Abstract
The heavy metal contamination of soils is a serious environmental issue because excessive metal concentrations pose risks to the health of humans, animals, and plants. For this reason, the interest in understanding the toxic effects of metals on crop growth and physiology has increased in the last decades. A pot trial was performed in a greenhouse to evaluate the effects of contaminated soil with different concentrations of cadmium (Cd) or chromium (Cr) on barley growth and development. Two cultivars of barley were studied, Pedrezuela and CB502. Growth, chlorophyll content, chlorophyll fluorescence, and relative water content (RWC) were analyzed during the plant-growing period. After harvesting, the Cd and Cr contents in plant were analyzed. No significant differences were observed for chlorophyll content and chlorophyll fluorescence between control plants and those treated with Cd. In the case of Cr, a significant decrease of plant growth, chlorophyll content, chlorophyll fluorescence, and RWC was detected with respect to the control. The tolerance index (TI) and translocation factor (TF) were calculated. Data indicated that both varieties are tolerant to these metals; CB502 showed higher tolerance to Cr and Pedrezuela to Cd. The effect of Cd or Cr addition on nutrient concentrations in plants varied among elements and organs of the plant analyzed. The correlations between the physiological and agronomic studied traits were significant (p < 0.01, p < 0.001), so the stress induced by these metals affected the physiology and water relations of the plant, which provoked a decrease of plant biomass, especially in the plants treated with Cr.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Soil metal contamination has increased in the last decades due to the impact of anthropogenic activities, leading to the increase in research related to phytotoxicity of contaminants and the mechanisms used by plants to balance their harmful effects. Soil contamination by metals affects the growth of plants and has a negative impact on soil microorganisms [1]. This decrease in growth in higher plants is due to metals which affect different physiological functions such as water relations, respiration, photosynthesis, and mineral nutrient inputs, particularly when they are in high concentrations. For this reason, there has been increased interest in determining the toxic effects of metals on the metabolism and development of crop plants, with the objective of understanding how the physiology of plants is affected when they accumulate toxic levels of metals in their biomass and what physiological mechanisms of tolerance actively work to minimize the concentration of metals in the aerial part of the plant [2].
The phytoextraction of metals is a promising technique, applicable to moderately contaminated soils and an alternative to ex situ decontamination techniques which are very expensive and unacceptable from an ecological point of view [3, 4]. The ability of land plants to absorb contaminants in the rhizosphere and transfer them to the aerial part has promoted the research about the use of higher plants to remediate soils contaminated with metals. Using crop plants for phytoremediation takes advantage of their ability to produce large amounts of biomass, rapid growth, and adaptability to different environmental conditions [5]. However, to be effective the plants must be tolerant to pollutants and accumulate significant amounts of toxic elements in their tissues.
Advances in plant breeding using genetic engineering techniques such as modifying the characteristics of metal absorption capacity, transportation, and storage as well as tolerance for various metals open new possibilities for phytoremediation of contaminated soils [6–10]. In this regard, cereal crops are attracting increasing interest for their processes of metal phytoextraction [11–14]. The presence of variability in Cd tolerance has been demonstrated in crops such as barley [15–18], rice [19], wheat [20], and soybean [21]. The presence of genotypes with different capabilities of tolerance to induced toxicity by Cr has also been observed in barley [22] and rice [23].
The Cd and Cr stress may lead to changes in plant nutrient concentration and composition depending on species, varieties, growth stages, and plant organ. The interactions of Cd or Cr and nutrients have been reported in some crops, such as wheat [20], rice [24–26], sunflower [27], and pea [28]. There are significant differences between species and varieties, and contradictory results have been observed.
In recent years, the response of plants to metal contamination has been mainly studied in hydroponic experiments. However, few studies have been conducted in soil to complete the crop cycle. The objectives of this study were (1) to study the response to various physiological traits of two barley cultivars grown in soil treated with increasing concentrations of Cd or Cr during the plant growth, (2) to analyze genotypic differences in biomass production, and (3) to study the interaction of these metals with mineral nutrients.
