Abstract
Effect of exogenous selenium at a dose of 10 μg/kg body weight on the testicular toxicity induced by nicotine in rats was investigated. Male albino rats were maintained for 60 days as follows: (1) control group (normal diet), (2) nicotine group (0.6 mg /kg body weight), (3) selenium (10 μg/kg body weight), and (4) nicotine (0.6 mg/kg body weight) + selenium (10 μg/ kg body weight). Administration of nicotine caused reduction in sperm count and sperm motility. Activity of HMG CoA reductase and concentration of cholesterol were increased in the testes of the nicotine administered group. Activities of testicular enzymes 3β hydroxysteroid dehyrogenase (3βHSD), 17β hydroxysteroid dehyrogenase (17βHSD) were decreased. Levels of testosterone in the serum were also reduced. However, the extent of these alterations was lesser in the group administered with nicotine along with selenium. Analysis of plasma revealed reduced quantity of cotinine in the group co-administered with nicotine along with selenium in comparison with the nicotine group. Nondetectable levels of nicotine were present in the co-administered group. This indicates altered metabolism of nicotine when administered along with selenium.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Nicotine is the most toxic component in the tobacco smoke [1]. In men, nicotine has been linked with decreased sperm density, a lower proportion of motile sperm decreased total sperm count, reduced testosterone secretion [2], and an increase in morphologically abnormal spermatids and spermatozoa, which can be a direct cause of various fetal malformations [3]. Nutritional studies indicate that selenium (Se) is essential for male fertility [4]. The best characterized effects of Se deficiency include loss of sperm motility, increased incidence of sperm abnormalities, and decreased reproductive ability [5]. The present study deals with the effect of exogenous selenium supplementation on testicular toxicity induced by nicotine in rats.
Materials and Methods
Experiment I
A dose response study was conducted to find out the most effective dose of selenium to combat nicotine-induced testicular toxicity induced by the administration of nicotine. Male albino rats (Sprague–Dawley; age 2 1/2 months), weighing 90–110 g, bred in our animal house were divided randomly into six groups of six rats each. Animals were housed in polypropylene cages and were maintained in a temperature-controlled environment under 12 h of light/dark cycle. Rats were fed with rat feedFootnote 1(Lipton India). Food and water was given ad libitum. Animals were handled using the laboratory animal welfare guidelines [6]. The rats were administered nicotine and selenium as follows:
-
1.
Group I: Control
-
2.
Group II: Nicotine (0.6 mg nicotine/kg body weight)
-
3.
Group III: Sodium selenite + nicotine (1 μg sodium selenite/100 g body weight + 0.6 mg nicotine/kg body weight)
-
4.
Group IV: Sodium selenite + nicotine (15 μg sodium selenite/100 g body weight + 0.6 mg nicotine/kg body weight)
-
5.
Group V: Sodium selenite + nicotine (20 μg sodium selenite/100 g body weight + 0.6 mg nicotine/kg body weight) and
-
6.
Group VI: Sodium selenite + nicotine (40 μg sodium selenite/100 g body weight + 0.6 mg nicotine/kg body weight).
Control rats were administered with physiological saline solution equal to the volume of nicotine solution. Duration of the experiment was 60 days.
Experiment II
Male albino rats (Sprague–Dawley; age 2 1/2 months) weighing average 90–110 g, bred in our animal house were divided randomly into four groups of six rats each. The rats were administered with nicotine and selenium as follows:
-
1.
Control
-
2.
Nicotine 0.6 mg nicotine/kg body weight
-
3.
Selenium 10 μg sodium selenite/kg body weight
-
4.
