Abstract
The aim of this study was to investigate the influence of mineral sources on broiler breeders and their offsprings. Broiler breeding hens were fed with diets containing either organic or inorganic trace minerals at equal levels, i.e., (1) control group was fed with basal diet supplemented with inorganic trace minerals; (2) OZ group was fed with organic Zn instead of sulfate; and (3) OTM group was fed with organic Cu, Mn, Zn, and Se instead of inorganic sources. Results indicated that OTM supplementation decreased plasma cholesterol and triglyceride and increased yolk triglyceride via increasing high-density lipid protein cholesterol and decreasing low-density lipid protein cholesterol and very low-density lipid protein (VLDL) in plasma. OZ diets decreased plasma cholesterol and triglyceride mainly by reducing VLDL concentration. For control group, increased lipid concentrations resulted in increased lipid peroxidation in serum and malondialdehyde retention in yolk. Zn retention was not affected. Otherwise, OZ diet was observed to decrease Cu in yolk and albumen. While for OTM group, albumen Cu, albumen Se, and hepatic Se of hatched chicks were increased, but yolk Cu was decreased. Moreover, organic mineral supplementations improved broilers’ growth performance. In conclusion, organic mineral supplementation in breeders’ diets protected breeders from lipid peroxidation, increased egg nutrition retention, and benefit for growth of broilers.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Maternal nutrition was found to play important roles in fetal growth and organogenesis and could permanently change growth, physiology, and metabolism of offsprings [1, 2]. Therefore, researchers paid more attention to the relations between maternal nutrition and “fetal programming” or “fetal origins of adult disease.” For animal husbandry, it is necessary and useful to understand the relations between maternal nutrition and growth of the offsprings. In poultry, embryos develop outside of hen’s body. Nutritional status in egg is crucial to its development. Maternal nutrition could affect embryo development by modulating nutrients delivery from breeders’ blood to egg. As essential elements for animals, minerals were proved to play critical roles in embryo development [3]. Triglyceride, as a key energy source during bovine oocyte maturation, could maintain normal blastocyst formation and cell oxygen consumption [4]. And cholesterol was found to be indispensable in maintenance of membrane structure and action of developmental gene expression [5]. On the other hand, maternal health condition could also affect forms of nutrient retention in egg, so as to affect embryo development.
Copper (Cu), zinc (Zn), manganese (Mn), and selenium (Se) participate in a wide variety of physiological processes of animals [6]. Cu, Zn, Mn, and Se were found to be important for apolipoprotein (apo), the major structural protein of lipoprotein, so as to participate in lipid metabolism [7–11]. Cu, Zn, Mn, and Se are also essential for catalytic activity and proper spatial conformation of antioxidant enzymes such as superoxide dismutase (SOD), catalase, and glutathione peroxidase (GSH-Px) and thereby play important roles in the antioxidant defense and lipid peroxidation [12]. In accordance with mechanism above, Se was observed to exhibit protective roles on risks of oxidative damage by lowering lipid concentrations and increasing antioxidant enzyme activities when animals were challenged with cigarette smoke and enflurane anesthetized [13, 14]. For poultry, lowered lipid peroxidation in plasma might reduce lipid peroxidation product retention in yolk via nutrient deposition so as to benefit embryo and offsprings’ development.
Recent data indicated that inclusion of organic trace minerals in broiler breeder diets improved livability [15], cardiac output, immune system endpoint [16], and mineral retention [17] but did not affect growth performance [15, 18] or organ development of their progeny [19]. But reasons behind were not well discussed. Based on the roles of Cu, Zn, Mn, and Se play in lipid metabolism and antioxidant defense and improved bioavailability of organic minerals, we hypothesized that organic minerals might alter lipid metabolism, antioxidant enzyme activities, and mineral metabolism in breeders so as to affect nutrients retention in egg and finally affect the development of offsprings. This study was designed to estimate the effects of Cu, Zn, Mn, and Se sources on blood lipid metabolism and antioxidant status of broiler breeders, mineral retention in yolk, and the growth of their offsprings.