Materials and Methods
Plant Material and Experimental Design
Two cultivars of barley (Hordeum vulgare L.) were used: one two-row cultivar, Pedrezuela, and one six-row cultivar, CB502. They were planted in a loam soil from Central Spain in greenhouse conditions, using eight pots of 4 L each per treatment and cultivar, with two barley seeds in each pot. The soil initial characteristics were as follows: pH 8.1, electrical conductivity 0.26 dS/m, organic matter 1.08 %, carbonates 8.6 %, nitrogen 0.08 %, phosphorus 23 mg/kg, calcium 2,946 mg/kg, magnesium 446 mg/kg, sodium 98 mg/kg, potassium 408 mg/kg, chromium 15 mg/kg, Cd not detected. The pots were watered with tap water until the plants reached stage 20 of the Zadoks scale [29]. From this stage until the end of the crop cycle, the control pots were irrigated with 500 mL of tap water. The pots with metal treatments were irrigated with 400 mL of tap water + 100 mL of the solution of the corresponding metal, prepared from CdSO4, 30, 40, 50, and 60 mM for Cd treatments and from K2Cr2O7, 0.2, 0.5, 1, and 1.5 mM for Cr(VI) treatments. All the results from agronomic and physiological characteristics obtained from plant material represent the mean of the four replicates. The results from nutrients and metal content represent the mean of the three replicates.
Chlorophyll Content
The estimation of the chlorophyll content was carried out using intact leaves by single-photon avalanche diode (SPAD), SPAD-502 chlorophyll meter (Minolta Co, Japan) [30]. The measurements were taken at the stages of the grain-filling period (78 on the Zadoks scale) in four pots. The measurements were made on the flag leaf of the main shoot of each plant. There were four measurements per plant, and the average value of each leaf was recorded. Throughout the experiment, the measurements were made on the same leaf.
Chlorophyll Fluorescence Measurements
The maximum photosynthetic efficiency of PSII (Fv/Fm), where Fv is the variable fluorescence and Fm the maximum fluorescence [30], was measured in the same state and the same leaf that the SPAD measured using a fluorometer F MS2 from Hansatech Instruments Ltd., UK. The fluorescence parameters were measured in the central part of the flag leaf of the main stem of each plant, after its adaptation to the dark for 30 min.
Relative Water Content and Osmotic Potential
To determine the relative water content (RWC), the flag leaves were cut from the main shoot from the three pots. The samples were at stage 78 on the Zadoks scale. In the laboratory, the leaves were cut longitudinally into two symmetrical portions. A portion was used to determine the relative water content immediately, according to the formula [31]:
The other part of the sheet was frozen and used to measure osmotic potential (OP) after thawing, using a thermocouple hygrometer (Wescor model C-52, Logan, UT, USA) set in a dew point mode [32].
Plant Growth, Tolerance Index, and Translocation Factor
The plant height was measured at the end of grain-filling period corresponding to stage 80 of the Zadoks scale. At the end of the crop cycle, the spikes of each plant were cut and weighed. Then the plants in each of the pots were cut to soil level to obtain the dry weight of the shoots. The roots of each plant, once washed, were dried in an oven at 80 ° C for 48 h to obtain the dry weight.
Two indicators were calculated to evaluate the availability of plants for phytoextraction purposes: tolerance index (TI) and translocation factor (TF).
A metal tolerance index of 50 % of the optimum growth is the desired minimum biomass production for plants growing in soils contaminated with metals [33].
To obtain the TI to the metals found in the plant biomass, the following formula was used: TI = [(biomass treatment) / (biomass control)] × 100.
The TF is used to evaluate the plant’s ability to translocate metals from roots to the harvestable aerial part [34].
The TF was calculated using the following formula: TF = (C shoots / C roots) where C shoots is the concentration of metal in the shoots and C roots is the concentration of metals in the roots.
Analysis of Nutrients and Metals in Plants
The vegetable dry samples (shoots and roots) were mill grounded and treated by a dry digestion in a muffle furnace (480 °C). The ashes were digested using HCl according to [35]. After cooling, the digests were filtered (Whatman filter paper no. 541) and diluted with MilliQ water to 50 mL. The total concentration of nutrients (Ca, Mg, Na, K, Fe) and metals (Cd, Cr) was measured by atomic absorption (Varian AA 240 FS, Varian, Palo Alto, CA, USA).
Statistical Analysis
The differences among control and treated soils with different concentrations of Cd or Cr were evaluated by one-way analysis of variance (ANOVA) at significance level of p < 0.05 using SAS. The mean values of the replicates were compared by Duncan’s test to detect significant differences.
