Abstract
To date, no study has critically reviewed the current literature on the association between magnesium (Mg) and sleep health. Therefore, we carried out a systematic review to assess the association between Mg and sleep patterns in adults’ population through observational and interventional studies. We searched for relevant studies through PubMed (http://www.ncbi.nlm.nih.gov/pubmed), Scopus (http://www.scopus.com), and ISI Web of Science (http://www.webofscience.com) from the earliest available date until November 2021. Eligibility criteria for study selection were guided by the following components identified using the PI(E)CO (Population, Intervention (Exposure), Comparison, Outcome) framework: P (adult population), I(E) (high dietary intake or supplementation of Mg), C (low dietary intake of Mg or placebo group), and O (sleep pattern including sleep duration, sleep-onset latency, night awakenings, sleep stages, and sleep phases). The present study involved 7,582 subjects from 9 published cross-sectional, cohort, and RCT systematically reviewed the possible links between Mg and sleep quality (daytime falling asleep, sleepiness, snoring, and sleep duration) in an adult population. Observational studies suggested an association between Mg statuses and sleep quality, while the RCTs reported contradictory findings. This systematic review revealed an association between magnesium status and sleep quality (daytime falling asleep, sleepiness, snoring, and sleep duration) according to the observational studies, while the randomized clinical trials showed an uncertain association between magnesium supplementation and sleep disorders. The association between dietary magnesium and sleep patterns needs well-designed randomized clinical trials with a larger sample size and longer follow-up time (more than 12 weeks) to further clarify the relationship.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sleep health, in terms of quantity and quality, is critical for a range of health and well-being outcomes, throughout the entire lifespan [1]. Sleep disturbances and insufficient sleep have been shown to be linked with a variety of chronic conditions [2], and with a higher rate of all-cause mortality [3, 4]. Besides, sleep problems are associated with accidents and human errors [5, 6]. These reasons, among others, underlie the increasing interest in sleep among scholars.
Various studies have pointed out the associations between intake of dietary macro-/micronutrients and sleep outcomes. For instance, carbohydrates with high glycemic index have been linked to sleep onset [7], whereas low fiber, high saturated fat, and sugar intake are associated with lighter and less restorative sleep with more arousals [8, 9]. Also, consumption of some micronutrients such as iron and zinc has positively been associated with sleep duration, but vitamin K and B12 have shown negative links [10].
Magnesium (Mg), a micronutrient mainly found in green leafy vegetables, whole grains, nuts, and legumes, is a cofactor involved in various enzymatic reactions and regulating biochemical pathways in the human body [11]. Low levels of Mg have been associated with a number of chronic diseases including migraine headaches, Alzheimer’s disease, cerebrovascular accident (CVA), hypertension, cardiovascular disease (CVD), type 2 diabetes mellitus (T2DM), and premenstrual syndrome [12].
More recently, Mg has been found to be beneficial in maintaining the normal circadian rhythms and sleep quality [13], though the results have been mixed. For example, in a study conducted on adults in Turkey, dietary intake of Mg was significantly higher in individuals with better sleep quality compared to those with poor sleep quality [14]. However, there are some datasets failing to show any association between Mg and sleep quality or quantity [15, 16].
To date, no study has critically reviewed the current literature on the association between Mg and sleep health. Moreover, the findings of previous studies are contradictory which precludes the clinicians to reach a firm and practical conclusion in this regard. Therefore, we carried out a systematic review to assess the association between Mg and sleep patterns in adults’ population through observational and interventional studies.
Methods
Data Source and Search Strategy
The present study was designed and conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement checklist [17]. We searched for relevant studies through PubMed (http://www.ncbi.nlm.nih.gov/pubmed), Scopus (http://www.scopus.com), and ISI Web of Science (http://www.webofscience.com) from the earliest available date until November 2021, using the following keywords: (Magnesium) AND (Sleep OR Insomnia OR Dyssomnia) (Table 1). The search terms were selected in consultation with expert scientists in the field of Mg and sleep to increase the relevance search strategy and to reduce the chance of missing relevant articles. The reference lists of eligible articles were also manually checked to diminish the chance of missing relevant studies. No restriction was made on publication year during the database searching.
Study Selection and Eligibility Criteria
Eligibility criteria for study selection were guided by the following components identified using the PI(E)CO (Population, Intervention (Exposure), Comparison, Outcome) framework: P (adult population), I(E) (high dietary intake or supplementation of Mg), C (low dietary intake of Mg or placebo group), and O (sleep pattern including sleep duration, sleep-onset latency, night awakenings, sleep stages, and sleep phases).
