Abstract
A gene of glucose oxidase (GOD) from Aspergillus niger Z-25 was cloned and sequenced. The entire open reading frame (ORF) consisted of 1,818 bp and encoded a putative peptide of 605 amino acids. The gene was fused to the pPICZαA plasmid and overexpressed in Pichia pastoris SMD1168. The recombinant GOD (rGOD) was secreted into the culture using MF-α factor signal peptide under the control of the AOX1 promoter. Sodium dodecyl sulfate polyacrylamide gel electrophoresis indicated that rGOD exhibited a single band at around 94 kDa. The maximal GOD activity of approximately 40 U/mL was achieved in shake flask by induction under optimal conditions after 7 days. rGOD was purified by ammonium sulfate precipitate leading to a final specific activity of 153.46 U/mg. The optimum temperature and pH of the purified enzyme were 40 °C and 6.0, respectively. Over 88% of maximum activity was maintained below 40 °C. And the recombinant enzyme displayed a favorable stability in the pH range from 4.0 to 8.0. The Lineweaver–Burk plotting revealed that rGOD exhibited a K m value of 16.95 mM and a K cat value of 484.26 s−1.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Glucose oxidase (GOD; β-d-glucose: oxygen 1-oxidoreductase, EC 1.1.3.4), a glycoprotein, catalyzes the oxidation of β-d-glucose to d-glucono-δ-lactone and hydrogen peroxide. Thus, the enzyme is widely used in the fields of food engineering, biotechnology, and medicine like as a source of hydrogen peroxidase in food preservation, as a means for gluconic acid production, and tested as the basis for glucose sensors and glucose detection kits [1, 2].
At present, Aspergillus niger and Penicillum amagasakiense are mostly utilized for industrial-scale fermentation of the enzyme. However, the production of GOD by the two species is difficult in the commercial process due to either low yield or concomitant production of other enzymes such as cellulase, catalase, and amylase [3]. Although several heterologous expression systems such as Escherichia coli [4], Hansenula polymorpha [5], Saccharomyces cerevisiae [6–8], and Pichia pastoris [9, 10] have been investigated for GOD production to solve those problems, other drawbacks still emerged. The recombinant GOD (rGOD) expressed in E. coli exhibited low activity and stability since it was accumulated as inclusion bodies and nonglycosylated form. And in the latter cases, distinct glycosylated patterns of the interest enzymes might diminish their usage such as in the pharmaceutical industry [11]. For the last several decades, the methylotrophic yeast P. pastoris has been developed as a successful system for heterologous proteins expression owing to simple genetic manipulation techniques, high expression levels, efficient secretion, and proper protein modification [12]. Two GOD genes from Penicillium variabile [9] and A. niger [10] were transformed into P. pastoris for secretory and constitutive expression, respectively, but both of the rGOD yields were achieved at a rather low level.
In this paper, we described heterologous overexpression in P. pastoris of the GOD gene cloning from A. niger Z-25, aiming to produce a higher amount of the interesting enzyme. The purification and characterization of the rGOD were also studied.
Materials and Methods
Strain and Plasmid
The GOD gene was cloned from A. niger Z-25, which was isolated and identified by our laboratory [13]. E. coli DH5α was utilized for the maintenance and manipulation of plasmids. P. pastoris SMD1168 (Invitrogen, USA) was utilized as a host for the secretion of GOD. The vector pMD19-T (Takara Biotechnology, Dalian Branch) was used for cloning of the GOD gene. The vector pPICZαA (Invitrogen, USA) was used for the expression of GOD in P. pastoris.
DNA Isolation and Cloning of the GOD Gene from Aspergillus niger
The chromosomal DNA was isolated from A. niger Z-25 as follows. The A. niger mycelia cultured for 24 h at 28 °C in PDA medium (20% potato extract and 2% sucrose) were harvested by sucking filtration, washed with distilled water, frozen in liquid nitrogen, and immediately disrupted in a chilled mortar. Subsequently, about 0.1 g of disrupted mycelia were suspended in 500 µL CTAB extraction buffer (100 mM Tris–HCl at pH 7.5, 700 mM NaCl, 20 mM EDTA, 1% dithiothreitol [DTT], and 1% CTAB). After incubation for 30 min at 65 °C, the mixture was centrifuged. DNA was then extracted with 500 µL phenol/chloroform (1:1), purified by ethanol perception, and eventually dissolved in 50 µL sterilized double-distilled water.