Materials and Methods
Experimental Diets and Breeder Husbandry
The design and conduct of this study were approved by the Institutional Animal Care and Use Committee of China Agricultural University. A total of 120 31-week-old Cobb 48 broiler breeder hens were randomly assigned to three treatments (five replicates of eight hens each) and fed with three different diets containing either organic or inorganic trace mineral supplementations at equal levels. The control group was fed with basal diet supplemented with inorganic trace minerals (Cu 8 mg from CuSO4, Zn 50 mg from ZnSO4, Mn 60 mg from MnSO4, and Se 0.3 mg from Na2SeO3 separately per kilogram of diet). The second (OZ) group was fed with organic Zn (Mintrex-Zn) instead of sulfate. The third (OTM) group was fed with organic Cu, Mn, Zn (Mintrex-Cu, Mn, Zn, Novus Intl., USA), and Se (selenium yeast, ALLtech Inc., USA) instead of inorganic sources. The diets were formulated based on NRC [20]. Feed composition is listed in Table 1. Trace minerals in basal diets were Cu 4.6 mg, Mn 35.3 mg, Zn 47 mg, and Se 0.18 mg/kg diet based on actual analysis. All diets were iso-energetic, iso-nitrogenous, and iso-methionine.
All birds were housed in a completely enclosed, ventilated, conventional caged-breeder house in which the lighting program was 16:8 L/D. Breeder hens were allotted 160 g of feed for each at 6:00 am every day. Male breeders were caged and given a commercial diet. Hens were artificially inseminated, and hatching eggs laid during 39 weeks of age were incubated.
Broiler Husbandry and Experimental Design
Hatched chicks were collected and vent sexed. Healthy male chicken were randomly selected and reared regarding to hens’ treatments, housed in six cages with six chicks for each treatment. All birds were fed with a common commercial ration with inorganic minerals (supplemental Cu 8 mg, Zn 50 mg, Mn 60 mg, and Se 0.3 mg per kg diet). Feed intake and body weight of male chicks were determined at 42 days.
Lipid and Antioxidant Profile Analysis
Plasma, serum, and yolk were obtained from breeders at the end of 35 and 39 weeks of age. Total cholesterol (TC), low-density lipid protein cholesterol (LDL-C), high-density lipid protein cholesterol (HDL-C), triglyceride (TG), very low-density lipid protein (VLDL) concentration, and total antioxidant capacity (T-AOC) were determined in plasma. CuZn-SOD, GSH-Px activity, and malondialdehyde (MDA) concentration were measured in serum. TC and TG in yolk were also determined.
TC, TG, LDL-C, HDL-C, T-AOC, CuZn-SOD, GSH-Px, and MDA were measured using commercial analytical kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). TC and TG concentration in yolk were determined with preparation methods described by Menge et al. [21]. VLDL concentration was determined as previously described by Griffin and Whitehead [22].
Mineral Retention
Cu, Mn, Zn, and Se were determined in yolk and albumen collected at 39 weeks of age and liver of 1-day-old chicks. Cu, Zn, and Mn were determined from microwave wet digests with inductively coupled plasma atomic emission spectrometry (Iris Intrepid, Thermo Elemental, USA) after microwave (MARS 5, CEM Corp., USA) wet digestion in nitric acid. For Se determination, samples were wet digested using nitric/perchloric acid mixture on sand-bath. Se was detected as 4,5-benzopiaselenol, product of reaction between Se and 2,3-diaminonaphthalene, by fluorescence spectrometer (Hitachi 850) at 520 nm after excitation at 376 nm according to method described by Li and Wang [23].
Statistical Analysis
All data were analyzed by one-way ANOVA using the SPSS for windows (version 16.0) program. And a post hoc Duncan’s multiple-range test was used to separate means that significantly differ at P < 0.05.
Results
Lipid Profile
Plasma TC, TG, HDL-C, LDL-C, and VLDL were measured in plasma (Table 2). No differences were observed at age 35 weeks except plasma TG. Breeders fed with OTM diet showed decreased (P < 0.05) TG in plasma collected at age 35 weeks. Plasma lipid profiles greatly differed among treatments at age 39 weeks. Plasma total cholesterol content was decreased (P < 0.01) by organic minerals supplementation and lowest in OTM group. Feeding with OTM diet significantly decreased (P < 0.01) LDL-C but increased (P < 0.05) HDL-C in plasma. No differences were observed in LDL-C and HDL-C in OZ group compared with control. However, VLDL was decreased (P < 0.05) by supplement of both OZ and OTM diets. A decrease in plasma TG concentration was also observed (P < 0.05) in both OZ and OTM groups.