The inclusion criteria were as follows: (1) original human studies either with observational or interventional design; (2) published in the English language; and (3) which explored the relationship between Mg and sleep pattern among the adult population. The exclusion criteria were as follows: (1) non-human studies; (2) case reports, poster abstract, editorial, and review articles; and (3) studies that assessed physiological sleep disorders.
Data Extraction
Data extraction was done in duplicate and blinded approach, using a predefined excel form by two independent investigators (A.A and F.S). The following information was extracted from each of the eligible studies: first author’s name, study year, country of origin, sample size, study design, participant features (i.e., age and sex), main findings, and strength and limitation. The included studies were categorized based on the design into observational studies (Table 2) and randomized controlled trials (RCTs) (Table 3).
Quality Assessment
The critical appraisal of the eligible studies was performed using the Newcastle–Ottawa Quality Assessment Scale (NOS) [18] for observational studies and the Cochrane Collaboration tool for RCTs [19]. The NOS consists of three domains including selection, comparability, and outcome for a total score of 9 points for cohort and 10 for cross-sectional studies (≥ 7, 4–6, and ≤ 3 points were identified as high, moderate, and low quality) [20]. The Cochrane Collaboration tool consisted of random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessments, incomplete outcome data, selective reporting, and other biases. Each item was scored as low, unclear, or high risk of bias.
Results
Search Results
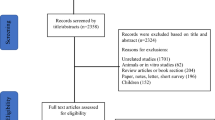
The initial search through databases identified 934 papers. After removing duplicates, the remaining 289 articles were reviewed based on the title and abstract by two independent reviewers (A.A and F.S). Totally, 19 articles were retrieved and reviewed based on full-text, and finally, nine studies met our inclusion criteria and entered this systematic review. The flow diagram of the study selection process is illustrated in Fig. 1.
Overview of Included Studies
Studies were conducted between 1999 and 2019 involving a total of 8398 participants. Among nine included studies, three were cross-sectional [14, 21, 22], one was a cohort study [13], and the other five datasets were RCTs [15, 16, 23,24,25]. Based on the country, two were from China [13, 21], two from the USA [16, 24], and the others were obtained from Turkey [14], Japan [22], Argentina [24], Iran [25], and Germany [23]. The Pittsburgh Sleep Quality Index (PSQI) was used to assess sleep quality in four studies [14, 16, 21, 24]. One study used the Epworth Sleepiness Scale (ESS) [21], one examined sleep electroencephalogram (EGG) [23], and one study used the Insomnia Severity Index (ISI) questionnaire [25]. In the remaining datasets, a self-report of sleep quality or quantity was used [13, 15, 22]. All studies recruited healthy subjects, except for one that included peritoneal dialysis patients [21]. The general characteristics of the included studies are presented in Tables 2 and 3.
Findings from the Systematic Review
Observational Studies
The association between Mg and sleep was examined in one cohort and three cross-sectional studies.
In the first cross-sectional study conducted on 3262 adults aged between 20 and 64 years in Turkey, consumption of Mg was significantly higher in individuals with better sleep quality compared to those with poor sleep quality assessed by the PSQI (P = 0.037). However, dietary Mg was not correlated with sleep duration [14].
In 2014, Lai et al. recruited 98 peritoneal dialysis patients in a cross-sectional study to explore the impact of possible factors on daytime sleepiness using both the PSQI and ESS questionnaires of excessive daytime sleepiness (EDS). In univariate analysis, ESS correlated with serum Mg (r = 0.376, P = 0.008). Multivariable linear regression indicated that urinary Mg was an independent predictor of ESS score [21].
In the last cross-sectional study on 3304 female Japanese dietetics students aged 18–20 years, the midpoint of sleep (calculated as halfway point between self-reported bedtimes and rise times) was negatively associated with dietary intake of Mg (P = 0.001) [22].
The only cohort study was conducted on 1487 adults aged ≥ 20 years to assess the association between Mg intake and self-reported sleep disorder symptoms. Compared with the lowest quartile of Mg intake, the highest quartile was associated with a decreased likelihood of falling asleep (odds ratio (OR): 0.12; 95% CI: 0.02, 0.57)) in women but not in men. No associations were found between dietary Mg intake and daytime sleepiness nor night snoring in either gender [13].