Results
Chlorophyll Content and Chlorophyll Fluorescence
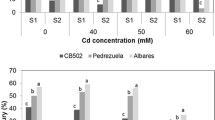
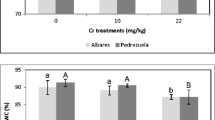
In both tested cultivars, the fluorescence and SPAD values were higher in plants treated with Cd than in plants treated with Cr. Increasing the dose of Cd did not adversely affect either the photosynthetic efficiency or the chlorophyll content. The Fv/Fm and SPAD values of various treatments were similar to the control in the first four samples taken (data not shown). In the last sampling, CB502 fluorescence and chlorophyll content showed a slight decrease which is not significantly relative to the control (Fig. 1a, b). In the Pedrezuela cultivar, these values were more affected by the metal dose, showing lower values than the control from the dose of 40 mM (Fig. 1a). These decreases that are relative to control represent 7 % for Fv/Fm and 17 % for SPAD values. For SPAD values, the differences between treatments were not statistically significant (Fig. 1b).
Effect of different treatments of cadmium or chromium on chlorophyll fluorescence (a), chlorophyll content (b), and relative water content (c) of two barley cultivars sampling at the end of grain filling period. For each cultivar, treatments followed by the same letter do not differ significantly (p < 0.05; Duncan test), lowercase letters CB502, and capital letters Pedrezuela. The data exposed are means of four replicates
In the Cr treatments, the effect of metal on Fv/Fm and the SPAD values was more important than in Cd treatments. The differences between treatments were significant from 0.5 mM treatment. The Fv/Fm and SPAD values for the cultivar CB502 showed a significant decrease relative to the control (18 and 45 % respectively) (Fig. 1a, b). In the cultivar Pedrezuela, the decrease was greater, 29 % for Fv/Fm and 87 % for SPAD values (p < 0.001).
Relative Water Content
In general, the RWC of the two cultivars of barley was lower with increasing concentration of the metal and the time in which the plants were subjected to different treatments (data not shown). In the last sampling, the RWC of the cultivar CB502 did not significantly decrease compared to the control in Cd treatments, but in plants grown in soils treated with Cr, the RWC decrease was significant from the lowest doses of metal applied (0.2 mM) (Fig. 1c). At the highest dose, the values were 31 % lower than in the control. In Pedrezuela, the RWC values relative to the control were significantly lower in plants treated with Cd at higher doses (50 and 60 mM). In Cr treatments, the RWC decrease was significant from the treatment, 1 mM, and with a decline of 44 % at higher metal concentrations (Fig. 1c).
The RWC decrease relative to the control was lower in CB502 than Pedrezuela, indicating a better adaptation of the first variety to the stress produced by these two metals. The greater capacity for osmotic adjustment of CB502 has contributed to maintain higher RWC, 79.80 %, than the Pedrezuela, 69.18 %, to an OP of −25 bars (Table 1). This benchmark was used because it was within the limit of OP reached by the two varieties studied.
Plant Growth, Tolerance Index, and Translocation Factor
The plant height was greater in plants treated with Cd than in plants treated with Cr in the two cultivars studied. The CB502 cultivar height was greater than Pedrezuela (Table 2). The Cd treatments did not affect the plant height. In the Cr treatments, the height was lower compared to the control along with the increase of metal concentration showing a decrease of 38 % in CB502 and 30 % in Pedrezuela at the highest metal concentration.
The dry weight of shoots and roots was also higher in plants treated with Cd than those treated with Cr in the two cultivars. Considering the genotypes, the dry weights of Pedrezuela were higher than those of CB502 treated with Cd and those treated with the lowest concentration of Cr. At higher concentrations of Cr, the dry weights of CB502 were higher than those of Pedrezuela both in the shoots and in the roots (Table 2). A dose-dependent decrease of dry weight was detected, and the major decrease was observed at 60 mM of Cd. At this concentration, the decrease of the dry weights relative to the control in CB502 was 29 % for the shoots and 22 % for the roots and in Pedrezuela, 21 % and 50 %, respectively. At the highest Cr concentration, weight reduction in CB502 was 87 % for the shoots and 63 % for the roots, and in Pedrezuela it was 90 % for the shoots and 89 % for the roots.
