Abstract
This study assessed the impacts of dietary arsenic trioxide on the contents of 26 elements in the pectoral muscle of chicken. A total of 100 Hy-line laying cocks were randomly divided into two groups (n = 50), including an As-treated group (basic diet supplemented with arsenic trioxide at 30 mg/kg) and a control group (basal diet). The feeding experiment lasted for 90 days and the experimental animals were given free access to feed and drinking water. The elements lithium (Li), boron (B), natrum (Na), magnesium (Mg), aluminium (AI), silicium (Si), kalium (K), calcium (Ca), vanadium (V), chromium (Cr), manganese (Mn), ferrum (Fe), cobalt (Co.), nickel (Ni), copper (Cu), zinc (Zn), arsenic (As), selenium (Se), molybdenum (Mo), cadmium (Cd), stannum (Sn), stibium (Sb), barium (Ba), hydrargyrum (Hg), thallium (Tl) and plumbum (Pb) in the pectoral muscles were determined using inductively coupled plasma mass spectrometry (ICP-MS). The resulted data indicated that Li, Na, AI, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Sn, Ba, Tl and Pb were significantly increased (P < 0.05) in chicken exposed to As2O3 compared to control chicken, while Mg, Si, K, As and Cd decreased significantly (P < 0.05). These results suggest that ICP-MS determination of elements in chicken tissues enables a rapid analysis with good precision and accuracy. Supplementation of high levels of As affected levels of 20 elements (Li, Na, AI, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Sn, Ba, Tl, Pb, Mg, Si, K, As and Cd) in the pectoral muscles of chicken. Thus, it is needful to monitor the concentration of toxic metal (As) in chicken for human health.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Monitoring chronic exposure of animals and humans to toxic metals and assessing their effects is a global health concern. Arsenic (As) is a poisonous trace element that is found in soil, water, food, dust, and some chemical materials [1]. In general, inorganic As is the more toxic form than organic forms and is present in water, which is readily absorbed by the animal and human body [2]. Although As contamination in the environment has been reported worldwide [3, 4], however, drinking water contamination with As has been a major public health concern in south-east, south-west and north-east USA; Inner Mongolia (China); south-west Taiwan coastal regions; Sonora (Mexico); Pamplonian Plain (Argentina); West Bengal (India); Northern Chile; and Bangladesh [5]. Chronic As exposure has induced increased incidence of all cancers, cytogenetic damage, nephritis, nephrosis, increased mortality from hypertensive heart disease, late foetal mortality, neonatal mortality and postnatal mortality in human [6].
Essential trace and toxic elements have positive and negative effects on human health and the environment. Essential trace minerals are important for a wide variety of physiological processes in all animals. Several hundred enzymes require the presence of minerals for their activity [7]. An optimal level of mineral elements is important for the health of animals and humans. The ingestion of food is an obvious means of exposure to metals, not only because many metals are natural components of foodstuffs but also in some cases the contamination occurs due to environmental exposure and during processing. Toxic elements can be very harmful even at low concentration when ingested over a long time period. The essential metals can also produce toxic effects when the metal intake is excessively elevated [8–10]. Therefore, some studies are interested in the analysis of the trace metal contents of the environmental samples and especially foods [11–13].
Studies on metal exposure have been mainly focused on Pb, Hg and Cd in birds, and several reviews have summarised the concentrations found in biota and their main effects [14–18]. With intensive areas of As-contaminated groundwater in recent years, the toxic effects have become a great threat to the health of human and animals. Chicken is a major source of meat for human consumption [19–21]. Therefore, chicken is routinely selected as en experimental model for the prediction the sensitivity of other avian species to the impact of the environmental contaminants [2, 22]. The previous studies have reported the adverse effects of As on chicken [23, 24]. In the present study, we investigated the effects of an As-supplemented diet on the levels of 26 elements in the pectora muscle of chicken.
