36 h) were shown. The data in this figure were representatives of more than three different experiments, similar results were obtained. The values in the figures are expressed as the means ± standard deviation (SD). # p < 0.05
Perifosine Activates JNK and p53 in Cultured Osteosarcoma Cells
As shown previously, blocking of PI3K/Akt activation by three different pharmacological inhibitors caused a much less osteosarcoma cell death compared to perifosine treatment, indicating that other mechanisms may also be involved. The Stress-Activated Protein Kinase (SAPK)/JNK pathway, mainly activated by cellular stress, plays an important role in mediating cell apoptosis [8]. Recent studies have shown that perifosine activates JNK [4, 11]. Expression of the dominant-negative form of c-Jun, a substrate of SAPK/JNK, nullified the apoptosis-inducing activity of perifosine [4]. We then tested the effect of perifosine on JNK activation in osteosarcoma cells. As shown in Fig. 4a–b, perifosine induced a significant JNK activation and c-fos, c-Jun up-regulation in U2OS cells, which was reversed by JNK inhibitor SP 600125. Notably, antioxidant MnTBAP largely inhibited perifosine-induced JNK activation (Fig. 4a–b), suggesting that reactive oxygen species (ROS) production might be involved in JNK activation by perifosine. Both SP 600125 and MnTBAP reduced perifosine-induced U2OS cell death (Fig. 4c) and apoptosis (Fig. 4d), suggesting that activation of JNK pathway contributes to perifosine-induced anti-U2OS effect. Further, perifosine induced p53 activation (phosphorylation and up-regulation) in U2OS cells (Fig. 4e–f). The blocking of JNK activation by SP 600125 reduced perifosine-induced p53 activation (Fig. 5g–h). These results imply that, beside Akt inhibition, perifosine may induce a ROS-JNK-p53 pro-apoptosis pathway to mediate cell apoptosis.
Perifosine activates JNK and p53 in cultured osteosarcoma cells. U2OS osteosarcoma cells were pretreated with JNK inhibitor SP 600125 (10 μM) or antioxidant MnTBAP (25 μM) for 1 h, followed by perifosine treatment (10 μM) for 6 and 12 h, activation of JNK and c-Jun as well as the expression of c-fos and c-jun were measured by Western blots, with Akt1 as loading control. Effects of JNK inhibitor SP 600125 (10 μM, 1 h pretreatment) or antioxidant MnTBAP (25 μM, 1 h pretreatment) on U2OS cell viability loss (c, “MTT” assay) and apoptosis (d, Histone DNA-ELISA assay) were also shown. U2OS cells were treated with perifosine (10 μM) for 24 and 48 h, phosphorylation and total level of p53 were measured (e–f), the effect of SP 600125 (10 μM, 1 h pretreatment) on perifosine (10 μM) induced p53 activation were shown in (g–h). Blot intensity was quantified using Image J software. The data in this figure were representatives of more than three different experiments, similar results were obtained. # p < 0.05
Perifosine induces survivin down-regulation in cultured osteosarcoma cells. U2OS osteosarcoma cells were either left untreated (Ctrl) or treated with perifosine (10 μM) for indicated time points (6 h, 12 h, 24 h, 36 h and 48 h) or treated with different concentration of perifosine (1, 5, 10 and 25 μM) for 36 h, expression level of survivin was measured by Western blots with Akt1 as loading control, survivin expression level was quantified using Image J software (a–b). The association between survivin and HSP-90 after perifosine (10 μM) treatment was measured by immunoprecipitation (IP) assay (c). d U2OS osteosarcoma cells were either left untreated (Ctrl) or treated with three different PI3K/Akt inhibitors LY 294002 (LY, 10 μM), wortmannin (WT, 10 μM) and Akt inhibitor X (Akt X, 10 μM) or two HSP-90 inhibitors 17-AAG (2 μM), 17-DMAG (10 μM) for 36 h, the expression level of survivin was measured. The effects of 17-AAG (2 μM), 17-DMAG (10 μM) or perifosine (10 μM) on U2OS cell viability loss (e, MTT assay) and apoptosis (f, Histone DNA-ELISA assay) were shown. The data in this figure were representatives of more than three different experiments, similar results were obtained. The values in the figures are expressed as the means ± standard deviation (SD). # p < 0.05