The TI of the plants treated with Cd exceeded 50 % of the optimal growth in all treatments, being comprised between 90 and 74 % in the barley CB502 and between 97 and 76 % for Pedrezuela (Table 3). In the Cr treatments, the TI was lower for all materials studied, being between 85 and 14 % in CB502 and between 83 and 11 % in Pedrezuela. The Cd tolerance was very high in the two cultivars, with TI greater than 74 %, even in plants treated with the highest dose of the metal. In plants treated with Cr there were greater differences; the TI was higher than 50 % only for the plants grown in soil treated with the lowest dose of metal. The tolerance indices to Cd for Pedrezuela were higher than those of CB502; for Cr treatments, CB502 had higher tolerance indices.
The mean values of TF for Cd were higher in CB502 (0.21) than in Pedrezuela (0.16). The opposite effect was observed for Cr, and the higher value was observed in Pedrezuela (0.17) in comparison to CB 502 (0.07) (Table 3).
The correlations between the physiological and agronomic traits studied were high, significant at 1 % and 0.1 % in all cases (Table 4).
Cd and Cr Contents in Plant Tissues
The amount of Cd and Cr accumulated in various parts of the plant was lowest in the controls and increased progressively with increasing concentrations of the metal applied in all Cr treatments, in both shoot and roots (Table 5). An exception was observed in the behavior of plants in soil treated with Cd; plants grown with 40 mM accumulated more metal than plants grown with 50 and 60 mM treatments. The higher doses of Cd affected more the CB502 cultivar than the Pedrezuela, decreasing its biological activity and therefore reducing the absorption of nutrients and metals. In the two treatments and in both varieties studied, the amount of metal accumulated in the roots was greater than in the shoot, being these differences significant in all cases.
The TF calculated for CB502 and Pedrezuela is reported in Table 3. The values indicate that CB502 is the most efficient in the Cd translocation with a higher TF (0.32 and 0.24) in plants treated with 50 and 60 mM of Cd, while the opposite behavior is observed in Pedrezuela with the lowest TF for those treatments (0.15 and 0.11). This cultivar was more efficient in the translocation of Cr to the shoot. The TF value for the highest Cr concentrations was 0.21 and 0.22. These values were lower for the cultivar CB502 (0.10 and 0.08).
Nutrient Element Concentration
The applied amount of metal does not seem to influence the studied nutrients status (Tables 6 and 7). The CB502 plants grown in soils treated with the highest Cd concentrations accumulated the highest Ca concentration in their roots (7,798 mg/kg) in comparison to control plants (3,203 mg/kg) (Table 6). However, in the shoot, highest Ca concentrations were found in the control plants (6,540 mg/kg) and lowest (2,843 mg/kg) in plants treated with the highest concentrations of Cd. In Pedrezuela, the behavior was similar. Plants grown in soil with the highest concentrations of Cd accumulated in their roots the highest concentrations of Ca (21,135 mg/kg), while control plants accumulated the lowest amounts (3,154 mg/kg). In shoots, the Ca concentration was similar in control and treated plants. Other nutrients studied did not show a clear trend with respect to different Cd concentrations applied.
The treatments of Cr did not affect regularly the concentration of nutrients studied. In both cultivars, the amount of Ca accumulated in the root and the amount of Na accumulated in shoot and grain of control plants were lower than the amount accumulated in plants grown in soils with Cr treatments (Table 7).
Significant differences in the amount of nutrients in various organs of the plant have been observed. In our experimental conditions, the higher concentration of nutrients was accumulated in roots and shoots. The amount of Ca and Fe accumulated in the roots was higher than in the shoot and grain in both studied cultivars, and the same behavior was observed in both treatments Cd and Cr. However, the highest amount of Na and K was accumulated in the shoot. K was the most abundant nutrient in the grain, followed by Na, Ca, and Fe, respectively.
The correlation was significant and positive between Cd or Cr and Ca and Fe and negative and not significant with Na and K (Table 8).
