Abstract
The objective of the present study was to evaluate the potential effect of maternal cadmium exposure on pregnancy outcome and development in the offspring at age 4.5 years. Between November 2002 and December 2003, 109 normal pregnant women were enrolled in our cohort from Da-Ye Country, Hubei Province in Central China. The placental, whole blood, and cord blood levels of cadmium were determined by inductively coupled plasma mass spectrometer (ICP-MS). The 106 children at 4.5 years of age given birth by the aforementioned women were followed up and the following rate was 97.25%. Detailed questionnaire surveys, anthropometric measurements were performed, and IQ development was evaluated by Wechsler Preschool and Primary Scale of Intelligence Revised Edition (WPPSI-R). Multiple linear regression analysis indicated that cord blood cadmium level was significantly negatively correlated with fetus development. Low birth weight (less than 2,500 g) occurred significantly more frequently in infants with higher cord blood cadmium than in those exposed to lower levels of cord blood cadmium. Significantly negative correlation was found between cord blood cadmium exposure and WPPSI-R IQ full score after controlling for confounding variables. It was concluded that cord blood cadmium concentration was a factor that influenced fetus growth and later IQ development.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Many studies have indicated that even low-level exposure to heavy metals, such as lead, cadmium, and mercury, have some toxic effects on development. Cadmium is a ubiquitous environmental pollutant that has no known biological function in human beings, and it is listed in the seventh place in the hazardous substances register of the United States Agency for Toxic Substances and Disease Registry (US-ATSDR) [1, 2]. Environmental and occupational exposure to cadmium is implicated in a number of clinical conditions, primarily renal dysfunction and bone disease [3], but also some cancers [4]. Compared with postnatal exposure, prenatal exposure to various toxins often has more adverse developmental effects [5]. Cadmium was found to be toxic to the fetus prior to and during gestation in rats and mice, resulting in reduced fetal or pup weights, malformations, and impaired neurobehavioral development [6–9]. Several epidemiological studies showed that cadmium exposure of pregnant women might influence pregnancy outcome and neonatal birth weight. A study on pregnant women living in an area close to a copper smelter suggested that exposure to lead and cadmium could influence the development of complications in pregnancy such as threatened spontaneous abortion, toxemia, and anemia, by increased lipid peroxidation [10–12]. Laudanski et al. [13] found that the mean blood concentration of cadmium in mothers delivered of preterm infants was higher than that of women who went to full term in an area with high amounts of lead and cadmium in the soil. Nishijo et al. reported that maternal exposure to cadmium seems to increase early delivery and transfer in part to the next generation through breast milk after birth [14].
However, most of conclusions about the relationship between cadmium exposure and fetus development were obtained from analyzing maternal blood cadmium concentration. In fact, placenta acts as a barrier protecting the fetus from toxic metals, such as mercury and cadmium. Grandjean et al. [15] reported that cord blood mercury was a better measure of fetal exposure than maternal blood mercury. Our early studies with limited samples revealed that higher cord blood cadmium level might be associated with a significant decrease in neonatal birth height [16]. Moreover, the relationship between children’s IQ and their prenatal cadmium exposure has not been studied. Given this point, the purpose of our study was to evaluate the effects of prenatal cadmium exposure on pregnancy outcomes and child development.
Materials and Methods
Subjects and Data Collecting
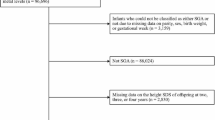
The prospective cohort study was conducted from November 2002 to August 2007. The Da-Ye county of Hubei Province in Central China was chosen as the study area. In 2002, the cadmium levels in water used for irrigation in this area were 0.26 mg/L, and those in soil were 1.41–9.01 mg/kg as a result of the operation of a metal smelter operating in Da-Ye. All healthy pregnant women with single birth, aged 20–34 years, and had lived in Da-Ye city at least 15 years were included in this study. Those with stillbirth, or with a history of occupational cadmium exposure, or with a history of calcium, iron, vitamin D, or compound trace element supplement were excluded. Those with hepatic disease, or nephropathy and endocrine disease were also excluded. According to this criterion, 109 pregnancy women were included in the study. The questionnaire and interview provided the necessary information about the subjects’ demographics, general state of health, history of past diseases and medication, habits of smoking and drinking, reproductive history, and gestational age, etc.