RCTs
The effect of Mg supplementation on sleep in healthy subjects was surveyed through 5 RCTs.
A study on the association between oral Mg supplementation and sleep EEG was conducted on 12 elderly subjects aged 60–80 years in Germany. Subjects received Mg in a dose of 10 mmol and 20 mmol each for 3 days followed by 30 mmol for 14 days. Authors revealed that the amount of wakefulness was reduced insignificantly; rapid eye movement (REM) sleep was not altered, while slow-wave sleep (SWS) was significantly increased (P ≤ 0.05) with major changes in the initial part of the night [23].
Another experiment was performed on 100 adults aged 51 to 85 years who had poor sleep quality based on a PSQI score > 5. Results showed that supplementation of 320 mg magnesium per day vs a placebo did not significantly affect sleep quality [24].
A crossover randomized double-blind placebo-controlled trial including 42 subjects with a mean age of 61.6 years showed no beneficial effects for an oral dose of 900 mg magnesium citrate twice daily for 1 month in relieving sleep disorders caused by nocturnal leg cramps [15].
Another study by Abbasi et al. was conducted to determine the effects of Mg supplementation on insomnia among the elderly population. This double-blind placebo-controlled study was carried out on 46 subjects (65 ± 4.6 years old) who were randomly allocated to the intervention (500 mg daily) or placebo group for 8 weeks. Mg supplementation increased sleep time, sleep efficiency, the concentration of serum renin, and melatonin. It also led to a decreased ISI score, sleep onset latency, and serum cortisol concentration [25].
Finally, a group of authors examined older adult subjects (age 50–70) treated with a synapse density enhancer containing Mg (n = 23) or placebo (n = 21) for 12 weeks. The treatment did not show any effect on sleep quality assessed by PSQI [16].
Discussion
The present study involved 7,582 subjects from 9 published cross-sectional, cohort, and RCT systematically reviewed the possible links between Mg and sleep quality (daytime falling asleep, sleepiness, snoring, and sleep duration) in an adult population. Observational studies suggested an association between higher Mg intake and sleep quality in healthy adults. The RCTs reported a positive effect of magnesium supplementation on PSQI score, sleep efficiency, and sleep time.
Good sleep quality plays a vital role in physical and mental health and well-being outcomes [1, 26]. Reduction in sleep quality and quantity may contribute to the accumulation of fatigue, drowsiness, and changes in the autonomic nervous system function as well as elevation of inflammatory markers and immune system dysfunction [27, 28] General population experiences impaired sleep, including insufficient sleep duration, long sleep-onset latency, frequent and long nocturnal awakenings, and other sleep disturbances. Mg seems to play an essential role in the regulation of sleep. However, little is known about the association between Mg and sleep disorder symptoms.
Results of the observational studies (one cohort and three cross-sectional studies) suggested an association between higher Mg intake and sleep quality (daytime falling asleep, sleepiness, snoring, and sleep duration). The cohort study showed that dietary Mg intake (332.5 mg/day) had no association with sleep disorder symptoms, including sleepiness and night snoring in in either men or women. While in women, an association between Mg intake and decreased likelihood of daytime falling asleep was found. Therefore, dietary magnesium intake may have long-term benefits in reducing daytime sleepiness in women.
Although these observational studies were of high quality, investigating individuals in different age groups and health conditions as well as lack of similar studies provided no possibility for comparing the results of other studies.
Held et al. revealed that oral Mg supplementation can lead to statistically insignificant maintenance of wakefulness and significantly greater increases in SWS in the 12 elderly subjects [23]. The study was conducted on a small population with limited generalizability. A study of fewer than 20 subjects is unlikely to be sufficient for investigation. In addition, aging is associated with an increased number of awakenings and reduced nocturnal sleep duration and decreased slow-wave sleep (34, 35). Therefore, the results of this study cannot be applied to an adult population.
A study performed by Nielsen et al. [8] investigated the role of magnesium supplementation in adults with sleep complaints. After 7 weeks 320 mg/day magnesium citrate supplementation, the patients had increased overall PSQI score.
A high-quality study by Abbasi et al. showed that Mg supplementation (500 mg Mg per day) in 46 elderly populations for 8 weeks increased sleep time and sleep efficiency and decreased ISI score and sleep onset latency [25]. The results demonstrate that magnesium supplementation with 320–900 mg of magnesium daily brings significant improvement sleep efficiency, sleep time, and sleep onset latency and early morning awakening, especially in older adults.