The open reading frame (ORF) encoding the GOD gene was amplified by the polymerase chain reaction (PCR) technique with Taq DNA Polymerase (Bipec, USA) using the following primers: GOD-F (5′-ATGCAGACTCTCCTTGTGAG-3′) and GOD-R (5′-TCACTGCATGGAAGCATAAT-3′). The PCR procedure was performed through 30 cycles of 50 s denaturation at 94 °C, 50 s annealing at 50 °C, 2 min extension at 72 °C, followed by a final extension for 10 min at 72 °C. The PCR product was recovered and purified from a 1.0% agarose gel using a DNA Gel Extraction kit (Sangon, Shanghai) according to the manufacturer's protocols, then subcloned into pMD19-T vector. The resulting plasmid was used for sequence analysis.
Construction of the Expression Plasmid
For the expression of mature protein with a native N terminus, the entire ORF of the GOD gene without native signal peptide sequence was amplified with the specific primers. The forward primer was GOD-F1: 5′-CCCTCGAG AAAAGAAGCAATGGCATCGAAGC-3′ (the italicized sequence indicates the restriction site XhoI, letters in bold indicated Kex2 signal cleavage). The reverse primer was GOD-R2: 5′-TTGCGGCCGCTCACTGCATGGAAGCATAAT-3′ (the italicized sequence indicates the restriction site NotI). DNA amplification was carried out through 30 cycles of 50 s denaturation at 94 °C, 50 s annealing at 62 °C, 2 min extension at 72 °C, followed by a final extension for 10 min at 72 °C. The purified PCR product was digested with XhoI and NotI and ligated at the XhoI–NotI restriction sites of the pPICZαA vector previously treated with the same enzymes, consequently creating the expression plasmid named pPICZαA-GOD. The sequence of the resulting plasmid was verified.
Transformation of P. pastoris
Transformation was performed utilizing the method reported by Wu and Letchworth [14]. The recombinant plasmid pPICZαA-GOD was linearized with PmeI (MBI). Competent P. pastoris SMD1168 cells were prepared by pretreating P. pastoris with lithium acetate (LiAc) and DTT for electroporation. One microgram of the linearized recombinant plasmid and 80 µL of fresh competent P. pastoris cells were mixed well, transferred into an 0.2-cm electroporation cuvette, and electroporated using a Bio-Rad MicroPulser apparatus (Bio-Rad, USA) working at 2.0 kV and 5.8 ms. The transformants were selected at 30 °C on YPDS (1% yeast extract, 2% peptone, 2% glucose, 1 M sorbitol, and 2% agar) plates containing 100 µg/mL zeocin (Invitrogen, USA) for 3–4 days. As a control, the empty vector pPICZαA was also transformed into P. pastoris SMD1168.
Screening of High-Level Expression Transformants
P. pastoris transformants were patched on YPDS plates containing 500, 1,000, and 2,000 µg/mL zeocin. Multicopy transformants which were supposed to highly express GOD were screened from the YPDS plates with high zeocin concentration (2,000 µg/mL) and analyzed by PCR with 5′ AOX1 and GOD-R2 primers. The PCR reaction was carried out through 30 cycles of 50 s denaturation at 94 °C, 50 s annealing at 55 °C, 2.5 min extension at 72 °C, followed by a final extension for 10 min at 72 °C. Positive transformants were inoculated into 5 mL BMGY medium (1% yeast extract, 2% peptone, 1.34% yeast nitrogen base [YNB], 4 × 10−5% biotin, 1% glycerol in 100 mM potassium phosphate, pH 6.0) in 50 mL shake flask (250 rpm) at 30 °C. After incubation for 24 h, the cells were harvested by centrifugation and resuspended in 25 mL BMMY medium (1% yeast extract, 2% peptone, 1.34% YNB, 4 × 10−5% biotin, 0.5% methanol [v/v] in 100 mM potassium phosphate, pH 6.0) in 100 mL shake flask (250 rpm) at 30 °C for 72 h. To maintain induction, 100% methanol was added to a final concentration of 0.5% every 24 h during the incubation phase, while the culture supernatant was obtained by centrifugation daily and used to assay for the GOD activity. The recombinant strain (SMD1168/pPICZαA) generated by transforming the empty vector pPICZαA into P. pastoris SMD1168 was also incubated under the same conditions as a control for GOD assay. The recombinant strain with the highest level of GOD (SMD1168/GOD) was screened and utilized for further experiments.