No difference was observed in TC concentration in yolk (Table 3). But yolk TG concentration was increased (P < 0.01) in OZ and OTM groups at age 35 weeks and OTM at age 39 weeks.
Antioxidant Profile
Plasma T-AOC and serum MDA concentration of breeders were not affected (P > 0.10) by mineral sources at age 35 weeks (Table 4). However, supplementation of OZ and OTM diets increased (P < 0.01) GSH-Px but decreased (P < 0.01) CuZn-SOD activity in serum at age 35 weeks. At 39 weeks of age, a decrease was observed (P < 0.01) in plasma T-AOC and serum MDA concentration when feeding breeders with organic minerals. No difference was observed (P > 0.10) in serum GSH-Px activity. But an increase in serum CuZn-SOD activity was detected (P < 0.01) in both OZ and OTM groups.
Yolk MDA concentration was also determined on samples collected at 39 weeks of age. OZ and OTM supplementations decreased yolk MDA concentration (control, 15.03 nmol/ml; OZ, 8.99 nmol/ml; OTM, 9.59 nmol/ml; SEM = 0.77; P < 0.01).
Mineral Retention
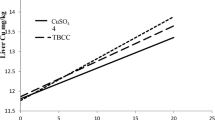
Effects of mineral sources on mineral retention in yolk, albumen, and liver of 1-day-old chicks were showed in Table 5. Zn was not affected by mineral sources in all tissues (P > 0.10). Cu concentration in yolk was decreased (P < 0.05) by supplementing organic minerals. While in albumen, Cu was increased (P < 0.05) in OTM group, while decreased in OZ group. No differences were observed (P > 0.10) in hepatic Cu concentration. Supplementation of OZ diet did not affect Se retention (P > 0.10). However, OTM supplementation showed increased Se concentration in albumen (P < 0.01) and liver of 1-day-old chicks (P < 0.05).
Broiler Performance
The average feed intake at 42 days did not differ among chicks from breeders fed different sources of minerals (Table 6). However, chicks from OZ and OTM dams weighed more than control at 42 days (P = 0.051). Feed conversion ratio was decreased due to replacing inorganic minerals by a combination of organic forms (P < 0.05).
Discussion
Lipid Profile
Cholesterol and triglyceride are considered as essential agents in embryonic development [4, 5]. Decreased cholesterol and triglyceride in plasma and increased in yolk could be good for breeders’ health and progeny development.
In this study, supplementations of organic Zn and a combination of organic Cu, Zn, Mn, and Se were found to decrease total cholesterol and total triglyceride levels in plasma at 39 weeks of age, also accompanied with alterations in plasma LDL-C, HDL-C, VLDL concentrations, and yolk TG level. LDL, HDL, and VLDL were proved to be major cholesterol and triglyceride transporters in laying hens [24]. HDL could clear cholesterol from plasma via promoting cholesterol delivery from the periphery to liver and excretion in the bile or to ovarian tissues [25]. In this study, OZ diets seemed to affect plasma TC and TG mainly by reducing VLDL concentration. While for the OTM group, increased HDL-C and decreased LDL-C and VLDL induced lowered TC and TG in plasma. Apo B, whose mRNA editing is zinc dependent [11], is the major structural protein for both VLDL and LDL [26]. Zn might affect lipid metabolism by alteration of apolipoprotein expression. Moreover, Cu, Mn, and Se were also reported to be associated with apo B [10], apo E [9], and apo A [7] synthesis, which playing critical roles in lipoprotein metabolism. Different sources of minerals might change lipid metabolism by regulating apolipoprotein synthesis.
Antioxidant Profile
Antioxidant defense is organized at three principal levels of protection: prevention, interception, and repair [27]. Antioxidant systems involved in interception period contain enzymatic and nonenzymatic antioxidant systems. GSH-Px and CuZn-SOD are two major enzymes in the enzymatic system and participate in antioxidant defense by clearance of reactive oxygen species (ROS) [27].
