Abstract
Twenty male buffalo calves (15 months, 200.2 ± 9.75) were divided into four groups of five animals in each and fed diets without (T1) or supplemented with 0.3 ppm selenium (Se) + 40 ppm zinc (Zn) (T2), 0.3 ppm Se + 40 ppm Zn + 10 ppm copper (Cu) (T3), and 40 ppm Zn + 10 ppm Cu (T4) for 120 days, during which blood samples were collected on days 0, 40, 80, and 120. Concentrations of glucose, total protein, albumin, globulin, urea, uric acid, and creatinine were similar in all the four groups. The level of different serum enzymes viz. lactate dehydrogenase, alkaline phosphatase, glutamate pyruvate transaminase, and glutamate oxaloacetate transaminase, and hormones viz. T3, T4, testosterone and insulin were similar (P > 0.05) among the four groups but the ratio of T4/T3 was reduced (P < 0.05) in the groups (T2 and T3) where selenium was supplemented at 120th day of supplementation. It was deduced that supplementation of 0.3 ppm Se and/or 10.0 ppm of Cu with 40 ppm Zn had no effect on blood metabolic profile in buffalo calves, except the ratio of T4 and T3 hormone which indicates that selenium plays an important role in converting T4 hormone to T3 which is more active form of thyroid hormone.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Essentiality of selenium [1], copper [2], and zinc [3], for life is well established, as these elements are essentially required for a number of biochemical functions in living organisms. Selenium-dependent enzymes, notably, the glutathione peroxidase, are components of the physiological antioxidative protection system. Selenium (Se) has also been shown to mediate a number of insulin-like actions both in vivo as well as in vitro [4] and it is also a component of enzyme type I iodothyronine-5′-deiodinase, which converts total thyroxine (T4) in to total tri-iodothyronine (T3) which is more active form of the thyroid hormone[5]. Copper is also an essential component of many enzymes (cuproenzymes) and proteins. Studies have shown that copper is required for growth, host defense mechanisms, bone strength, red and white cell maturation, iron transport, and glucose metabolism [6]. Zinc is associated with more than 300 enzymes either as component or as activator [7–9]. In many of the enzymatic reactions, all three, i.e., selenium, copper, and zinc, are essential. For example, superoxide radicals are dismutated into hydrogen peroxide and oxygen by enzyme superoxide dismutase in the presence of copper and zinc as cofactors. Hydrogen peroxide is then reduced to water by the selenium–glutathione peroxidase couple. Efficient removal of these precursors of reactive oxygen species maintains the integrity of membranes, reduces the risk of cancer, and slows the aging process [10]. However, requirements of these trace elements for buffaloes are yet not specified and their interaction also affects their utilization and till the date levels recommended for cattle [11] are being used. Moreover, recent studies [12] have shown better performance of animals with the higher levels of these elements than their recommended levels by National Research Council. In view of these facts, the present study was conducted to assess the effect of supplementation of Se, copper (Cu), and zinc (Zn) on blood metabolic profile in male buffalo calves.
Materials and Methods
Animals: Selection and Grouping
Twenty healthy male buffalo (Bubalus bubalis) calves (15 months, 200.2 ± 9.75 kgs) were taken for this study, vaccinated against common contagious diseases, and dewormed 1 month before the start of experimental feeding. These calves were divided into four groups of five animals each on the basis of their body weight after randomized block design.
Housing and Management
The calves were housed in a well-ventilated, clean and concrete-floored shed, and fed individually. Strict management and hygienic practices were adopted throughout the experimental period. Clean drinking water was provided ad libitum twice a day at about 9 A.M. and 3 P.M.
Feeds and Feeding
Buffalo calves in all the four groups were offered concentrate mixture (20% ground maize grain, 27% soybean meal, 50% wheat bran, 2% mineral mixture, and 1% common salt) and ad libitum wheat straw to meet nutrient requirements for 500 g/day body weight gain [13]. The amount of concentrate mixture offered to calves was regularly revised every fortnight based on their body weight. All the calves were provided with about2.5 kg of available (maize/oats/berseem) green fodder, twice a week to meet vitamin A requirements. Feeding schedule was similar in all the four groups, except for mineral supplementation, which was no mineral supplementation in group T1 (control), 0.3 ppm Se + 40 ppm Zn in group T2, 0.3 ppm Se + 40 ppm Zn + 10 ppm Cu in group T3, and 40 ppm Zn + 10 ppm Cu in group T4. Supplementation of Se, Zn, and Cu was done as aqueous solutions of sodium selenite, zinc sulfate, and cupric sulfate, respectively, which were mixed daily in the concentrate mixture of each animal. The quantity of mineral solution was revised every week according to total dry matter intake of the animals. Experimental feeding was done for a period of 120 days.