Nicotine + Selenium 0.6 mg nicotine/kg body weight + 10 μg sodium selenite/kg body weight
The dose of nicotine was selected according to studies conducted in our department [7]. Dose of selenium was selected by conducting a dose response study in which 10 μg sodium selenite/kg body weight was found to be the optimum dose with maximum protective effect. Rats were fed with rat feed *(Lipton India Ltd). Food and water was given ad libitum. Sodium selenite and nicotine were administered as detailed above. Sodium selenite was freshly dissolved in distilled water. Nicotine 99% (Sigma-Aldrich Pvt.Lt.) was dissolved in physiological saline and pH adjusted to 7.2 with sterile 0.1 N HCl. Both were given orally by gastric tube for 60 days. Control rats received the same volume of saline. On the 61st day, animals were allowed to fast for overnight and were given intraperitoneal injections of sodium pentothal and subjected to euthanasia. The weight of the body and testis were also determined. Tissues were collected in ice -cold containers for various biochemical estimations.
Biochemical Analysis
Sperm parameters, such as motility, and count were determined by the methods given in World Health Organization (WHO) Manual [8]. The tissues were extracted for lipids according to the procedure of Folch et al. [9]. The total cholesterol was estimated by the method of Abell et al. [10]. HMG CoA reductase activity in the testes was estimated as described by Rao and Ramakrishnan [11] by determining the ratio of HMG CoA/mevalonic acid.
Tissues for estimations of 3β hydroxysteroid dehyrogenase (3βHSD) and 17β hydroxysteroid dehyrogenase (17βHSD) were homogenized in 0.1 M Tris HCl. The assay of 3βHSD was done as by the colorimetric assay of Shivanandappa and Venkatesh [12]. 17βHSD was assayed by the method described by Jarabak [13]. Protein in the enzyme extract was determined after trichloroacetic acid (TCA) precipitation by the method of Lowry et al. [14].The serum testosterone was estimated by RIA (RIA kit was supplied by BRIT India).The procedure described by Hariharan and VanNoord [15] was used for the extraction of alkaloids in the plasma. The nicotine and cotinine content in the plasma were estimated by high performance liquid chromatography (HPLC) [Shimadzu SPD-10A] [HPLC column reverse phased C-18 ion pair column, flow rate 1.5 ml/min].The solvent system or the mobile phase was a mixture of citrate and dibasic phosphate (30 mmol/l) containing 1 mmol of sodium heptanesulfonate and 50 ml of acetonitrile/l (pH 6.1).
Statistical Analysis
The results were analyzed using a statistical program SPSS/PC+, version 5.0 (SPSS, Chicago, IL, USA). A one-way analysis of variance (ANOVA) was employed for comparison among the six groups. Duncan’s post-hoc multiple comparison tests of significant differences among groups were determined. A p ≤ 0.05 was considered to be significant.
Results
Experiment I
Motility of spermatozoa (Table 1), removed from the cauda epididymides of the rats treated with nicotine, was depressed when compared with control animals. The sperm motility decreased as the concentration of selenium increased on co-administration along with nicotine. Maximum motility was seen in the group co administered with a dose of 1 μg selenium along with nicotine.
Experiment II
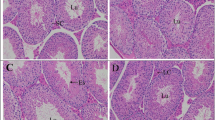
Body weight and testis weight (Table 2) of the rats were decreased in the nicotine administered group, when compared to the control. On Se supplementation and co-administration of nicotine along with selenium, the weight was increased in comparison to the nicotine group.
Sperm count (Table 2) decreased significantly in the nicotine-treated group as compared to the control and selenium groups. On co-administration of nicotine along with selenium, the counts were increased as compared to the nicotine group. The sperm motility in the nicotine group was drastically reduced in comparison with the selenium and control groups. However, co-administration of selenium and nicotine enhanced the percentage of motility.
The HMG Co A/mevalonate ratio was measured to assay the activity of HMG CoA reductase activity. Lower ratio indicates higher enzyme activity. HMG CoA reductase activity (Table 3) increased significantly in the testes of the nicotine-treated group when compared to the control group. Whereas on co-administration of selenium along with nicotine showed a significant decrease in the concentration of cholesterol.
in the testes was significantly increased in the nicotine group compared to the control. Selenium supplemented group showed no significant change as compared to the control. Co-administration of nicotine along with selenium decreased the activity of the enzyme as compared to the nicotine. The concentration of total cholesterol (Table 3) increased significantly in the testes of the nicotine-treated group when compared to the control group. Whereas on co-administration of selenium along with nicotine showed a significant decrease in the concentration of cholesterol.