From this study, organic Zn or Cu, Mn, Zn, and Se combinations was observed to play protective roles in lipid oxidation, such as decreased TC, TG, and MDA concentrations in plasma. Protective roles of Se were also found when animals were challenged with cigarette smoke and enflurane anesthetized [13, 14]. But an inconsistence was observed between T-AOC and MDA (product of lipid peroxidation). In this study, no differences were observed in T-AOC in plasma at 35 weeks of age, but an increase was found in control group at age 39 weeks. Higher T-AOC indicated stronger antioxidant defense and might decrease oxidative damage. However, MDA was increased accompanied with the increased T-AOC. The inconsistent results between T-AOC and MDA can be explained by adaptive response. The body exhibits adaptive responses to oxidative challenge for cell survival [27]. Increased plasma TC and TG levels in control group lead to increased risks of lipid peroxidation. The body modulates antioxidant system, such as CuZn-SOD and GSH-Px activity, to scavenge ROS. And when the ROS scavenging fails, these species lead to increased MDA formation which results in an increased MDA deposition in yolk.
In accordance with the adaptive response hypothesis above, CuZn-SOD, as the first line of defense against ROS [28], was transiently increased in control group during the initial antioxidant defense at age 35 weeks due to the increased risks of lipid peroxidation and then decreased at age 39 weeks which indicated a lower bioavailability of inorganic mineral in maintenance of CuZn-SOD activity. GSH-Px is a selenoprotein which reduces lipidic or nonlipidic hydroperoxides as well as H2O2 [28]. Supplementation of Se yeast in broiler diet increased plasma GSH-Px activity via modification of enzyme structure [29]. But it is hard to explain why GSH-Px activity was increased by organic Zn supplementation. Increased GSH-Px activity was also observed by Bun et al. [30] when feeding broilers with increased organic zinc levels.
Mineral Retention
Antagonism between Zn and Cu was observed by Hall et al. [31]. Skrivan and his co-workers also found a decreased Cu concentration in yolk when fed with 80 mg/kg Zn. In accordance with results above, Cu concentration in yolk was decreased by supplementation of organic Zn and the combination of organic Cu, Zn, Mn, and Se. However, an increase was observed in albumen Cu by OTM supplementation, which may indicate increased Cu absorption by organic Cu supplementation. One perceived benefit for organic minerals is facilitating the absorption of other minerals [32]. However, Zn-MHA did not benefit for Cu absorption, which may indicate exist of antagonism between Zn and Cu and separation of Zn and its ligands in the body. Supplementation of OTM showed increased Se deposition in albumen and increased uptake of Se of embryo which was consistent with results of Paton et al. [33].
Broiler Growth Performance
Maternal nutritions, such as Zn, Mn, and Se, play important roles in embryo development [3]. Some researchers found no differences in growth, livability, and carcass composition of chicks from breeders fed with different sources of Cu, Zn, or Mn [15, 18, 34]. However, others found increased livability [14] and bone development [35] of 1-day-old chicks from breeders fed with organic minerals.
In this study, increased body weight and feed efficiency were observed in chicks from dams fed OTM diets at 42 days. Supplementation of organic zinc slightly increased body weight but did not affect feed efficiency. Minerals, triglyceride, and cholesterol were proved to be critical to embryo development [3–5]. Increased egg nutrition deposition and decreased lipid peroxidation product retention in yolk might play a role in the increased growth performance of chicks.
Conclusion
In conclusion, supplementations of organic Zn and a combination of organic Cu, Zn, Mn, and Se in breeders’ diets were observed to have protective effects on breeders via increasing cholesterol and triglyceride clearance from plasma and decreasing plasma lipid peroxidation. Maternal nutrition could affect chicks’ development by regulating amounts and forms of nutrients retention in egg. Supplementations of organic minerals increased triglyceride delivery and decreased lipid peroxidation product retention in yolk, which may be reasons induced improved growth performance of chicks. Moreover, the combination of organic Cu, Zn, Mn, and Se appeared to be much more effective than that of organic Zn. For poultry husbandry, these results seem to be useful. We suggest the uses of organic Zn and combination of organic Cu, Zn, Mn, and Se in breeders’ diets due to its beneficial effects on offsprings’ growth and development. However, more studies should be done to uncover the underlying causes of long-term effects of growth of chicks.