Collection of Blood Samples and Separation of Serum
Blood samples from buffalo calves were collected initially (0 day) and subsequently at 40 days interval through jugular venipuncture, observing all aseptic precautions in the morning (before watering and feeding), into clean and dry test tubes and kept in slanting position for 45 min, followed by centrifugation at 700×g for 15 min to separate out serum. Serum samples were stored in 2 mL plastic vials at −20°C for further analysis.
Serum Biochemical Constituents and Enzyme Activity
Serum samples were analyzed for glucose [14], total protein (TP) and albumin [15], urea [16], creatinine [17], uric acid [18], alkaline phosphatase (ALP) [19], glutamate oxaloacetate transaminase (SGOT), and glutamate pyruvate transaminase (SGPT) [20], and lactate dehydrogenase (LDH) [21]. Concentration of globulin was calculated by subtracting serum albumin from TP.
Serum Hormone Levels
Serum hormone profile of calves was determined by radioimmunoassay (RIA) kits using a gamma counter (Model COBRA II, Packard, USA). The RIA kits used for the estimation of T3, T4 were from Immunotech, Radiova Czech Republic; testosterone from Immunotech, France; and insulin from Medicorp, Canada.
Statistical Analysis
All the data generated in the experiment were analyzed statistically using analysis of variance [22] and Duncan’s multiple range test was used to compare the means [23].
Results and Discussion
Chemical Composition of Feeds
The chemical composition of the concentrate mixture (CM), wheat straw (WS) and complete basal diet (calculated on the basis of proportion of CM and WS consumed during entire experimental period) is presented in Table 1. The contents of crude protein, ether extract, total carbohydrates, neutral detergent fiber, and acid detergent fiber contents were comparable to the levels recommended for growing buffalo calves for 500 g daily gain [13]. The Se (0.28 ppm), Zn (31.60 ppm), and Cu (11.47 ppm) contents in the diet were also comparable to the levels recommended for cattle calves [11].
Blood Biochemical Parameters
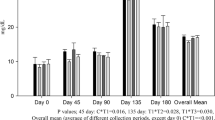
Results showed that supplementation of Se (0.3 ppm) and/or Cu (10 ppm) with Zn (40 ppm) had no effect on different blood biochemical parameters in the buffalo calves as their values were similar (P > 0.05) among the four groups throughout the experimental period (Table 2). In contrast to our observations, a declined plasma glucose level was reported in buffalo calves supplemented with Se [24]; but the level of Se supplementation in that experiment [24] was very high (8.54 ppm) compared to 0.3 ppm level used by us. However, similar to our findings, kids supplemented with 10 ppm Cu in their diet had no effect on blood glucose levels [25]. Similarly, no effect on plasma protein concentration has been reported with the supplementation of 0.1 ppm Se in steers [26], 8.54 ppm Se in buffalo calves [24], and 10 ppm of Cu in kids [25]. Similarly in one of our experiment [27], we also did not observed any effect on serum glucose and total protein level in buffalo calves supplemented with either Se (0.3 ppm) and Cu (10 ppm) alone on in combination.
However, the concentration of serum globulin was increased and albumin and A/G ratio were found to be reduced in our previous experiment when Se (0.3 ppm) either alone or with 10 ppm Cu was supplemented in the diet of buffalo calves [27]. But in most of the reports, supplementation of Se [24, 26] in buffalo calves and steers, respectively, or Cu [25] in kids had no effect on either serum albumin and globulin levels or their ratios.
Results also revealed no significant effect of supplementation of Se and/or Cu with Zn on serum urea, creatinine, and uric acid, as values of these constituents were comparable (P > 0.05) among the four groups throughout the experimental period (Table 2). In contrast to our results, steers supplemented with 0.1 ppm of Se [26] had higher levels of urea and creatinine in plasma; but the basal diet used in their experiment had very low levels of Se (0.015 ppm), which might be the reason for an increase in the levels of plasma urea and creatinine on 0.1 ppm of Se supplementation in that experiment. However, our findings are supported by an experiment conducted on buffalo calves where supplementation of as high as 8.54 ppm selenium had no effect on serum creatinine levels [24], similarly we also did not find any effect on either serum urea, creatinine, or uric acid levels when supplemented 0.3 ppm Se either alone or in combination with 10 ppm Cu in buffalo calves [27].