Discussion
Cadmium treatments affected the chlorophyll content and Fv/Fm only in the highest metal concentrations in the longest treatment period. In the Cr treatments, both the chlorophyll content and photosynthetic efficiency decreased to low concentrations, showing the most important differences at highest metal concentration, with a decrease compared to the control of 45 % of chlorophyll content and 18 % in the maximum photochemical efficiency of PSII (Fv/Fm) in CB502 and 87 % for chlorophyll content and 29 % for photosynthetic efficiency in Pedrezuela (Fig. 1a, b). The reduction in chlorophyll content and photosynthetic efficiency of PSII at different concentrations of Cd or Cr has been observed by other authors [36, 37] and in other species [38–40]. The inhibition of photosynthesis is the result of damage to the PSII reaction centers in the leaf. Photosynthesis is the main source for biomass production. Our results indicate that the stress produced by the metal reduces the growth of plants due to the decrease in chlorophyll content and consequently the inhibition of photosynthesis. These results are consistent with those obtained for wheat [41], corn [42], and soybean [43]. The treatments with the metals used affected the variety Pedrezuela more than CB502 in both the growth parameters and the physiological characteristics studied. The variability of different barley genotypes regarding tolerance to Cd or Cr has been observed by other authors [15, 16, 22, 44, 45]. The existence of variability is important for achieving genotypes better adapted to soils polluted with metals and therefore more effective for use in decontamination processes.
The wilting of the plant due to metal toxicity has been observed in different crops [46], as well as the existence of osmotic adjustment in Lolium perenne plants treated with different concentrations of Cr [47]. The results obtained in this study corroborate the observation by these authors since the CB502 barley maintained a higher RWC in the leaves than the Pedrezuela during the stress increase produced by the metal (Fig. 1c) due to its higher osmotic adjustment capacity (Table 1). To this, the capacity of adaptation to the stress produced by metals may have contributed the fact that the metal was incorporated in successive irrigations during the crop cycle, allowing the adaptation of plants to stress produced by metals. Further studies are needed to evaluate the behavior of the plants when they are subjected to high concentrations of the metal since the beginning of their development.
Exposing barley plants to increasing concentrations of Cd or Cr produced a reduction in dry matter yield with respect to the control plants. These results are in agreement with those found by other authors in wheat [20] and barley [22, 36]. The reduction in root dry weight was higher than in the shoots, particularly in treatments of Cr, and higher in Pedrezuela, 89 % at the highest concentration, while it was 63 % in CB502 (Table 2). These results confirm those found in barley treated with Cd [16, 17]. The reason for the increased sensitivity of the root to the Cd and Cr concentrations could be that the root is the first organ in contact with the metals, thus accumulating more metal in the root than in the shoot [45, 48, 49]. The metals are immobilized in the vacuoles of the root cells, making them less toxic to the plant, then the root can be considered a barrier to the translocation of metals to the aerial part, reflecting the potential tolerance mechanism operating in the root cells [23, 38].
The stress caused by metals induced a reduction in growth parameters in other crops such as almonds [50], ryegrass [47], and peas [28]. Differences were also observed between genotypes in cereal crops that suffered stress from Cd or Cr [15, 23].
The TI was greater than 50 % for Cd treatments, in which the values were higher for Pedrezuela than for CB502 (Table 3). The TF value for CB502 was twice that of Pedrezuela TF at the highest Cd concentrations (Table 3). These values indicate that barley CB502 is the most efficient in the translocation of Cd to the shoot and less tolerant to this metal than Pedrezuela. The decrease of dry weight of Pedrezuela shoots was lower than in CB502 shoots. The amount of Cd accumulated in Pedrezuela was greater than the amount accumulated in CB502 because of its greater tolerance to Cd. For Cr treatments, CB502 presented higher tolerance indices. The TF value for Pedrezuela was twice than that of CB502 TF at the highest Cr concentrations. The first is the most efficient in Cr translocation to the shoot and less tolerant to this metal than CB502 which accumulated more metal in both roots and shoots. These high concentrations of Cd and Cr are those that caused increased stress in the cultivars studied.
The amount of Cd and Cr accumulated in the root was higher than that accumulated in the aboveground part in the two studied cultivars (Table 5). The highest concentration of Cd in roots has been showed in other crops like wheat [20], rice [24], and sunflower [27]. High retention of metals in roots and shoots is particularly desirable in cereal crops because these parts are not generally utilized as food or feed, reducing the risk of transfer to the food chain. The use of TI and TF to evaluate plants’ availability for phytoextraction purposes has been used by different authors [34, 51]. Under conditions of high stress caused by metals, cultivar CB502 showed higher values of TF when plants were grown in soil treated with Cd, and the biomass was lower than in Pedrezuela. The latter shows higher values of TF in Cr-treated plants, and the biomass obtained was lower than in CB502 (Tables 2 and 3).