Follow-up information about their children’s development was obtained when the children were, on average 54 months of age. Three children were excluded, one of them changed residence, the other two died from leukemia and drowning, respectively. 106 children were followed-up and all these children lived in their birthplace. The follow-up questionnaire and interview provided the necessary information about the children’s general state of health, variables likely to affect children’s development such as maternal gestational weeks, gravidity, breast feeding, method of delivery, maternal educational status, environmental tobacco smoke (ETS) exposure, per capita annual income of family, and so on were obtained. The performance, verbal, and full IQ scores from the WPPSI-R administered were used to evaluate IQ development of these children. We used the WPPSI-R (Wechsler 1989), rather than the third edition (Wechsler 1991) because it is widely used in China [17]. Assessments were performed at the children’s home; on the WPPSI-R (standardized mean, 100 ± 15), a score of 89–80 is low average, 79–70 is borderline, and ≤69, intellectually deficient.
Biological Sample Collection
Hospital staff collected maternal blood within 1 week before delivery and cord blood at delivery. Cord blood samples were drawn by puncture right after parturition. In each case, 3 ml blood was collected and stored at −20℃ until needed for analysis. The placentas were divided into eight average-size sections through the center, from which two symmetric sections were sampled. The sampled two sections were freeze-dried, ground to a powder, and stored at −20℃. The blood and placental samples were digested by HNO3/HClO4, and diluted with HNO3.
Elements Determination
The cadmium at m/z 111 and lead at m/z 208 in blood and tissue samples were measured by means of inductively coupled plasma mass spectrometer (ICP-MS) model DRCII from Perkin-Elmer (USA). The detection limits of cadmium and lead were 0.005 and 0.0007 ng/mL, respectively. Certified Reference Materials GBW09133 (Beijing, China) and GBW08551 were used for quality control. The analytical results of Certified Reference Materials were along with the certified values (Table 1). In addition, 10% of the samples were selected for parallel analysis. The coefficient of variance for the duplicate determinations of maternal blood cadmium, cord blood cadmium, placenta cadmium, and cord blood lead was 6.7%, 5.7%, 2.5%, and 1.4%, respectively.
Statistical Analysis
If needed, variables were natural log-transformed, and nonparametric test was used. Student’s t test was used in the analysis of data to find differences between two groups. Stepwise multiple linear regression was used to analyze the influence of the gestational cadmium exposure. The significance level for entering the regression model was set as 0.30 and for staying it was set as 0.10. Statistical analyses were performed with SPSS 13.0 software, and p ≤ 0.05 indicated statistical significance.
Results
General Characteristics of the Subjects
The average height and weight of the women prior to delivery was 159.5 cm and 66.0 kg. Of the 109 pregnant women, 45.9% were farmers, and 13.8% had over 9 year of schooling. In addition, 48.6% had a history of previous gestation, 43.1% had a history of previous lactation. None of the women participating in the study had a history of smoking, which is a factor resulting in elevated levels of cadmium. The cadmium and lead concentration in maternal blood, cord blood, and placenta were shown in Table 2.
Correlations Between Cord Blood Cadmium Concentration and Pregnancy Outcomes
Given that cord blood cadmium, in fact, represents the absolute amount of cadmium that reaches the fetus through the placenta, we used cord cadmium for all outcome analyses. The infants were divided into two groups according to the median of cord blood cadmium concentration (0.6 µg/L).
No significant differences was found in the sex constituent ratio between the low- and high-cadmium groups (χ 2 = 0.0324, p > 0.05). Length and weight at birth of the infants exposed to higher cord blood cadmium concentration were significantly lower than those of infants exposed to lower cord blood cadmium (t = 2.89, p < 0.01; t = 4.58, p < 0.01). Similarly, Fisher’s exact test revealed that the rate of low birth weight (less than 2500 g) of infants with higher cord blood cadmium was significantly higher than those of infants with lower cord blood cadmium (χ 2 = 6.829, p < 0.05) (Table 3).
We also analyzed the relationship between maternal blood cadmium level and fetus growth. The neonatal birth height was negatively associated with the concentration of maternal blood cadmium (r = 0.2568, t = 2.71, p < 0.01). Negative correlation was found between the weights of neonates with maternal blood cadmium level. No correlation was found between the weight and height of neonates with the level of placenta cadmium (data not shown).These data further confirmed that cord blood cadmium level was a more reasonable marker in the evaluation of the effect of prenatal cadmium exposure.