Serum Enzyme Profile
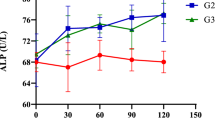
Supplementation of Se (0.3 ppm) and/or Cu (10 ppm) with Zn (40 ppm) had no effect on the activity of different blood enzymes, i.e., ALP, SGPT, SGOT, and LDH in buffalo calves, as their values were comparable (P > 0.05) in the four groups throughout the experimental period (Table 3). As these enzymes are the indicator of muscular or liver damages and the observations of the present experiment suggested that the level of mineral supplementations were within the safe limits in the diet of buffalo calves.
Consistent to our finding in Holstein Friesian calves [28], no effect was observed on ALP activity when supplemented Zn (30 or 60 ppm) with Cu (20 ppm) also. Similarly in ewes [29], no effect on ALP activity was observed due to supplementation of Zn with Cu (90 and 20 ppm, respectively) also. Similar observations were made in the study on buffalo calves [27] where no effect of Se (0.3 ppm) and/or Cu (10 ppm) supplementation on ALP activity was observed.
Contrary to our observations, reduced serum ALP activity was observed in steers supplemented with 0.1 ppm of Se [26], but it was probably due to a very low level of Se (0.015 ppm) in their basal diet. However, our basal diet had about 0.28 ppm Se; and probably therefore, we did not observe any change in the ALP activity due to further supplementation of Se. Our results are supported by an experiment in Holstein Friesian calves, where ALP activity remained unaltered on supplementation of Cu at 10 and 20 mg/kg of diet [28].
Contrary to our results, buffalo calves supplemented with 8.54 ppm of Se in their diet [24] and adult goats supplemented with 0.15–0.30 mg Se per kilogram body weight [29] showed increased SGOT and SGPT activity. But in both these experiments, the level of Se supplementation was too high compared to our experiment (0.3 ppm of the diet). Similarly, significant increase in SGOT activity has been reported in cows and heifers on Cu supplementation [30], but probably, it was also due to very high levels (80 ppm) of Cu supplementation, which might have been toxic to the animals. However, our results are supported by a study on male Holstein calves, where no effect was observed on SGOT and LDH activity on supplementation of 1 and 10 ppm of Se and Cu, respectively [31]. Similarly, supplementation of 10 ppm Cu in the diet had no effect on SGOT activity in male cattle calves [32]. The results of our previous study [27] also supports that supplementation of either 0.3 ppm Se or 10 ppm Cu either alone or in combination did not have any effect on the serum levels of enzyme SGOT, SGPT, and LDH.
Serum Hormone Profile
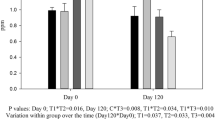
Supplementation of 0.3 ppm of Se and/or 10 ppm of Cu with 40 ppm Zn had no effect on insulin, testosterone, T3 and T4 levels as their values were comparable (P > 0.05) among the four groups throughout the experimental period (Table 4). However, there was an overall increasing tendency in the testosterone levels with increasing postfeeding period in all the groups, which may be associated with physiological changes that accompany male sexual maturation [33]. Similar to the present findings, Angus steers supplemented with 10 and 20 ppm Cu also did not show any effect on serum insulin values [34]. In buffalo calves [27], also no difference on the insulin level was observed when supplemented with 0.3 ppm Se with 10 ppm Cu.