Perifosine Induces Survivin Down-regulation in Cultured Osteosarcoma Cells
Survivin is a structurally unique member of the inhibitor of apoptosis protein family, which is important for mitotic progression, cancer survival, and apoptosis inhibition. Its marked expression in cancers versus normal tissues and its association with unfavorable disease outcome have made survivin a promising new target for anti-cancer interventions [19–21]. Survivin is important for osteosarcoma cell survival, growth, and apoptosis resistance [22–25], and down-regulating survivin induces osteosarcoma cell apoptosis and growth inhibition [23–25]. We then tested perifosine’s effect on survivin expression in cultured U2OS osteosarcoma cells, as shown in Fig. 5a–b; perifosine down-regulated survivin expression in U2OS cells. Heat shock protein-90 (HSP-90) is a ubiquitous molecular chaperone that promotes the conformational maturation and stabilization of numerous client proteins such as survivin, deregulation of this process, by its inhibitors 17-AAG and 17-DMAG for example, prevents the stabilization of client proteins, ultimately resulting in their degradation [26]. Here we found that perifosine led to the down-regulation of survivin (Fig. 5a–b), probably by disassociating it with HSP-90 (Fig. 5c). HSP-90 inhibitors 17-AAG and 17-DMAG, but not Akt inhibitors (WT, LY and Akt X), caused survivin down-regulation (Fig. 5d). Further, 17-AAG and 17-DMAG both induced significant U2OS cell viability loss and apoptosis, though both inhibitors were not as efficient as perifosine (Fig. 5e–f).
Perifosine’s Effect on Ceramide Production in Cultured Osteosarcoma Cells
Recent studies demonstrate that perifosine induces ceramide accumulation through de novo synthesis pathway, which contributes to cell apoptosis [11, 13]. We tested whether this was also true in osteosarcoma cells. Perifosine administration resulted in short term (24 h) and moderate ceramide production (less than three folds v.s. Ctrl); however, the ceramide level went back after 48 h (Fig. 6a). Interestingly, the ceramide de novo synthase inhibitor fumonisin B1(F-B1) [27, 28], which almost blocked ceramide accumulation (Fig. 6a), had almost no effect on perifosine-induced cell death and apoptosis in U2OS cells (Fig. 6b–c), suggesting that ceramide production may not be so critical for perifosine-induced cell death in cultured osteosarcoma cells. As a matter of fact, when we added short-chain cell-permeable C2 ceramide to osteosarcoma cells directly, a relative large amount of C2 ceramide (10 μg/ml) was needed to kill the least amount of osteosarcoma cells (30 %) (Fig. 6d). However, with further increase in cellular ceramide level by exogenously adding short-chain ceramide (C2), or by administration of the ceramide metabolism inhibitor l-threo-1-phenyl-2-decanoylamino-3-morpholino-1-propanol (PDMP), more cytotoxicity was achieved (Fig. 6e). Both C2 ceramide and PDMP enhanced perifosine-induced U2OS cytotoxicity (Fig. 6e). Based on these data, we speculate that the moderate ceramide accumulation by perifosine is not necessary for its cytotoxic effects; however, further increase in ceramide production can enhance perifosine-induced U2OS cell death. Figure 6f confirmed enhanced ceramide production by PDMP in perifosine-treated U2OS cells.