In general, no differences that are associated with the applied metal concentrations in the behavior of nutrients in plants were observed, but the correlation between the two metals and Ca and Fe was positive and significant except for Cr-Ca in CB502 and Cr-Fe in Pedrezuela (Table 8). These results indicate that some synergistic relation existed in the absorption and translocation between Cd or Cr and Ca and Fe. Metal concentrations affected the translocation of Ca and Fe from the root to the shoot since the amount of the two metals accumulated in the root was greater than in the shoot. Liu et al. in 2003 showed that the interactions of Cd and Fe, Zn, and Cu are synergetic in uptake and translocation from root to shoot in rice. A similar interaction was observed between Cd contamination and Cu, Zn, and Fe contents in sunflower [27]. The inhibition of Ca and Fe uptake and translocation may play an important role in chlorophyll biosynthesis as they are cofactors of the polypeptide enzymes in PSI and PSII [39].
In summary, given the tolerance index and translocation factor, both varieties may be used in the remediation of soil contaminated with Cd at all assayed concentrations; however, the plants only showed a tolerance index higher than 50 % at the lowest concentration of Cr. It should be noted that Pedrezuela is more tolerant to Cd and CB502 more tolerant to Cr.
The high correlations found between the studied traits (Table 4) indicate that the stress due to Cd and Cr affected the physiology and water relations of the plant and resulted in a decrease in biomass, especially in plants grown in soils treated with Cr. CB502 barley is more tolerant to these metals, because of the water status, chlorophyll content, and photosynthetic efficiency of PSII remained higher during the crop cycle at the different metal concentrations assayed. The increasing concentrations of Cd and Cr had no significant effect on the concentration of nutrients in plants. A potential synergistic effect between metals and nutrients, Ca, and Fe in both varieties was observed.
References
McGrath SP, Zhao FJ, Lombi E (2001) Plant and rhizosphere processes involved in phytoremediation of metal-contaminated soils. Plant Soil 232:207–214
Krämer U (2010) Metal hyperaccumulation in plants. Annu Rev Plant Biol 61:517–534
McGrath SP, Zhao FJ, Lombi E (2002) Phytoremediation of metals, metalloids, and radionuclides. Adv Agron 75:1–56
Nascimento CWA, Xing B (2006) Phytoextraction: a review on enhanced metal availability and plant accumulation. Sci Agric 63:299–311
Soriano MA, Federes E (2003) Use of crops for in situ phytoremediation of polluted soils following a toxic flood from a mine spill. Plant Soil 256:253–264
Salt DE, Blaylock M, Kumar N, Dushenkov V, Ensley BD, Chet I, Raskin I (1995) Phytoremediation: a novel strategy for the removal of toxic metals from the environment using plants. Nat Biotechnol 13:468–474
Lasat MM, Pence NS, Garvin DF, Ebbs SD, Kochian LV (2000) Molecular physiology of zinc transport in the Zn hyperaccumulator Thlaspi caerulescens. J Exp Bot 51:71–79
Rascio N, Navari–Izzo F (2011) Heavy metal hyperaccumulating plants: how and why do they do it? And what makes them so interesting? Plant Sci 180:169–181
Hao D, Chao M, Yin Z, Yu D (2012) Genome-wide association analysis detecting significant single nucleotide polymorphisms for chlorophyll and chlorophyll fluorescence parameters in soybean (Glycine max) landraces. Euphytica 186:919–931
Kumar S, Sehgal SK, Kumar U, Prasad PV, Joshi AK, Gill BS (2012) Genomic characterization of drought tolerance-related traits in spring wheat. Euphytica 186:265–276
Clark JM, Norvell WA, Clark FR, Buckley WT (2002) Concentration of cadmium and other elements in the grain of near-isogenic durum lines. Can J Anim Sci 82:27–33