Se supplementation has been reported to increase T3 and decrease T4 concentrations [26, 35]; but it was probably due to very low levels of Se being 0.015 [26] and 0.02–0.03 ppm [35] in the basal diets in these experiments. On the contrary, basal diet in our experiment had much higher levels of Se (0.28 ppm) and probably for that reason; we did not observe any significant effect of Se supplementation on T3 and T4 hormone levels as such in the buffalo calves. Similar to our observations, sheep (0.11 ppm Se in the basal diet) supplemented with 0.2 ppm of Se did not show any effect on thyroid (plasma T3 and T4 levels) metabolism [34]. In buffalo calves [27] also, no effect on thyroid metabolism was observed by supplementation of Se (0.3 ppm) with Cu (10 ppm). Goats [36] also did not show any effect on thyroid hormone levels when even a high level (1 ppm) of selenium was supplemented in their diet. Though, statistically there was no difference in values of T4 and T3 among different treatment groups, the ratio of T4/T3 was lower (P < 0.05) in groups supplemented with Se (T2 and T3), as compared to the groups where Se was not supplemented (T1 and T4). Thus, supplementation of Se had an effect on T4/T3 ratio in the buffalo calves. The results were also supported by the previous findings where in two different experiments conducted on Holstein Friesian calves [28] and reported decreased T4/T3 ratio (P < 0.01) by intraruminal Se pellet infusion. Similarly, in Frisian steers [26] also lower plasma T4/T3 ratio was observed by supplementation of 1 ppm Se.
As Se is a component of enzyme Type I Iodothyronine-5′-deiodinase, which converts T4 in to T3 [5] and a lower T4/T3 ratio is indicative of better thyroid status of the animals because T3 is more active form of the thyroid hormone as compared to T4. Thus the findings of present experiment suggested that supplementation of Se at 0.3 ppm along with either Zn or Zn + Cu had a beneficial effect on thyroid status of animals.
The serum testosterone concentration among different groups remained similar (P > 0.05) during the entire experimental period. Similar to our previous work in buffalo calves [27] supplementation of Se (0.3 ppm) and/or Cu (10 ppm) had no effect on serum testosterone levels.
On the basis of these results, it is deduced that supplementation of 0.3 ppm Se and/or 10 ppm of Cu with 40 ppm Zn in the basal diet of buffaloes having 0.28 ppm Se, 11.47 ppm Cu and 31.60 ppm Zn had no effect on their blood metabolic profile, except the ratio of T4 and T3 hormone which indicates that selenium plays an important role in converting T4 hormone to T3 which is more active form of thyroid hormone.
References
Patterson EL, Milstrey R, Stokstad ELR (1957) Effect of selenium in preventing exudative diathesis in chicks. Proc Soc Exp Biol Med 95:617–620
Davis KG, Mertz W (1987) Copper. In: Mertz W (ed) Trace elements in human and animal nutrition, 5th edn. Academic, New York, pp 301–364
Todd WR, Elvehjem CA, Hart EB (1934) Zinc in the nutrition of the rat. Am J Physiology 107:146–156
Stapleton SR (2000) Selenium: an insulin-mimetic. Cell Mol Life Sci 57:1874–1879
Lee WS, Berry MJ, Hediger MA, Larsen PR (1993) The type I iodothyronine 5′-deiodinase messenger ribonucleic acid is localized to the S3 segment of the rat kidney proximal tubule. Endocrinology 132:2136–2140
Uauy R, Olivares M, Gonzalez M (1998) Essentiality of copper in humans. Am J Clin Nutr 67:952S–959S
Coleman JE (1992) Zinc proteins: enzymes, storage proteins, transcription factors and replication proteins. Annu Rev Biochem 16:897–946
Coleman JE (1992) Structure and mechanism of alkaline phosphatase. Annual Review of Biophysico and Biomolecular Structure 21:441–483
Vallee BL, Falchuk KH (1993) The biochemical basis of zinc physiology. Physiol Rev 73:79–118
Chan S, Gerson B, Subramaniam S (1998) The role of copper, molybdenum, selenium, and zinc in nutrition and health. Clin Lab Med 18:673–685
NRC (2001) Nutrient requirements of dairy cattle, 7th edn. National Academy, Washington, DC
Pimentel JL, Cook ME (1987) Suppressed humoral immunity in chicks fed diets deficient in sodium, chloride or both sodium and chloride. Poultry Sci 66:2005