Perifosine’s effect on ceramide production in cultured osteosarcoma cells. U2OS osteosarcoma cells were pretreated with fumonisin B1 (F-B1, 15 μM) for 1 h, followed by perifosine treatment (10 or 25 μM) for indicated time points, ceramide level was measured (a), cell viability loss (b, “MTT” assay) and cell apoptosis (c, Histone DNA ELISA) were measured. Effects of C2 ceramide on U2OS viability was measured by MTT assay (d). U2OS cells were pretreated with C2 Ceramide (5 μg/ml) or PDMP (20 μM) for 1 h, followed by perifosine (10 μM) treatment, cell viability was analyzed by “MTT” assay 48 h after incubation (e), and cellular ceramide level was measured after 24 h incubation (f). The data in this figure were representatives of more than three different experiments, similar results were obtained. The values in the figures are expressed as the means ± standard deviation (SD). * Values of p < 0.05 compared with Ctrl group (d). # p < 0.05
Chemo-sensitization Effect of Perifosine Against Osteosarcoma Cells
Studies show that perifosine sensitizes multiple anti-cancer agents including paclitaxel (Taxol) [11], histone deacetylase inhibitor (HDACi) [13], and soy isoflavones [29]. It also enhances radiation-induced cell apoptosis [30]. Doxorubicin and etoposide are two clinically used anti-osteosarcoma drugs [2], we then tested perifosine’s effect on doxorubicin and etoposide in cultured osteosarcoma cells. Results in Fig. 7a–b demonstrated that a relative low dose of perifosine (0.5–1 μM) significantly enhanced etoposide-induced U2OS cell death (viability loss) (Fig. 7a–b), with combination index (CI) < 1 indicating synergism. Further, perifosine facilitated doxorubicin-induced U2OS cell death, as co-administration of perifosine with doxorubicin caused more cell viability loss than either single agent alone (Fig. 7c) (CI) < 1. Perifosine also enhanced etoposide- or doxorubicin-induced apoptosis in U2OS (Fig. 7d), SaOs-2 (Fig. 7e), and MG-63 (Fig. 7f) osteosarcoma cell lines. These results suggest that perifosine sensitized doxorubicin/etoposide’s anti-osteosarcoma effect in vitro.
Chemo-sensitization effect of perifosine against osteosarcoma cells. U2OS osteosarcoma cells were treated with the combination of various concentrations of etoposide and perifosine, cells were cultured for indicated time period, cell viability loss was measured by MTT assay (a and b). U2OS cells were treated with treated with doxorubicin (10 μM) in the presence or absence of perifosine (1 μM), cell viability was measured by MTT assay (c). U2OS, SaOs-2 and MG-63 osteosarcoma cells were treated with doxorubicin (10 μM), etoposide (5 μM) with or without perifosine (1 μM) for 36 h, cell apoptosis was analyzed by Histone-DNA ELISA assay (d, e and f). The data in this figure were representatives of more than three different experiments, similar results were obtained. The values in the figures are expressed as the means ± standard deviation (SD). * Values of p < 0.05 compared with Ctrl group (a–b). # p < 0.05
Discussion
In cultured human osteosarcoma cells, we demonstrate that perifosine blocks Akt/mTORC1 activation, while inducing pro-apoptotic caspase-3, JNK, and p53 activation. Perifosine induces down-regulation of survivin probably by disrupting its association with HSP-90. These signal events together may count for a marked increase of osteosarcoma cell apoptosis. Further, low dose of perifosine sensitizes etoposide- or doxorubicin-induced osteosarcoma cell apoptosis in vitro. Our study suggests that perifosine induces a short-term moderate ceramide accumulation in osteosarcoma cells, which by itself may not be enough to promote cell apoptosis.
The Akt/mTOR signal transduction is critical to control processes integral in cancer development, such as protein translation, growth, metabolism, therapeutic resistance, and survival [31, 32]. This pathway is frequently over-expressed or over-activated in osteosarcoma, providing a strong rationale to target it in cancer therapy [31, 32].