Multiple linear regression analysis showed that cord blood cadmium level, cord blood lead level, maternal age, height, weight, gravidity, and gestational weeks, EST, could explain 83.62% and 78.40% of the variation of neonatal birth length and weight, respectively. After control of those potential confounding factors, cord blood cadmium level was still significantly negatively associated with neonatal birth height (t = -2.79, p < 0.05) and weight (t = -2.05, p < 0.05), showing that cord blood cadmium concentration was an independent factor affecting fetus growth (Table 4). These results are partially consistent with our early study [16].
Correlations Between Cord Blood Cadmium Concentration and Children Development at 4.5 Years Old
The height of children in higher cord blood cadmium group was significantly lower than those in lower group (t = 4.44, p < 0.01). Moreover, the WPPSI-R IQ full score and performance of children in lower cord blood cadmium group was significantly higher than those in higher group (t = 2.44, p < 0.05; t = 2.04 p < 0.05). No significant differences were found in verbal abilities between higher and lower cord blood cadmium groups (Table 3). After adjustment for cord blood lead, maternal age, height, weight, gestational weeks, maternal educational status, method of delivery, feeding methods, nursery school age, ETS and per capita annual income of the family, a significantly negative correlation was found between cord blood cadmium and children’s height. Similarly, significantly negative correlations were found between cord blood cadmium and WPPSI-R IQ full and performance score (Tables 5 and 6).
Discussion
Exposure to cadmium is a serious public health concern, and a number of previous studies have reported an inverse relationship between maternal cadmium exposure and infant body weight [11, 13, 18–22]. In addition to occupational and environmental exposure, pregnant women are also exposed through smoking. In the present study, no subject has the history of occupational exposure and smoking. So cadmium exposure of these subjects mainly came from environmental exposure. It has been reported that maternal cadmium exposure can produce several adverse reproductive outcomes, including preterm deliveries and reduced infant body weight. But most of studies mainly analyzed the correlation between maternal blood or placenta cadmium level and pregnancy outcome. In fact, placenta acts as a barrier protecting the fetus from toxic metals, such as lead, cadmium, and mercury [15, 23, 24]. These metals, especially cadmium, accumulate in human placenta [24–26]. Cord blood cadmium, in fact, represents the absolute amount of cadmium that reaches the fetus through the placenta. So in the present study, we analyzed the effect of cord blood cadmium on pregnancy outcomes and later development of children.
Pregnancy outcome and development in children were affected by a lot of heredity and environmental factors. To study the effects of cadmium exposure duration pregnancy on offspring, many variables that we could measure were introduced into the analysis, such as maternal age, gravidity, gestational weeks, maternal educational status, methods of delivery, feeding methods, nursery school age, EST, and per capita annual income. It is known to all that no level of lead exposure appears to be “safe,” and even the current “low” levels of exposure in children are associated with neurodevelopmental deficits [27]. Besides postnatal exposure, prenatal exposure to lead also has adverse effects on the IQ development of children. A Yugoslavia study found that increased maternal blood Pb concentration in mid-pregnancy (12–20 weeks) was significantly associated with decreased 3- to 7-year IQ regardless of pattern of postnatal exposure [28]. Another Cincinnati, Ohio (USA) study showed lasting significant effects of maternal blood Pb between 6 and 28 weeks on factor scores representing attention and visuoconstruction in adolescents when prenatal blood Pb was tested without simultaneously considering postnatal blood Pb exposure history [29]. So in the present study, cord blood Pb has been controlled when evaluating the effects of Cd exposure on pregnancy outcome and IQ development of offspring,
Compared with infant exposed to lower cord blood cadmium concentration, the body weight of infant exposure to higher cord blood cadmium was lower, and the body length was shorter. After excluding the effects of potential confounding factors, cord blood cadmium level was still an independent factor influencing fetal development. But Nishijo et al. reported that cadmium exposure did not greatly affect fetal development [14]. The reason leading to these different results may be due to the fact that a different biological sample (maternal urinary cadmium) was analyzed in their study.
We also observed the long-term effects of prenatal cadmium exposure on anthropometric measurement results and their offspring’s later IQ development. The height and WPPSI-R IQ full score of children at 4.5 years in the higher cord blood cadmium group were significantly lower than those of children in the lower group. This kind of correlation did not change after controlling potential confounding factors. Most researches about the adverse effects of prenatal low-level exposure to heavy metals on later IQ development have focused on lead. Needleman et al reported an adverse impact of low-level lead exposure on the intellectual and behavioral functioning of children with no overt lead-related symptomatology [30]. Many, but not all subsequent studies have found that low-level lead exposure did adversely impact on functioning in otherwise asymptomatic children [31]. But to our knowledge, there were few reports about the effects of prenatal cadmium exposure on later IQ development. Our results in the present that cadmium exposure during embryonic period had negative effects on offspring’s development are innovative, which made the understanding of cadmium toxic effect go a step further.