Materials and Methods
Animals and Experimental Design
All procedures used in the present study were approved by the Institutional Animal Care and Use Committee of Northeast Agricultural University. A total of 100 Hy-line laying cocks were randomly divided into two groups (n = 50), including an As-treated group and a control group. The control group was fed with the basal diet, and the As-treated group fed with the basal diet supplemented with arsenic trioxide (As2O3) at 30 mg/kg according to 1/20 of the median lethal dose (LD50) for cocks. The feeding experiment lasted for 90 days and the experimental animals were given free access to feed and water. On day 90, the pectoral muscle tissues were removed from individual chicks (n = 15) after killing with sodium pentobarbital. The tissues were rinsed with ice-cold 0.9 % NaCl solution, frozen immediately in liquid nitrogen and stored at −80 °C until required.
Element Analysis
The elements (Li, B, Na, Mg, AI, Si, K, Ca, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, As, Se, Mo, Cd, Sn, Sb, Ba, Hg, Tl and Pb) in the pectoral muscles were determined using inductively coupled plasma mass spectrometry (ICP-MS) (Thermo iCAPQ, American). The instrumental parameters of the equipment used are summarised in Table 1.
The element concentrations were determined in acid digest of the samples according to the method of Uluozlu et al. [11]. One gramme of each sample was digested with 5 mL HNO3 (65 %) and 2 mL H2O2 (30 %) in microwave digestion system and diluted to 10 mL with deionised water. A blank digest was carried out in the same way. All sample solutions were clear. Digestion conditions for microwave system were applied as 3 min for 1800 W at 100 °C, 10 min for 1800 W at 150 °C and 45 min for 1800 W at 180 °C. The digested samples were diluted with ultrapure water to a final volume of 50 mL for ICP-MS analysis.
Statistical Analysis
Statistical analysis of all data was performed using SPSS for Windows (version 13, SPSS Inc., Chicago, IL). The differences between the As-treated groups and the control group were assessed by using paired t test. The data were expressed as the mean ± standard deviation. Differences were considered to be significant at P < 0.05. In addition, principal component analysis (PCA) was used to define the most important parameters, which could be used as key factors for individual variations using Statistic 6.0.
Results and Discussion
Toxic metal As can naturally occur in the environment, due to weathering, biological activity, and volcanic activity. Anthropogenic inputs from agricultural and industrial practices can also increase the levels of As contamination in the environment. Animals are capable of accumulating non-essential elements such as As, Hg and Pb in their muscle tissues [1, 25–28]. Chicken is the most consumed meat in North America and Chinese. Concentrations of As in chicken range from microgram per kilogram to milligram per kilogram. ICP-MS is well established as a method for multi-elemental analysis and the determination of isotope ratios. Chevallier et al. determined concentrations of 31 elements in foodstuffs by ICP-MS [29]. Giannenas et al. reported the contents of the trace minerals Se, Zn, Mn, Co, Cu, Mo, V, Cr, Ni, Tl, As and Cd in yolk and albumen from hen eggs using ICP-MS [30]. This methodology allows simultaneous analysis of a wide range of mineral elements in the same sample and has been used in this study.
Standard curves of 26 elements are summarised in Table 2. Correlation coefficient of standard curves was found to be in the range of 0.9904–1.0000. The results indicate that ICP-MS has a good linear relationship within the scope of the work. The levels of 26 elements in the pectoral muscles of chicken exposed to As2O3 are listed in Table 3. The resulted data indicates that the levels of 20 elements were significantly changed (P < 0.05) in chicken exposed to As2O3 compared to control chicken, except for the B, Ca, Se, Mo, Sb and Hg levels. Thereinto, the levels of 15 elements (Li, Na, AI, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Sn, Ba, Tl and Pb) were significantly decreased (P < 0.05) in chicken exposed to As2O3 compared to control chicken; levels of five elements (Mg, Si, K, As and Cd) increased significantly (P < 0.05). A proper amount of mineral is required for the normal function of all biochemical processes in animal body. The obtained results indicate that the levels of 20 elements (Li, Na, AI, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Sn, Ba, Tl, Pb, Mg, Si, K, As and Cd) in the pectoral muscles of chicken were significantly influenced by As supplementation. Mondal et al. reported that major elements’ (Ca, P and Mg) balance was not affected by the dietary Cu-salt and soybean oil supplementation both at days 21 and 42 [12]. These inconsistent results might be due to different species and levels of supplementation element. Chronic exposure to high levels of the toxic metals can cause variety of adverse health effects, including skin and internal cancers and cardiovascular and neurological effects [6, 31]. Hence, it is critical to balance the animal’s requirement to maintain growth performance and the element levels in the diet.