Several Akt inhibitors have entered early-phase clinical trials with few success due to less response or unexpected toxicity, these inhibitors include triciribine, which is a nucleoside analog that causes dephosphorylation of active Akt, and GSK690693, which are adenosine triphosphate (ATP)-competitive inhibitors [33]. In addition, there are other ATP-competitive and allosteric Akt inhibitors in preclinical development [34, 35]. A potentially more selective and novel approach is to target the lipid-binding PH domain of Akt, PH domain is essential for Akt translocation and activation. Perifosine and other lipid-based Akt inhibitors are developed based on this rationale [36]. In vitro, perifosine inhibits Akt at low micromolar concentrations [36, 37], and inhibition has been measured in many xenograft models in vivo [36, 37]. Here, we found that perifosine blocked pro-survival Akt/mTOR complex 1 (mTORC1) activation in osteosarcoma cells. However, three different pharmacological inhibitors, LY 294002, wortmanin, and Akt inhibitor X, caused a much less cell viability loss and apoptosis compared to perifosine treatment, which suggests that other mechanisms besides Akt/mTOR inhibition may also contribute to perifosine’s anti-osteosarcoma cell effects.
A number of recent studies have confirmed that activation of JNK pathway is important for perifosine-induced apoptosis [11, 31, 38], and expression of a dominant-negative form of c-Jun, a substrate of SAPK/JNK, nullified the apoptosis-inducing activity of perifosine [4]; JNK inhibitor also limited perifosine’s pro-apoptosis effect [11]. We found that perifosine induced JNK activation and c-fos, c-Jun accumulation in cultured osteosarcoma cells. Interestingly, antioxidant MnTBAP largely inhibited JNK activation to suggest that ROS production may be involved in this process. As a matter of fact, recent studies have shown that perifosine facilitates ROS production, which is involved in JNK activation [13, 39] and cancer cell apoptosis [13, 38, 39]. The fact that JNK inhibitor and antioxidant MnTBAP inhibited perifosine-induced osteosarcoma cell apoptosis suggests that JNK activation is involved in this process. Under a number of stimuli, p53 activation promotes osteosarcoma cell apoptosis [40, 41]. Activation of JNK is involved in this process. Notably, compared to U2OS cells where p53 is normal, perifosine induced less cell death and apoptosis in MG-63 osteosarcoma cells that are deficient in p53 to imply that p53 accumulation is involved in perifosine-induced cell apoptosis. More evidence is needed to support this.
One of the major characters of cancer that promotes enhanced adaptation to environmental challenges is a constitutive up-regulation of the cellular stress response. This preserves folding of nascent polypeptides, prevents protein aggregation, and ensures specialized intracellular trafficking of client proteins [26, 42–44]. The protein folding quality control machinery is orchestrated by HSP, a family of evolutionary conserved ATPase-directed molecular chaperones. In particular, HSP-90 controls the balance between folding/maturation and proteasomal destruction of a restricted number of client proteins that are typically involved in signal transduction and cell proliferation [26, 42–44]. One of the client proteins is survivin. Disruption of the survivin–HSP-90 interaction with specific inhibitors or antibodies of HSP-90 destabilizes survivin, promotes survivin ubiquitin degradation, and initiates mitochondrial apoptosis. Here our co-IP results confirmed that perifosine disrupted the association between survivin and HSP-90 in osteosarcoma cells, which led to survivin degradation to contribute to cell apoptosis.
Abbreviations
- mTORC1:
-
mTOR complex 1
- JNK:
-
c-Jun N-terminal kinases
- ERK:
-
Extracellular signal-regulated kinase
- MTT:
-
3-[4,5-dimethylthylthiazol-2-yl]-2,5 diphenyltetrazolium bromide
- HSP-90:
-
Heat shock protein-90
- PBS:
-
Phosphate-buffered saline
- ROS:
-
Reactive oxygen species
- HDACi:
-
Histone deacetylase inhibitor
- PI3K:
-
Phosphoinositide 3-kinase
References
Ham, S. J., Schraffordt Koops, H., van der Graaf, W. T., van Horn, J. R., Postma, L., & Hoekstra, H. J. (1998). Historical, current and future aspects of osteosarcoma treatment. European Journal of Surgical Oncology, 24, 584–600.