A few animal and human researches have focused on Mg and sleep deprivation [13,14,15,16, 24, 26]. It has been suggested that Mg deficiency may exert a potential effect on chronic inflammatory stress and poor sleep by increasing periods of wakefulness and reducing slow-wave sleep. Although the effect of Mg on brain functions and sleep patterns is not fully understood, however, several mechanisms could explain the connections between sleep behavior and dietary magnesium. Evidence suggested that magnesium regulates sleep because it acts as an N-Methyl-D-aspartate antagonist (NMDA) and a γ-aminobutyric acid (GABA) agonist, sleep architecture, especially slow-wave sleep, is closely associated with the glutamatergic and GABAergic system [23, 26]. In addition, magnesium has an essential role in ion channels conductivity including an endogenous inhibitor of NMDA receptor and the unilateral entrance of potassium channels, thus helping to improve sleep. It has also been suggested that Mg has a relaxant action and anti-depressant effect and may increase melatonin and renin while reducing cortisol levels [23, 26]. Recent researches have revealed that Mg regulates cellular timekeeping, energy balance, and circadian rhythm; therefore, it seems to play a critical role in sleep regulation.
The available studies do not provide adequate and sufficient information for a certain association between dietary magnesium intake and sleep parameters. These findings not give a straight answer to a day dosing of magnesium to improving sleep quality. It seems that Mg supplementation ≥ 500 mg per day for more than 8 weeks could improve sleep time and sleep efficiency. Due to the insufficient evidence, or poor quality of evidence as well as the differences over geographical areas and individuals’ genetic background, further rigorous research is needed.
The critical strength of the current study is that it is the first systematic review conducted on this topic that covered all clinical trials and observational studies. The major limitation of this review is due to the overall low quality of some evidence and high risk of bias in included studies as well as the evidence of heterogeneity across the studies, especially in terms of their design and methodologies. Controlling possible confounding factors including age, gender, health status, Mg intake, BMI, waist-hip ratio, and physical activity level could enhance the quality of works.
Conclusion
This systematic review revealed an association between magnesium status and sleep quality. Observational studies suggested an association between higher Mg intake and daytime falling asleep, sleepiness, snoring, and sleep duration in healthy adults. The RCTs reported a positive effect of magnesium supplementation on PSQI score, sleep efficiency, and sleep time. The association between dietary magnesium and sleep patterns needs well-designed randomized clinical trials with a larger sample size and longer follow-up time (more than 12 weeks) to further clarify the relationship.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Simonelli G, Marshall NS, Grillakis A, Miller CB, Hoyos CM, Glozier N (2018) Sleep health epidemiology in low and middle-income countries: a systematic review and meta-analysis of the prevalence of poor sleep quality and sleep duration. Sleep Health 4(3):239–250
Buxton OM, Marcelli E (2010) Short and long sleep are positively associated with obesity, diabetes, hypertension, and cardiovascular disease among adults in the United States. Soc Sci Med 71(5):1027–1036
Cappuccio FP, D’Elia L, Strazzullo P, Miller MA (2010) Sleep duration and all-cause mortality: a systematic review and meta-analysis of prospective studies. Sleep 33(5):585–592
Gallicchio L, Kalesan B (2009) Sleep duration and mortality: a systematic review and meta-analysis. J Sleep Res 18(2):148–158
Åkerstedt T, Fredlund P, Gillberg M, Jansson B (2002) A prospective study of fatal occupational accidents–relationship to sleeping difficulties and occupational factors. J Sleep Res 11(1):69–71
Pandi-Perumal SR, Verster JC, Kayumov L, Lowe AD, Santana MGd, Pires MLN et al (2006) Sleep disorders, sleepiness and traffic safety: a public health menace. Braz J Med Biol Res 39(7):863–71
Afaghi A, O’Connor H, Chow CM (2007) High-glycemic-index carbohydrate meals shorten sleep onset. Am J Clin Nutr 85(2):426–430