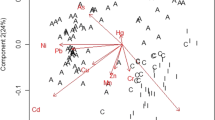
Using PCA, all biochemical parameters measured in the present study were distinguished on the ordination plots corresponded to the first and second principle components (Fig. 1). The first two principal components took into account 87.90 % (PC1 = 79.53 %, PC2 = 8.37 %). Furthermore, the observed relationships amongst the parameters were confirmed and quantified according to the Spearman’s test (Table 4).
Conclusion
In summary, ICP-MS determination of elements in chicken tissues enables a rapid analysis with good precision and accuracy. The obtained results indicate that supplementation of high levels of As affected levels of 20 elements (Li, Na, AI, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Sn, Ba, Tl, Pb, Mg, Si, K, As and Cd) in the pectoral muscles of chicken. Thus, results of the work suggest that it is needful to monitor the concentration of toxic metal (As) in chicken for human health.
References
Kazi TG, Shah AQ, Afridi HI, et al (2013) Hazardous impact of organic arsenical compounds in chicken feed on different tissues of broiler chicken and manure. Ecotoxicol Environ Saf 87:120–123
Shah AQ, Kazi TG, Arain MB, et al (2009) Comparison of electrothermal and hydride generation atomic absorption spectrometry for the determination of total arsenic in broiler chicken. Food Chem 113:1351–1355
Sohel N, Persson LA, Rahman M, et al (2009) Arsenic in drinking water and adult mortality a population-based cohort study in rural Bangladesh. Epidemiology 20:824–830
Li G, Sun GX, Williams PN, et al (2011) Inorganic arsenic in Chinese food and its cancer risk. Environ Int 37:1219–1225
Argos M, Kalra T, Rathouz PJ, et al (2010) Arsenic exposure from drinking water, and all-cause and chronic-disease mortalities in Bangladesh (HEALS): a prospective cohort study. Lancet 376:252–258
Rodrıguez VM, Jimenez-Capdeville ME, Giordano M (2003) The effects of arsenic exposure on the nervous system. Toxicol Lett 145:1–18
Yuan J, Xu Z, Huang C, et al (2011) Effect of dietary Mintrex-Zn/Mn on performance, gene expression of Zn transfer proteins, activities of Zn/Mn related enzymes and fecal mineral excretion in broiler chickens. Anim Feed Sci Technol 168:72–79
Celik U, Oehlenschlager J (2007) High contents of cadmium, lead, zinc and copper in popular fishery products sold in Turkish supermarkets. Food Control 18:258–261
Tuzen M (2003) Determination of heavy metals in fish samples of the middle Black Sea (Turkey) by graphite furnace atomic absorption spectrometry. Food Chem 80:119–123
Pouretedal HR, Rafat M (2007) Simultaneous determination of nickel(II) and cop-per(II) by second-derivative spectrophotometric method in micellar media. J Chin Chem Soc 54:157–164
Uluozlu OD, Tuzen M, Mendil D, et al (2009) Assessment of trace element contents of chicken products from Turkey. J Hazard Mater 163:982–987
Mondal MK, Das TK, Biswas P, et al (2007) Influence of dietary inorganic and organic copper salt and level of soybean oil on plasma lipids, metabolites and mineral balance of broiler chickens. Anim Feed Sci Technol 139:212–233
Lin Y, Munroe P, Joseph S, et al (2013) Chemical and structural analysis of enhanced biochars: thermally treated mixtures of biochar, chicken litter, clay and minerals. Chemosphere 91:35–40
Burger J, Gochfeld M (1997) Risk, mercury levels, and birds: relating adverse laboratory effects to field biomonitoring. Environ Res 75:160–172
Burger J, Gochfeld M (2000) Effects of lead on birds (Laridae): a review of laboratory and field studies. J Toxicol Environ Health B Crit Rev 3:59–78