Rosen, G., Marcove, R. C., Caparros, B., Nirenberg, A., Kosloff, C., & Huvos, A. G. (1979). Primary osteogenic sarcoma: The rationale for preoperative chemotherapy and delayed surgery. Cancer, 43, 2163–2177.
Kondapaka, S. B., Singh, S. S., Dasmahapatra, G. P., Sausville, E. A., & Roy, K. K. (2003). Perifosine, a novel alkylphospholipid, inhibits protein kinase B activation. Molecular Cancer Therapeutics, 2, 1093–1103.
Ruiter, G. A., Zerp, S. F., Bartelink, H., van Blitterswijk, W. J., & Verheij, M. (1999). Alkyl-lysophospholipids activate the SAPK/JNK pathway and enhance radiation-induced apoptosis. Cancer Research, 59, 2457–2463.
Cirstea, D., Hideshima, T., Rodig, S., Santo, L., Pozzi, S., Vallet, S., et al. (2011). Dual inhibition of akt/mammalian target of rapamycin pathway by nanoparticle albumin-bound-rapamycin and perifosine induces antitumor activity in multiple myeloma. Molecular Cancer Therapeutics, 9, 963–975.
Crul, M., Rosing, H., de Klerk, G. J., Dubbelman, R., Traiser, M., Reichert, S., et al. (2002). Phase I and pharmacological study of daily oral administration of perifosine (D-21266) in patients with advanced solid tumours. European Journal of Cancer, 38, 1615–1621.
Van Ummersen, L., Binger, K., Volkman, J., Marnocha, R., Tutsch, K., Kolesar, J., et al. (2004). A phase I trial of perifosine (NSC 639966) on a loading dose/maintenance dose schedule in patients with advanced cancer. Clinical Cancer Research, 10, 7450–7456.
Fei, H. R., Chen, G., Wang, J. M., & Wang, F. Z. (2010). Perifosine induces cell cycle arrest and apoptosis in human hepatocellular carcinoma cell lines by blockade of Akt phosphorylation. Cytotechnology, 62, 449–460.
Papa, V., Tazzari, P. L., Chiarini, F., Cappellini, A., Ricci, F., Billi, A. M., et al. (2008). Proapoptotic activity and chemosensitizing effect of the novel Akt inhibitor perifosine in acute myelogenous leukemia cells. Leukemia, 22, 147–160.
Chiarini, F., Del Sole, M., Mongiorgi, S., Gaboardi, G. C., Cappellini, A., Mantovani, I., et al. (2008). The novel Akt inhibitor, perifosine, induces caspase-dependent apoptosis and downregulates P-glycoprotein expression in multidrug-resistant human T-acute leukemia cells by a JNK-dependent mechanism. Leukemia, 22, 1106–1116.
Sun, H., Yu, T., & Li, J. (2011). Co-administration of perifosine with paclitaxel synergistically induces apoptosis in ovarian cancer cells: More than just AKT inhibition. Cancer Letters, 310, 118–128.
Kumar, A., Fillmore, H. L., Kadian, R., Broaddus, W. C., Tye, G. W., & Van Meter, T. E. (2009). The alkylphospholipid perifosine induces apoptosis and p21-mediated cell cycle arrest in medulloblastoma. Molecular Cancer Research, 7, 1813–1821.
Rahmani, M., Reese, E., Dai, Y., Bauer, C., Payne, S. G., Dent, P., et al. (2005). Coadministration of histone deacetylase inhibitors and perifosine synergistically induces apoptosis in human leukemia cells through Akt and ERK1/2 inactivation and the generation of ceramide and reactive oxygen species. Cancer Research, 65, 2422–2432.
Paoloni, M. C., Mazcko, C., Fox, E., Fan, T., Lana, S., Kisseberth, W., et al. (2011). Rapamycin pharmacokinetic and pharmacodynamic relationships in osteosarcoma: A comparative oncology study in dogs. PLoS ONE, 5, e11013.