Nonetheless, it is clear that various other social, familial, and hereditary factors can make a contribution to pregnancy outcome and development in the offspring, and their effects should be controlled in future research about the adverse developmental effects of prenatal cadmium exposure.
References
Staessen, J. A., Vyncke, G., Lauwerys, R. R., Roels, H. A., Celis, H. G., Claeys, F., Dondeyne, F., Fagard, R. H., Ide, G., Lijnen, P. J., and et al. Transfer of cadmium from a sandy acidic soil to man: a population study. Environ Res, 58: 25–34, 1992.
Nampoothiri, L. P. and Gupta, S. Biochemical effects of gestational coexposure to lead and cadmium on reproductive performance, placenta, and ovary. J Biochem Mol Toxicol, 22: 337–344, 2008.
Kuriwaki, J., Nishijo, M., Honda, R., Tawara, K., Nakagawa, H., Hori, E., and Nishijo, H. Effects of cadmium exposure during pregnancy on trace elements in fetal rat liver and kidney. Toxicol Lett, 156: 369–376, 2005.
Jarup, L., Berglund, M., Elinder, C. G., Nordberg, G., and Vahter, M. Health effects of cadmium exposure—a review of the literature and a risk estimate. Scand J Work Environ Health, 24 Suppl 1: 1–51, 1998.
Slotkin, T. A., Kavlock, R. J., Cowdery, T., Orband, L., Bartolome, M., Gray, J. A., Rehnberg, B. F., and Bartolome, J. Functional consequences of prenatal methylmercury exposure: effects on renal and hepatic responses to trophic stimuli and on renal excretory mechanisms. Toxicol Lett, 34: 231–245, 1986.
Nagymajtenyi, L., Schulz, H., and Desi, I. Behavioural and functional neurotoxicological changes caused by cadmium in a three-generational study in rats. Hum Exp Toxicol, 16: 691–699, 1997.
Shen, H. M., Dong, S. Y., and Ong, C. N. Critical role of calcium overloading in cadmium-induced apoptosis in mouse thymocytes. Toxicol Appl Pharmacol, 171: 12–19, 2001.
Sorell, T. L. and Graziano, J. H. Effect of oral cadmium exposure during pregnancy on maternal and fetal zinc metabolism in the rat. Toxicol Appl Pharmacol, 102: 537–545, 1990.
Christensen, M. M., Keith, I., Rhodes, P. R., Graziano, F. M., Madsen, P. O., Bruskewitz, R. C., and Saban, R. A guinea pig model for study of bladder mast cell function: histamine release and smooth muscle contraction. J Urol, 144: 1293–1300, 1990.
Kuhnert, P. M., Kuhnert, B. R., Bottoms, S. F., and Erhard, P. Cadmium levels in maternal blood, fetal cord blood, and placental tissues of pregnant women who smoke. Am J Obstet Gynecol, 142: 1021–1025, 1982.
Kuhnert, B. R., Kuhnert, P. M., Debanne, S., and Williams, T. G. The relationship between cadmium, zinc, and birth weight in pregnant women who smoke. Am J Obstet Gynecol, 157: 1247–1251, 1987.
Kuhnert, P. M., Kuhnert, B. R., Erhard, P., Brashear, W. T., Groh-Wargo, S. L., and Webster, S. The effect of smoking on placental and fetal zinc status. Am J Obstet Gynecol, 157: 1241–1246, 1987.
Laudanski, T., Sipowicz, M., Modzelewski, P., Bolinski, J., Szamatowicz, J., Razniewska, G., and Akerlund, M. Influence of high lead and cadmium soil content on human reproductive outcome. Int J Gynaecol Obstet, 36: 309–315, 1991.
Nishijo, M., Nakagawa, H., Honda, R., Tanebe, K., Saito, S., Teranishi, H., and Tawara, K. Effects of maternal exposure to cadmium on pregnancy outcome and breast milk. Occup Environ Med, 59: 394–396; discussion 397, 2002.
Grandjean, P., Budtz-Jorgensen, E., Jorgensen, P. J., and Weihe, P. Umbilical cord mercury concentration as biomarker of prenatal exposure to methylmercury. Environ Health Perspect, 113: 905–908, 2005.