Vassilev A, Vangronsveld J, Yordanov I (2002) Cadmium phytoextraction: present state, biological backgrounds and research needs. Bulg J Plant Physiol 28:68–95
Ueno D, Koyama E, Yamaji N, Ma JF (2011) Physiological, genetic, and molecular characterization of a high-Cd-accumulating rice cultivar, Jarjan. J Exp Bot 62:2265–2272
Zhang X, Zhang G, Guo L, Wang H, Zeng D, Dong G, Qian O, Xue D (2011) Identification of quantitative trait loci for Cd and Zn concentrations of brown rice grown in Cd-polluted soils. Euphytica 180:173–179
Wu FB, Zhang G (2002) Genotypic differences in effect of Cd on growth and mineral concentrations in barley seedlings. Bull Environ Contam Toxicol 69:219–227
Tiryakioglu M, Eker S, Ozkutlu F, Husted S, Cakmak I (2006) Antioxidant defense system and cadmium uptake in barley genotypes differing in cadmium tolerance. J Trace Elem Med Biol 20:181–189
Wu F, Zhang G, Dominy P, Wu H, Bachir DML (2007) Differences in yield components and kernel Cd accumulation in response to Cd toxicity in four barley genotypes. Chemosphere 70:83–92
Chen F, Wang F, Zhang G, Wu F (2008) Identification of barley varieties tolerant to cadmium toxicity. Biol Trace Elem Res 121:171–179
Xue D, Chen M, Zhang G (2009) Mapping of QTLs associated with cadmium tolerance and accumulation during seedling stage in rice (Oryza sativa L.). Euphytica 165:587–596
Zhang GP, Fukami M, Sekimoto H (2002) Influence of cadmium on mineral concentrations and yield components in wheat genotypes differing in Cd tolerance at seedling stage. Field Crop Res 77:93–98
Sugiyama M, Ae N, Hajika M (2011) Developing of a simple method for screening soybean seedling cadmium accumulation to select soybean genotypes with low seed cadmium. Plant Soil 341:413–422
Ali S, Zeng F, Qiu L, Zhang G (2011) The effect of chromium and aluminum on growth, root morphology, photosynthetic parameters and transpiration of the two barley cultivars. Biol Plantarum 55:291–296
Qiu B, Zhou W, Xue D, Zeng F, Ali S, Zhang G (2010) Identification of Cr-tolerance lines in a rice (Oryza sativa L.) DH population. Euphytica 174:199–207
Liu J, Li K, Xu J, Liang J, Lu X, Yang J, Zhu Q (2003) Interaction of Cd and five mineral nutrients for uptake and accumulation in different rice cultivars and genotypes. Field Crop Res 83:271–281
Zeng F, Qiu B, Ali S, Zhang G (2010) Genotypic differences in nutrient uptake and accumulation in rice under chromium stress. J Plant Nutr 33:518–528
Hayat S, Khalique G, Irfan M, Wani AS, Tripathi BN, Ahmad A (2012) Physiological changes induced by chromium stress in plants: an overview. Protoplasma 249:599–611
Rivelli AR, Puschenreiter M, De Maria S (2014) Assessment of cadmium uptake and nutrient content sunflower plants grown under Cd stress. Plant Soil Environ 60:80–86
Sandalio LM, Dalurzo HC, Gómez M, Romero-Puertas MC, del Río LA (2001) Cadmium-induced changes in the growth and oxidative metabolism of pea plants. J Exp Bot 52:2115–2126
Zadoks JC, Chang TT, Konzak CF (1974) A decimal code for the growth stages of cereals. Weed Res 14:415–421
Fang S, SunW HH, Zhang H, Zheng K (2012) Chlorophyll content of barley (Hordeum vulgare L.) estimation from leaf SPAD, chlorophyll fluorescence and reflectance properties. Adv Sci Lett 11:702–705
Ritchie SW, Nguyen HT, Holaday AS (1990) Leaf water content and gas-exchange parameters of two wheat genotypes differing in drought resistance. Crop Sci 30:105–111
Morgan JM (1983) Osmoregulation as a selection criterion for drought tolerance in wheat. Aust J Agric Res 34:607–614
Shahandeh H, Hossner LR (2000) Plant screening for chromium phytoremediation. Int J Phytoremediat 2:3–51
Mattina MJ, Lannucci-Berger W, Musante C, White JC (2003) Concurrent plant uptake of heavy metals and persistent organic pollutants from soil. Environ Pollut 124:375–378