Abbreviations
- Cu:
-
Copper
- Zn:
-
Zinc
- Mn:
-
Manganese
- Se:
-
Selenium
- Apo:
-
Apolipoprotein
- SOD:
-
Superoxide dismutase
- GSH-Px:
-
Glutathione peroxidase
- TC:
-
Total cholesterol
- TG:
-
Total triglyceride
- LDL-C:
-
Low-density lipid protein cholesterol
- HDL-C:
-
High-density lipid protein cholesterol
- VLDL:
-
Very low-density lipid protein
- T-AOC:
-
Total antioxidant capacity
- MDA:
-
Malondialdehyde
- ROS:
-
Reactive oxygen species
References
Jou M, Philipps AF, Lonnerdal B (2010) Maternal zinc deficiency in rats affects growth and glucose metabolism in the offspring by inducing insulin resistance postnatally. J Nutr 140:1621–1627
Vonnahme KA, Hess BW, Hansen TR, McCormick RJ, Rule DC, Moss GE, Murdoch WJ, Nijland MJ, Skinner DC, Nathanielsz PW, Ford SP (2003) Maternal undernutrition from early- to mid-gestation leads to growth retardation, cardiac ventricular hypertrophy, and increased liver weight in the fetal sheep. Biol Reprod 69:133–140
Wilson HR (1997) Effects of maternal nutrition on hatchability. Poult Sci 76:134–143
Ferguson EM, Leese HJ (2006) A potential role for triglyceride as an energy source during bovine oocyte maturation and early embryo development. Mol Reprod Dev 73:1195–1201
Roux C, Wolf C, Mulliez N, Gaoua W, Cormier V, Chevy F, Citadelle D (2000) Role of cholesterol in embryonic development. Am J Clin Nutr 71:1270–1279
Richards JD, Zhao J, Harrell RJ, Atwell CA, Dibner JJ (2010) Trace mineral nutrition in poultry and swine. Asian Austral J Anim 23:1527–1534
Bleys J, Navas-Acien A, Stranges S, Menke A, Miller ER, Guallar E (2008) Serum selenium and serum lipids in US adults. Am J Clin Nutr 88:416–423
Faure P, Roussel AM, Richard MJ, Foulon T, Groslambert P, Hadjian A, Favier A (1991) Effect of an acute zinc depletion on rat lipoprotein distribution and peroxidation. Biol Trace Elem Res 28:135–146
Kawano J, Ney DM, Keen CL, Schneeman BO (1987) Altered high density lipoprotein composition in manganese-deficient Sprague-Dawley and Wistar rats. J Nutr 117:902–906
Nassir F, Mazur A, Sérougne C, Gueux E, Rayssiguier Y (1993) Hepatic apolipoprotein B synthesis in copper-deficient rats. FEBS Lett 322:33–36
Reaves SK, Fanzo JC, Wu JYJ, Wang YR, Wu YW, Zhu L, Lei KY (1999) Plasma apolipoprotein B-48, hepatic apolipoprotein B mRNA editing and apolipoprotein B mRNA editing catalytic subunit-1 mRNA levels are altered in zinc-deficient rats. J Nutr 129:1855–1861
Barandier C, Tanguy S, Pucheu S, Boucher F, De Leiris J (1999) Effect of antioxidant trace elements on the response of cardiac tissue to oxidative stress. Ann Ny Acad Sci 874:138–155
Cay M, Naziroglu M, Koylu H (2009) Selenium and vitamin E modulates cigarette smoke exposure-induced oxidative stress in blood of rats. Biol Trace Elem Res 131:62–70
Naziroglu M (1999) Protective role of intraperitoneally administered vitamin E and selenium in rats anesthetized with enflurane. Biol Trace Elem Res 69:199–209
Virden WS, Yeatman JB, Barber SJ, Zumwalt CD, Ward TL, Johnson AB, Kidd MT (2003) Hen mineral nutrition impacts progeny livability. J Appl Poult Res 12:411–416
Virden WS, Yeatman JB, Barber SJ, Willeford KO, Ward TL, Fakler TM, Wideman RF Jr, Kidd M (2004) Immune system and cardiac functions of progeny chicks from dams fed diets differing in zinc and manganese level and source. Poultry Sci 83:344–351
Surai PF, Karadas F, Pappasa AC, Sparks NHC (2006) Effect of organic selenium in quail diet on its accumulation in tissues and transfer to the progeny. Br Poultry Sci 47:65–72
Hudson BP, Dozier WA, Wilson JL (2005) Broiler live performance response to dietary zinc source and the influence of zinc supplementation in broiler breeder diets. Anim Feed Sci Tech 118:329–335