Zhang, Y. L., Zhao, Y. C., Wang, J. X., Zhu, H. D., Liu, Q. F., Fan, Y. G., Wang, N. F., Zhao, J. H., Liu, H. S., Ou-Yang, L., Liu, A. P., and Fan, T. Q. Effect of environmental exposure to cadmium on pregnancy outcome and fetal growth: a study on healthy pregnant women in China. J Environ Sci Health A Tox Hazard Subst Environ Eng, 39: 2507–2515, 2004.
Carvajal, H. H., Parks, J. P., Bays, K. J., Logan, R. A., Lujano, C. I., Page, G. L., and Weaver, K. A. Relationships between scores on Wechsler Preschool and Primary Scale of Intelligence—revised and Stanford-Binet IV. Psychol Rep, 69: 23-26, 1991.
Huel, G., Boudene, C., and Ibrahim, M. A. Cadmium and lead content of maternal and newborn hair: relationship to parity, birth weight, and hypertension. Arch Environ Health, 36: 221–227, 1981.
Sikorski, R., Radomanski, T., Paszkowski, T., and Skoda, J. Smoking during pregnancy and the perinatal cadmium burden. J Perinat Med, 16: 225–231, 1988.
Kostrzewska, A., Laudanski, T., and Bergelin, I. Influence of cadmium ions on the reactivity of isolated human uterine arteries. J Toxicol Environ Health, 34: 187–195, 1991.
Berlin, M., Blanks, R., Catton, M., Kazantzis, G., Mottet, N. K., and Samiullah, Y. Birth weight of children and cadmium accumulation in placentas of female nickel-cadmium (long-life) battery workers. IARC Sci Publ: 257–262, 1992.
Odland, J. O., Nieboer, E., Romanova, N., Thomassen, Y., and Lund, E. Blood lead and cadmium and birth weight among sub-arctic and arctic populations of Norway and Russia. Acta Obstet Gynecol Scand, 78: 852–860, 1999.
Baranowska, I. Lead and cadmium in human placentas and maternal and neonatal blood (in a heavily polluted area) measured by graphite furnace atomic absorption spectrometry. Occup Environ Med, 52: 229–232, 1995.
Osman, K., Akesson, A., Berglund, M., Bremme, K., Schutz, A., Ask, K., and Vahter, M. Toxic and essential elements in placentas of Swedish women. Clin Biochem, 33: 131–138, 2000.
Baghurst, P. A., Robertson, E. F., Oldfield, R. K., King, B. M., McMichael, A. J., Vimpani, G. V., and Wigg, N. R. Lead in the placenta, membranes, and umbilical cord in relation to pregnancy outcome in a lead-smelter community. Environ Health Perspect, 90: 315–320, 1991.
Semczuk, M. and Semczuk-Sikora, A. New data on toxic metal intoxication (Cd, Pb, and Hg in particular) and Mg status during pregnancy. Med Sci Monit, 7: 332–340, 2001.
Wright, R. O., Amarasiriwardena, C., Woolf, A. D., Jim, R., and Bellinger, D. C. Neuropsychological correlates of hair arsenic, manganese, and cadmium levels in school-age children residing near a hazardous waste site. Neurotoxicology, 27: 210–216, 2006.
Wasserman, G. A., Liu, X., Popovac, D., Factor-Litvak, P., Kline, J., Waternaux, C., LoIacono, N., and Graziano, J. H. The Yugoslavia Prospective Lead Study: contributions of prenatal and postnatal lead exposure to early intelligence. Neurotoxicol Teratol, 22: 811–818, 2000.
Ris, M. D., Dietrich, K. N., Succop, P. A., Berger, O. G., and Bornschein, R. L. Early exposure to lead and neuropsychological outcome in adolescence. J Int Neuropsychol Soc, 10: 261–270, 2004.
Needleman, H. L., Leviton, A., and Bellinger, D. Lead-associated intellectual deficit. N Engl J Med, 306: 367, 1982.
Faust, D. and Brown, J. Moderately elevated blood lead levels: effects on neuropsychologic functioning in children. Pediatrics, 80: 623–629, 1987.
Acknowledgements
Support by the International Atomic Energy Agency for this project is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tian, LL., Zhao, YC., Wang, XC. et al. Effects of Gestational Cadmium Exposure on Pregnancy Outcome and Development in the Offspring at Age 4.5 Years. Biol Trace Elem Res 132, 51–59 (2009). https://doi.org/10.1007/s12011-009-8391-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-009-8391-0