The activities of 3βHSD and 17βHSD (Table 3) in the testes decreased significantly in the nicotine-treated group compared to the control. However, the group co-administered nicotine along with selenium showed a significant increase in the activities of these enzymes when compared to the nicotine-treated group. The selenium-treated group showed no significant change as compared to control. Testosterone concentration (Table 3) in the serum was significantly decreased in the nicotine-treated group. However, the group given with nicotine along with selenium had an increased concentration of testosterone when compared to the nicotine-treated group. The selenium-treated group showed an increase in the testosterone level when compared to that of nicotine. A high concentration of nicotine (Table 3) was detected in the nicotine-treated group. However, nicotine was not detected in the group co-administered with nicotine along with selenium. Concentration of cotinine, which is a metabolite of nicotine, increased significantly after nicotine administration. However, on co-administration of nicotine along with selenium, there was a reduction in its level in comparison with nicotine group.
Discussion
The dose response study revealed the optimum dose of selenium that had maximum beneficial effects on the rat testes to be 10 μg Se/kg body weight. Our earlier studies had also shown that 10 μg Se/kg body weight reduced ethanol-induced testicular toxicity in rats [16]. The body weight and testes weight of rats decreased in the nicotine treated group. This is in accordance with the reports that cigarette smoking results in weight loss, and the smokers gain weight, when they quit smoking [17]. However, administration of nicotine and selenium together reversed these changes. In agreement with the reports that cigarette smoking is associated with decreased sperm count and alteration in sperm motility [3], we also observed reduction in sperm count and motility. The co-administered group showed increased sperm count and motility. In this connection, it is important to note that Se is a key component of a protein present in the sperm, which has a role in maintaining normal fertility. Kaur and Bansal [18] observed reduction in the reproductive ability of selenium-deficient mice. Selenium plays a major role in the spermatogenesis especially through the selenoenzyme phospholipid glutathione peroxidase (PHGPx, GPx-4) [19].
The elevated levels of cholesterol in the nicotine group may be due to the enhanced biosynthesis, as the activity of HMG CoA reductase had been increased. This is in agreement with the earlier reports [7]. Whereas co-administration of nicotine and selenium showed decreased cholesterol concentration and lowered HMG CoA reductase activity. This may account for the hypolipidemic effect of selenium [20–22] Cholesterol is the substrate for testosterone biosynthesis. Although the nicotine-treated group shows increased concentration of cholesterol, it is not utilized for the production of testosterone, as the testosterone level in the nicotine-treated group significantly decreased when compared to the control group. This is supported by the decreased activities of the two main enzymes of the testosterone biosynthetic pathway-17β hydroxy steroid dehydrogenase and 3β hydroxy steroid dehydrogenase. This inhibition of testosterone biosynthesis supports the observation of other workers [7, 23]. However, this was reversed by the supplementation of selenium along with nicotine proving its protective effects on reproductive performance.
Cotinine, the major metabolite of nicotine, was detected in very low amounts in the plasma of the group-administered selenium along with nicotine, whereas nicotine was not detected in the plasma of this group. This may be due to the enhanced metabolism of nicotine in the presence of selenium. Dawson et al. [24] reported that the supplementation of the antioxidant lowers the urinary excretion of cotinine. Hence, from the above studies, it is suggested that exogenous selenium supplementation has protective effects on the testicular toxicity induced by nicotine, as selenium increases testosterone synthesis and enhances nicotine degradation.
Notes
Composition of the rat feed: The selenium content of the feed was 0.0035 μg/g, crude protein 24%, ether extract 1–3.5%, crude fiber 12%, ash 8%, calcium 1.2%, phosphorus0.06%, and nitrogen free extract 43%.