Cao Y, Wittert G, Taylor AW, Adams R, Shi Z (2016) Associations between macronutrient intake and obstructive sleep apnoea as well as self-reported sleep symptoms: results from a cohort of community dwelling Australian men. Nutrients 8(4):207
St-Onge M-P, Roberts A, Shechter A, Choudhury AR (2016) Fiber and saturated fat are associated with sleep arousals and slow wave sleep. J Clin Sleep Med 12(1):19–24
Ji X, Grandner MA, Liu J (2017) The relationship between micronutrient status and sleep patterns: a systematic review. Public Health Nutr 20(4):687–701
Wacker WE, Parisi AF (1968) Magnesium metabolism. N Engl J Med 278(13):712–717
Volpe SL (2013) Magnesium in disease prevention and overall health. Adv Nutr 4(3):378S-S383
Cao Y, Zhen S, Taylor AW, Appleton S, Atlantis E, Shi Z (2018) Magnesium intake and sleep disorder symptoms: findings from the Jiangsu Nutrition Study of Chinese adults at five-year follow-up. Nutrients 10(10):1354
Cakir B, Kılınç FN, Uyar GÖ, Özenir Ç, Ekici EM, Karaismailoğlu E (2020) The relationship between sleep duration, sleep quality and dietary intake in adults. Sleep Biol Rhythms 18(1):49–57
Frusso R, Zarate M, Augustovski F, Rubinstein A (1999) Magnesium for the treatment of nocturnal leg cramps: a crossover randomized trial. J Fam Pract 48:868–871
Liu G, Weinger JG, Lu Z-L, Xue F, Sadeghpour S (2016) Efficacy and safety of MMFS-01, a synapse density enhancer, for treating cognitive impairment in older adults: a randomized, double-blind, placebo-controlled trial. J Alzheimers Dis 49(4):971–990
Moher D, Liberati A, Tetzlaff J, Altman DG (2010) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg 8(5):336–341
Peterson J, Welch V, Losos M, Tugwell PJ (2011) The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa: Ottawa Hospital Research Institute. 2(1):1–2
Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD et al (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928
Uhland AM, Kwiecinski GG, DeLuca HF (1992) Normalization of serum calcium restores fertility in vitamin D-deficient male rats. J Nutr 122(6):1338–1344
Lai X, Chen W, Bian X, Wang T, Li J, Wang H et al (2015) Predictors of poor sleep quality and excessive daytime sleepiness in peritoneal dialysis patients. Ren Fail 37(1):61–65
Sato-Mito N, Sasaki S, Murakami K, Okubo H, Takahashi Y, Shibata S et al (2011) The midpoint of sleep is associated with dietary intake and dietary behavior among young Japanese women. Sleep Med 12(3):289–294
Held K, Antonijevic I, Künzel H, Uhr M, Wetter T, Golly I et al (2002) Oral Mg2+ supplementation reverses age-related neuroendocrine and sleep EEG changes in humans. Pharmacopsychiatry 35(04):135–143
Nielsen FH, Johnson LK, Zeng H (2010) Magnesium supplementation improves indicators of low magnesium status and inflammatory stress in adults older than 51 years with poor quality sleep. Magnes Res 23:158–168
Abbasi B, Kimiagar M, Sadeghniiat K, Shirazi MM, Hedayati M, Rashidkhani B (2012) The effect of magnesium supplementation on primary insomnia in elderly: a double-blind placebo-controlled clinical trial. J Res Med Sci 17(12):1161
Peuhkuri K, Sihvola N, Korpela R (2012) Diet promotes sleep duration and quality. Nutr Res 32(5):309–319
Simpson N, Gibbs E, Matheson G (2017) Optimizing sleep to maximize performance: implications and recommendations for elite athletes. Scand J Med Sci Sports 27(3):266–274
Fullagar HH, Duffield R, Skorski S, Coutts AJ, Julian R, Meyer T (2015) Sleep and recovery in team sport: current sleep-related issues facing professional team-sport athletes. Int J Sports Physiol Perform 10(8):950–957
Funding
This research was supported by Isfahan University of Medical Sciences.
Author information
Authors and Affiliations
Contributions
Conception and design: A.A and F.S
Acquisition of data: A.A, F.S, and N.R
Analysis and interpretation of data: A.A, F.S, and N.R
Drafting the manuscript: F.S, N.R, and A.A
Revising it for intellectual content: A.A, F.S, N.R, and R.A
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Arab, A., Rafie, N., Amani, R. et al. The Role of Magnesium in Sleep Health: a Systematic Review of Available Literature. Biol Trace Elem Res 201, 121–128 (2023). https://doi.org/10.1007/s12011-022-03162-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-022-03162-1