Pathak NN, Verma DN (1993) Nutrient requirement of Buffalo, 1st edn. International Book, Lucknow, UP, India
Henry RJ (1963) Standard methods of clinical chemistry, 1st edn. Harper and Row, New York, USA
Dumas BT, Watson WA, Briggs HG (1971) Albumin standards and the measurement of serum albumin with bromocresol green. Clin Chem Acta 31:87–96
Wybenga DR, Digiorgio J, Pileggi VJ (1971) Manual and automated methods for urea nitrogen measurement in whole serum. Clin Chem 17:891
Bonses RW, Tausskay HH (1945) The colorimetric determination of creatinine by the Jaffe reaction. J Biol Chem 158:581
Caraway WT (1963) Standard methods of clinical chemistry, volume 4. Seligson D (eds). Academic: New York. p 239
Kind PRM, King EJ (1954) Estimation of serum alkaline phosphatase activity by colorimetric method. J Clin Pathol 7:322
Reitman S, Frankel SE (1957) A colorimetric method for the determination of serum glutamic oxaloacetic transaminase and serum glutamic pyruvic transaminase. Am J Clin Pathol 28:56–63
Wroblewski VF, Duean LS (1955) Bestimmung der Aktivital der lactat-dehydrogenase. Proc Soc Exp Biol 90:210–214
Snedecor GW, Cochran WG (1989) Statistical methods, 8th edn. Iowa State University Press, Ames, Iowa
Steel RGD, Torrie JH (1980) Principles and procedures of statistics. A biometrical approach, 2nd edn. McGraw-Hill, New Delhi, India
Singh R, Randhawa SS, Dhillon KS (2002) Changes in blood biochemical and enzyme profile in experimental chronic selenosis in buffalo calves (Bubalus bubalis). Indian J Anim Sci 72:230–232
Mondal MK, Roy B, Biswas P (2004) Effect of supplementation of Cu on nutrient utilization by black Bengal kids. Indian J Anim Nutr 21:261–264
Arthur JR, Morrice PC, Beckett GJ (1988) Thyroid hormone concentrations in selenium deficient and selenium sufficient cattle. Res Vet Sci 45:122–123
Mudgal V, Garg AK, Dass RS, Varshney VP (2008) Effect of selenium and copper supplementation on blood metabolic profile in male buffalo (Bubalus bubalis) Calves. Biol Trace Elem Res 121:31–38
Khan SA (1978) Interaction of copper and zinc and its influence on the metabolism of major nutrients in growing calves. Ph.D. Thesis. Aligarh Muslim University. Aligarh, India
Shashidhar G, Prasad T (1993) Influence of selenite and selenomethionin administration on serum transaminases and hematology of goats. Indian J Anim Nutr 10:1–6
Du Z, Hemken RW, Harmon RJ (1996) Copper metabolism of Holstein and Jersey cows and heifers fed diets high in cupric sulphate or copper propionate. J Dairy Sci 79:1873–1880
Fehrs MS, Miller WJ, Gentry RP, Neathery MW, Blackmon DM, Heinmiller SR (1981) Effect of high but non-toxic dietary intake of copper and selenium on metabolism in calves. J Dairy Sci 64:1700–1706
Jenkins KJ, Hidiroglou M (1989) Tolerance of the calf for excess copper in milk replacer. J Dairy Sci 72:150–156
Terasawa E, Fernandez DL (2001) Neurobiological mechanisms of the onset of puberty in primates. Endocr Rev 22:111–151
Chadio SE, Kotsampasi BM, Menegatos JG, Zervas GP, Kalogiannis DG (2006) Effect of selenium supplementation on thyroid hormone levels and selenoenzyme activities in growing lambs. Biol Trace Elem Res 109:145–154
Wichtel JJ, Craigie AL, Freeman DA, Varela-Alvarez H, Williamson NB (1996) Effect of selenium and iodine supplementation on growth rate and on thyroid and somatotropic function in dairy calves at pasture. J Dairy Sci 79:1865–1872
Feng Q, Xiaoping Z, Wei Z, Jiaping Z, Shiwei Z, Zhihai J (2011) Effects of dietary iodine and selenium on nutrient digestibility, serum thyroid hormones, and antioxidant status of Liaoning cashmere goats. Biol Trace Elem Res. doi:10.1007/s12011-011-8981-5
Acknowledgment
First author is thankful to Indian Council of Agricultural Research, Krishi Bhawan, New Delhi, for providing financial assistance in the form of Senior Research Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mudgal, V., Garg, A.K., Dass, R.S. et al. Effect of Selenium, Zinc, and Copper Supplementation on Blood Metabolic Profile in Male Buffalo (Bubalus bubalis) Calves. Biol Trace Elem Res 145, 304–311 (2012). https://doi.org/10.1007/s12011-011-9209-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-011-9209-4