Zhang, B., Shi, Z. L., Liu, B., Yan, X. B., Feng, J., & Tao, H. M. (2010). Enhanced anticancer effect of gemcitabine by genistein in osteosarcoma: The role of Akt and nuclear factor-kappaB. Anti-Cancer Drugs, 21, 288–296.
Zhou, R., Zhang, Z., Zhao, L., Jia, C., Xu, S., Mai, Q., et al. (2010). Inhibition of mTOR signaling by oleanolic acid contributes to its anti-tumor activity in osteosarcoma cells. Journal of Orthopaedic Research, 29, 846–852.
Jin, S., Pang, R. P., Shen, J. N., Huang, G., Wang, J., & Zhou, J. G. (2007). Grifolin induces apoptosis via inhibition of PI3K/AKT signalling pathway in human osteosarcoma cells. Apoptosis, 12, 1317–1326.
Hideshima, T., Catley, L., Yasui, H., Ishitsuka, K., Raje, N., Mitsiades, C., et al. (2006). Perifosine, an oral bioactive novel alkylphospholipid, inhibits Akt and induces in vitro and in vivo cytotoxicity in human multiple myeloma cells. Blood, 107, 4053–4062.
Altieri, D. C. (2003). Survivin, versatile modulation of cell division and apoptosis in cancer. Oncogene, 22, 8581–8589.
Altieri, D. C., & Marchisio, P. C. (1999). Survivin apoptosis: An interloper between cell death and cell proliferation in cancer. Laboratory Investigation, 79, 1327–1333.
Altieri, D. C. (2003). Validating survivin as a cancer therapeutic target. Nature Reviews Cancer, 3, 46–54.
Trieb, K., Lehner, R., Stulnig, T., Sulzbacher, I., & Shroyer, K. R. (2003). Survivin expression in human osteosarcoma is a marker for survival. European Journal of Surgical Oncology, 29, 379–382.
Wu, Y. F., Liang, X. J., Liu, Y. Y., Gong, W., Liu, J. X., Wang, X. P., et al. (2010). +Antisense oligonucleotide targeting survivin inhibits growth by inducing apoptosis in human osteosarcoma cells MG-63. Neoplasma, 57, 501–506.
Zhang, H. Z., Wang, Y., Gao, P., Lin, F., Liu, L., Yu, B., et al. (2006). Silencing stathmin gene expression by survivin promoter-driven siRNA vector to reverse malignant phenotype of tumor cells. Cancer Biology & Therapy, 5, 1457–1461.
Zou, J., Gan, M., Mao, N., Zhu, X., Shi, Q., & Yang, H. (2010). Sensitization of osteosarcoma cell line SaOS-2 to chemotherapy by downregulating survivin. Archives of Medical Research, 41, 162–169.
Fortugno, P., Beltrami, E., Plescia, J., Fontana, J., Pradhan, D., Marchisio, P. C., et al. (2003). Regulation of survivin function by Hsp90. Proceedings of the National Academy of Sciences of the United States of America, 100, 13791–13796.
Merrill, A. H., Jr, van Echten, G., Wang, E., & Sandhoff, K. (1993). Fumonisin B1 inhibits sphingosine (sphinganine) N-acyltransferase and de novo sphingolipid biosynthesis in cultured neurons in situ. Journal of Biological Chemistry, 268, 27299–27306.
Charles, A. G., Han, T. Y., Liu, Y. Y., Hansen, N., Giuliano, A. E., & Cabot, M. C. (2001). Taxol-induced ceramide generation and apoptosis in human breast cancer cells. Cancer Chemotherapy and Pharmacology, 47, 444–450.
Vinall, R. L., Hwa, K., Ghosh, P., Pan, C. X., Lara, P. N., Jr, & de Vere White, R. W. (2007). Combination treatment of prostate cancer cell lines with bioactive soy isoflavones and perifosine causes increased growth arrest and/or apoptosis. Clinical Cancer Research, 13, 6204–6216.