Garate A, Ramón AM, Carpena OR (1984) Influencia del boro sobre el manganeso y otros nutrientes en extractos de tejidos vasculares. Anales de Edafología y Agrobiología 43:1467–1477
Vassilev A, Lidon FC, Ramalho JC, do Céu Matos M, Bareiro MG (2004) Shoot cadmium accumulation and photosynthetic performance of barley plants exposed to high cadmium treatments. J Plant Nutr 27:775–795
Ali S, Farooq MA, Jahangir MM, Abbas F, Bharwana SA, Zhang GP (2013) Effect of chromium and nitrogen form on photosynthesis and anti-oxidative system in barley. Biol Plantarum 57:758–763
Bonnet M, Camares O, Veisseire P (2000) Effects of zinc and influence of Acremonium lolii on growth parameters, chlorophyll a fluorescence and antioxidant enzyme activities of ryegrass (Lolium perenne L. cv Apollo). J Exp Bot 51:945–953
Shukla UC, Singh J, Joshi PC, Kakkar P (2003) Effect of bioaccumulation of cadmium on biomass productivity, essential trace elements, chlorophyll biosynthesis, and macromolecules of wheat seedlings. Biol Trace Elem Res 29:257–273
Hörcsik ZT, Kovács L, Láposi R, Mészáros I, Lakatos G, Garab G (2007) Effect of chromium on photosystem 2 in the unicellular green alga, Chlorella pyrenoidosa. Photosynthetica 45:65–69
Ci D, Jiang D, Wollenweber B, Dai T, Jing Q, Cao W (2010) Cadmium stress in wheat seedlings: growth, cadmium accumulation and photosynthesis. Acta Physiol Plant 32:365–373
Wang H, Zhao SC, Liu RL, Zhou W, Jin JY (2009) Changes of photosynthetic activities of maize (Zea mays L.) seedlings in response to cadmium stress. Photosynthetica 47:277–283
Xue ZC, Gao HY, Zhang LT (2013) Effects of cadmium on growth, photosynthetic rate, and chlorophyll content in leaves of soybean seedlings. Biol Plantarum 57:587–590
Burzynsky M, Żureka A (2007) Effects of copper and cadmium on photosynthesis in cucumber cotyledons. Photosynthetica 45:239–244
González A, Lobo MC (2013) Growth of four varieties of barley (Hordeum vulgare L) in soils contaminated with heavy metals and their effects on some physiological traits. Am J Plant Sci 4:1799–1810
Poschenrieder C, Barceló J (2004) Water relations in heavy metal stressed plants. In: Prasad MNV (ed) Heavy metal stress in plants: from biomolecules to ecosystems. Springer, Berlin, pp 249–270
Vernay P, Gauthier–Moussard C, Hitmi A (2007) Interaction of bioaccumulation of heavy metal chromium with water relation, mineral nutrition and photosynthesis in developed leaves of Lolium perenne L. Chemosphere 68:1563–1575
Grant CA, Bailey LD (1998) Nitrogen, phosphorus and zinc management effects on grain yield and cadmium concentration in two cultivars of durum wheat. Can J Plant Sci 78:63–70
Nocito FF, Lancilli C, Dendena B, Lucchini G, Sacchi GA (2011) Cadmium retention in rice roots is influenced by cadmium availability, chelation and translocation. Plant Cell Environ 34:994–1008
Nada E, Ferjani BA, Ali R, Bechir BR, Imed M, Makki B (2007) Cadmium-induced growth inhibition and alteration of biochemical parameters in almond seedlings grown in solution culture. Acta Physiol Plant 29:57–62
Marchiol L, Assolari S, Sacco P, Zerbi G (2004) Phytoextraction of heavy metals by canola (Brassica napus) and radish (Raphanus sativus) grown on multicontaminated soil. Environ Pollut 132:21–27
Acknowledgments
This work has been supported by Project FP13-APL from IMIDRA and EIADES Program S2009/AMB-1478 from Consejería de Educación, Comunidad de Madrid. The authors gratefully acknowledge Mrs. Martin for reviewing the English language.
Conflict of Interest
The authors have declared no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
González, A., Gil-Díaz, M. & Lobo, M.C. Response of Two Barley Cultivars to Increasing Concentrations of Cadmium or Chromium in Soil During the Growing Period. Biol Trace Elem Res 163, 235–243 (2015). https://doi.org/10.1007/s12011-014-0182-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-014-0182-6