García-Fernández AJ, Calvo JF, Martínez-López E, et al (2008) Raptor ecotoxicology in Spain: a review on persistent environmental contaminants. Ambio 37:432–439
García-Fernández AJ (2014) Avian ecotoxicology. In: Wexler P (ed) Encyclopedia of toxicology 2, 3rd edn. Elsevier Inc., Academic Press, New York, pp. 289–294
Scheuhammer AM (1987) The chronic toxicity of aluminium, cadmium, mercury, and lead in birds: a review. Environ Pollut 46:263–295
Yao H, Wu Q, Zhang Z, Li S, Wang X, et al (2013) Selenoprotein W serves as an antioxidant in chicken myoblasts. Biochim Biophys Acta 1830:3112–3120
Yao H, Wu Q, Zhang Z, Li S, Wang X, et al (2013) Gene expression of endoplasmic reticulum resident selenoproteins correlates with apoptosis in various muscles of Se-deficient chicks 1–3. J Nutr 143:613–619
Yao H, Liu W, Zhao W, et al (2014) Different responses of selenoproteins to the altered expression of selenoprotein W in chicken myoblasts. RSC Adv 4:64032–64042
Nachman KE, Francesconi GKA, Navas-Acien A, Love DC (2012) Raber arsenic species in poultry feather meal. Sci Total Environ 417–418:183–188
Sánchez-Virosta P, García-Fernández S, Espín AJ, et al (2015) A review on exposure and effects of arsenic in passerine birds. Sci Total Environ 512–513:506–525
Naraharisetti SB, Aggarwal M, Ranganathan V, Sarkar SN, Kataria M, Malik JK (2009) Effects of simultaneous repeated exposure at high levels of arsenic and malathion on hepatic drug-biotransforming enzymes in broiler chickens. Environ Toxicol Pharmacol 28:213–218
Ooi MS, Townsend KA, Bennett MB, et al (2015) Levels of arsenic, cadmium, lead and mercury in the branchial plate and muscle tissue of mobulid rays. Mar Pollut Bull 94:251–259
Ambrosio F, Brown E, Stolz D, et al (2014) Arsenic induces sustained impairment of skeletal muscle and muscle progenitor cell ultrastructure and bioenergetics. Free Radic Biol Med 74:64–73
D’Amico AR, Gibson AW, Bain LJ (2014) Embryonic arsenic exposure reduces the number of muscle fibers in killifish (Fundulus heteroclitus). Aquat Toxicol 146:196–204
Palaniappan PLRM, Vijayasundaram V (2008) Fourier transform infrared study of protein secondary structural changes in the muscle of Labeo rohitadue to arsenic intoxication. Food Chem Toxicol 46:3534–3539
Chevallier E, Chekri R, Zinck J, et al (2015) Simultaneous determination of 31 elements in foodstuffs by ICP-MS after closed-vessel microwave digestion: method validation based on the accuracy profile. J Food Compos Anal 41:35–41
Giannenas I, Nisianakis P, Gavriil A, et al (2009) Trace mineral content of conventional, organic and courtyard eggs analysed by inductively coupled plasma mass spectrometry (ICP-MS). Food Chem 114:706–711
Li JL, Jiang CY, Li S, et al (2013) Cadmium induced hepatotoxicity in chickens (Gallus domesticus) and ameliorative effect by selenium. Ecotoxicol Environ Saf 96:103–109
Acknowledgments
The Postdoctoral Scientific Research Developmental Foundation of Heilongjiang Province (Grant No. LBH-Q13012) supported the study. The authors thank the members of the Veterinary Internal Medicine Laboratory in the College of Veterinary Medicine, Northeast Agricultural University, for their help in collecting tissue samples.
Conflict of Interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, B., Xing, M. Evaluated the Twenty-Six Elements in the Pectoral Muscle of As-Treated Chicken by Inductively Coupled Plasma Mass Spectrometry. Biol Trace Elem Res 169, 359–364 (2016). https://doi.org/10.1007/s12011-015-0418-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-015-0418-0