Ruiter, G. A., Verheij, M., Zerp, S. F., & van Blitterswijk, W. J. (2001). Alkyl-lysophospholipids as anticancer agents and enhancers of radiation-induced apoptosis. International Journal of Radiation Oncology Biology Physics, 49, 415–419.
Vivanco, I., & Sawyers, C. L. (2002). The phosphatidylinositol 3-Kinase AKT pathway in human cancer. Nature Reviews Cancer, 2, 489–501.
Hennessy, B. T., Smith, D. L., Ram, P. T., Lu, Y., & Mills, G. B. (2005). Exploiting the PI3K/AKT pathway for cancer drug discovery. Nature Reviews Drug Discovery, 4, 988–1004.
Garcia-Echeverria, C., & Sellers, W. R. (2008). Drug discovery approaches targeting the PI3K/Akt pathway in cancer. Oncogene, 27, 5511–5526.
LoPiccolo, J., Granville, C. A., Gills, J. J., & Dennis, P. A. (2007). Targeting Akt in cancer therapy. Anti-Cancer Drugs, 18, 861–874.
Lu, Y., Wang, H., & Mills, G. B. (2003). Targeting PI3K-AKT pathway for cancer therapy. Reviews in Clinical & Experimental Hematology, 7, 205–228.
Gills, J. J., & Dennis, P. A. (2009). Perifosine: Update on a novel Akt inhibitor. Current Oncology Reports, 11, 102–110.
Hennessy, B. T., Lu, Y., Poradosu, E., Yu, Q., Yu, S., Hall, H., et al. (2007). Pharmacodynamic markers of perifosine efficacy. Clinical Cancer Research, 13, 7421–7431.
Wang, Q., Wang, X., Hernandez, A., Hellmich, M. R., Gatalica, Z., & Evers, B. M. (2002). Regulation of TRAIL expression by the phosphatidylinositol 3-kinase/Akt/GSK-3 pathway in human colon cancer cells. Journal of Biological Chemistry, 277, 36602–36610.
Tazzari, P. L., Tabellini, G., Ricci, F., Papa, V., Bortul, R., Chiarini, F., et al. (2008). Synergistic proapoptotic activity of recombinant TRAIL plus the Akt inhibitor Perifosine in acute myelogenous leukemia cells. Cancer Research, 68, 9394–9403.
Simbulan-Rosenthal, C. M., Rosenthal, D. S., Luo, R., & Smulson, M. E. (1999). Poly(ADP-ribosyl)ation of p53 during apoptosis in human osteosarcoma cells. Cancer Research, 59, 2190–2194.
Schuler, M., Bossy-Wetzel, E., Goldstein, J. C., Fitzgerald, P., & Green, D. R. (2000). p53 induces apoptosis by caspase activation through mitochondrial cytochrome c release. Journal of Biological Chemistry, 275, 7337–7342.
Workman, P., & Powers, M. V. (2007). Chaperoning cell death: A critical dual role for Hsp90 in small-cell lung cancer. Nature Chemical Biology, 3, 455–457.
Whitesell, L., & Lindquist, S. L. (2005). HSP90 and the chaperoning of cancer. Nature Reviews Cancer, 5, 761–772.
Neckers, L. (2002). Hsp90 inhibitors as novel cancer chemotherapeutic agents. Trends in Molecular Medicine, 8, S55–61.
Conflict of interest
The authors declare no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
Perifosine inhibits Akt phosphorylation in cultured osteosarcoma cells. U2OS, SaOs-2 and MG-63 osteosarcoma cells were treated with indicated concentration of perifosine for 12 h, p-Akt (Ser 473) and Akt1 were examined by Western blot (EPS 814 kb)
Rights and permissions
About this article
Cite this article
Yao, C., Wei, Jj., Wang, Zy. et al. Perifosine Induces Cell Apoptosis in Human Osteosarcoma Cells: New Implication for Osteosarcoma Therapy?. Cell Biochem Biophys 65, 217–227 (2013). https://doi.org/10.1007/s12013-012-9423-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-012-9423-5