References
Barnes B, Bradley SG (1990) Planning for a healthy baby. Ebury Press, London, p 96
Briggs MH (1973) Cigarette smoking and infertility in men. Med J Aust 12:616
Evans HJ, Fletcher J, Torrance M, Hardgreave TB (1981) Sperm abnormalities and cigarette smoking. Lancet 1:627
Behne D, Weiler H, Kyriakopoulos A (1996) Effects of selenium deficiency on testicular morphology and function in rats. J Reprod Fertil 106:291
Calvin HI (1978) Selective incorporation of selenium75 into a polypeptide on the rat sperm tail. J Exp Zool 204:445
Hume CW (1972) The UFAW Handbook on the care and management of laboratory animals. Churchill Livingstone, Edinburg
Kavitharaj NK, Vijayammal PL (1999) Nicotine administration induced changes in the gonadal functions in male rats. Pharmacol 58:2
WHO laboratory manual for the examination of human semen and sperm-cervical mucus interaction, 4th ed. Pub. On behalf of WHO. Cambridge University Press, Cambridge
Folch J, Less M, Stanley GHS (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497
Abell LL, Levy BB, Brodie BB, Kendall FE (1952) A simplified method for the estimation of total cholesterol in serum and demonstration of its specificity. J Biol Chem 195:357
Rao V, Ramakrishnan S (1975) Indirect assessment of hydroxy methyl glutaryl CoA reductase activity in liver tissue. J Clin Chem 21:1523
Shivanandappa T, Venkatesh S (1997) A colorimetric assay method for 3β-hydroxy. 5. -steroid dehydrogenase. Anal Biochem 254:57
Jarabak J (1969) Soluble 17β-hydroxysteroid dehydrogenase of human placenta. In: Clayton RB (ed) Methods in enzymol, vol 15. Academic, New York, p 746
Lowry OH, Rosebrough NJ, Farr AL, Randekk RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265
Hariharan M, VanNoord T (1988) A high-performance liquid-chromatographic method for routine simultaneous determination of nicotine and cotinine in plasma. Clin Chem 34(4):724
Swathy SS, Panicker S, Indira M (2006) Effect of exogenous selenium on the testicular toxicity induced by ethanol in rats. Indian J Physiol Pharmacol 50:215–224
Williamson DF, Madans J, Anda RF et al (1991) Smoking cessation and severity of weight gain in a national cohort. N Engl J Med 324:739
Kaur P, Bansal MP (2005) Effect of selenium-induced oxidative stress on the cell kinetics in testis and reproductive ability of male mice. Nutrition 21:351–357
Shalini S, Bansal MP (2005) Role of selenium in regulation of spermatogenesis: involvement of activator protein 1. Biofactors 23:151–162
Dhingra S, Bansal MP (2006) Hypercholesterolemia and LDL receptor mRNA expression: modulation by selenium supplementation. Biometals 19:493–501
Asha GS, Indira M (2004) Combined effect of selenium and ascorbic acid on alcohol induced hyperlipidemia in male guinea pigs. Comp Biochem Physiol C Toxicol Pharmacol 137:109–114
Nassir F, Moundras C, Bayle D et al (1997) Effect of selenium deficiency on hepatic lipid and lipoprotein metabolism in the rat. Br J Nutr 78(3):493
Yeh J, Barbieri RL, Friedman AJ (1989) Nicotine and cotinine inhibit rat testis androgen biosynthesis in vitro. J Steroid Biochem 33(4A):627
Dawson EB, Evans DR, Harris WA, McGanity WJ (1999) The effect of ascorbic acid supplementation on the nicotine metabolism of smokers. Prev Med 29:451
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seema, P., Swathy, S.S. & Indira, M. Protective Effect of Selenium on Nicotine-Induced Testicular Toxicity in Rats. Biol Trace Elem Res 120, 212–218 (2007). https://doi.org/10.1007/s12011-007-8021-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-007-8021-7