Hudson BP, Fairchild BD, Wilson JL, Dozier WA, Buhr RJ (2004) Breeder age and zinc source in broiler breeder hen diets on progeny characteristics at hatching. J Appl Poult Res 13:55–64
National Research Council (1994) Nutrient requirements of poultry. National Academy of Sciences, Washington, D.C
Menge H, Littlefield LH, Frobish LT, Weinland BT (1974) Effect of cellulose and cholesterol on blood and yolk lipids and reproductive efficiency of the hen. J Nutr 104:1554–1566
Griffin H, Whitehead C (1982) Plasma lipoprotein concentration as an indicator of fatness in broilers: development and use of a simple assay for plasma very low density lipoproteins. Brit Poult Sci 23:307–313
Li J, Wang X (2004) Effect of dietary organic versus inorganic selenium in laying hens on the productivity, selenium distribution in egg and selenium content in blood, liver and kidney. J Trace Elem Med Bio 18:65–68
Elkin RG, Freed MB, Kieft KA, Newton RS (1993) Alteration of egg yolk cholesterol content and plasma lipoprotein profiles following administration of a totally synthetic HMG-CoA reductase inhibitor to laying hens. J Agr Food Chem 41:1094–1101
Grummer R, Carroll D (1988) A review of lipoprotein cholesterol metabolism: importance to ovarian function. J Anim Sci 66:3160–3173
Egusa G, Brady DW, Grundy SM, Howard BV (1983) Isopropanol precipitation method for the determination of apolipoprotein B specific activity and plasma concentrations during metabolic studies of very low density lipoprotein and low density lipoprotein apolipoprotein B. J Lipid Res 24:1261–1267
Sies H (1993) Strategies of antioxidant defense. Eur J Biochem 215:213–219
Michiels C, Raes M, Toussaint O, Remacle J (1994) Importance of SE-glutathione peroxidase, catalase, and Cu/Zn-SOD for cell survival against oxidative stress. Free Radic Bio Med 17:235–248
Wang Y, Xu B (2008) Effect of different selenium source (sodium selenite and selenium yeast) on broiler chickens. Anim Feed Sci Tech 144:306–314
Bun SD, Guo YM, Guo FC, Ji FJ, Cao H (2011) Influence of organic zinc supplementation on the antioxidant status and immune responses of broilers challenged with Eimeria tenella. Poult Sci 90:1220–1226
Hall AC, Young BW, Bremner I (1979) Intestinal metallothionein and the mutual antagonism between copper and zinc in the rat. J Inorg Biochem 11:57–66
Mateos GG, Lazaro R, Astillero JR, Serrano MP (2005) Trace minerals: what text books don't tell you. In: Taylor-Pickard J, Tuker L (eds) Re-defining mineral nutrition. Nottingham University Press, Nottingham, pp 41–43
Paton ND, Cantor AH, Pescatore AJ, Ford MJ, Smith CA (2002) The effect of dietary selenium source and level on the uptake of selenium by developing chick embryos. Poult Sci 81:1548–1554
Kidd MT, Anthony NB, Lee SR (1992) Progeny performance when dams and chicks are fed supplemental zinc. Poult Sci 71:1201–1206
Oviedo-Rondon E, Meija-Sanchez L, Small J, Brannan K, Leksrisompong N, Mitchell A, York T, Brake J (2008) Organic trace minerals (Mintrex P) in broiler breeder diets and pre-incubation conditions on bone development and leg health of broilers. In: International Poultry Science Forum Proceedings, p 17
Acknowledgments
This work was supported by the earmarked fund for Modern Agro-Industry Technology Research System and the Yangtz River Scholar and Innovation Research Team Development Program (project no. IRT0945).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, Q., Guo, Y., Ma, S. et al. Dietary Mineral Sources Altered Lipid and Antioxidant Profiles in Broiler Breeders and Posthatch Growth of Their Offsprings. Biol Trace Elem Res 145, 318–324 (2012). https://doi.org/10.1007/s12011-011-9196-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-011-9196-5