Optimization of Expression Conditions in Shake Flask
To improve GOD production, four parameters were examined in shake flask cultures, including methanol concentration, initial pH value, temperature, and rotary speed. The recombinant strain SMD1168/GOD was inoculated into 10 mL YPG (1% glycerol, 1% yeast extract, and 2% peptone) medium and incubated in a rotary shaker overnight at 30 °C until the cells were in logarithmic phase growth. The inoculum was harvested by centrifugation and resuspended by 50 mL BMMY medium in 250 mL shake flask. Methanol was added by various concentrations from 0.5% to 4.0% daily to screen the optimum concentration. Initial pH values (4.0–8.0) in BMMY medium were tested. To determine the most suitable temperature, induction was performed at 15, 20, 25, and 30 °C, respectively. The influence of dissolved oxygen was investigated with different rotary speeds (160, 200, 240, and 280 rpm). The GOD activity and cell density for each triplicate of shake flask cultures were detected after an induction period of 72 h. The profiles for cell growth and GOD production were studied under the optimal expression conditions for 7 days eventually.
Enzyme Assay and SDS-PAGE Analysis
The assay for GOD activity was carried out following the modified protocol of Sigma; 2.4 mL of 0.21 mM o-dianisidine (Sigma, USA) solution (dissolved in 0.1 M sodium phosphate buffer, pH 6.0), 0.5 mL of 10% (w/w) β-d-glucose solution (prepared in 0.1 M sodium phosphate buffer, pH 6.0), and 100 µL of 60 U/mL horseradish peroxidase solution were mixed well in a cuvette. After incubation at 35 °C, 100 µL of sample solution was added and immediately mixed by inversion. The increase in absorbance at 500 nm (ε = 7.5 mM−1 cm−1) was measured every 30 s using a spectrophotometer (Shimadazu, Japan) for 5 min continuously. The definition of 1 U of GOD activity per volume of enzyme solution (in milliliters) was that this activity oxidized 1 µmol of β-d-glucose to d-glucono-δ-lactone and H2O2 per minute under the assay condition.
To estimate homogeneity and molecular mass, sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) was performed with an 8% polyacrylamide gel. The protein bands were stained with Coomassie Brilliant Blue R-250. Protein molecular weight marker (Takara Biotechnology, Dalian Branch) was used.
Purification of Recombinant GOD and Protein Concentration Measurement
The culture supernatant was attained by centrifugation (5,000×g for 10 min) and then treated with ammonium sulfate precipitation. The precipitate salted out by 60–90% saturated ammonium sulfate was collected by centrifugation (1,000×g for 10 min), dissolved in 20 mM Tris–HCl buffer (pH 7.0), and dialyzed at 4 °C overnight against the same buffer. Subsequently, the resulting solution was concentrated by polyethylene glycol 20000 and used as purified enzyme.
The protein concentration was determined by the method of Bradford [15] with bovine serum albumin (TianWei Biotechnology, Beijing) as the standard.
Biochemical Characterizations of Recombinant GOD
The optimum temperature of rGOD was determined by performing the standard assay as described in the “Enzyme Assay and SDS-PAGE Analysis” section at 10° intervals between 20 and 80 °C. To estimate thermal stability, the enzyme was preincubated for 1 h at different temperatures and then assayed for residual activity using the standard method.
The optimum pH was determined by measuring the enzyme activity at the pH range of 2.0–10.0. The buffers (0.1 M) included HCl–KCl (pH 2.0), sodium citrate (pH 3.0–5.0), sodium phosphate (pH 6.0–8.0), and glycine–NaOH (pH 9.0–10.0). Substrate solution was prepared in the respective buffers. To investigate pH stability, the enzyme was preincubated in different buffers for 12 h at 25 °C and then assayed for residual activity as described above.
To study enzyme kinetics, the rGOD activity was assayed at 35 °C in 0.1 M sodium phosphate (pH 6.0) with β-d-glucose (0–200 mM) as substrate. The K m and K cat values for rGOD were determined by Lineweaver–Burk plotting.
Results
Cloning and Sequence Analysis of GOD Gene from Aspergillus niger Z-25
The entire ORF of the GOD gene consisted of 1,818 bp, encoding 605 amino acid residues (GenBank accession number FJ979866). No introns were present in the GOD sequence similar to those found in another two strains, A. niger NRRL-3 [16] and A. niger ATCC 9029 [6], respectively. The GOD nucleotide sequence from A. niger Z-25 exhibited 93% identity to those from the two strains (GenBank accession numbers X16061 and J05242), while comparison of the A. niger Z-25 amino acid sequence with its counterparts showed an identity degree of 97% with 17 amino acids replaced (Fig. 1). Similar to those from the two strains, the deduced amino acid sequence of GOD from A. niger Z-25 contained a total of three cysteine residues at conserved position and a signal peptide of 22 amino acids, the latter contributing to a mature peptide with a calculated molecular weight of 64 kDa. Nevertheless, it showed seven possible putative N-glycosylation sites conforming to the consensus sequence Asn-X-Thr/Ser, whereas eight potential N-glycosylation sites were identified by Frederick et al. [6] and six were indicated by Hecht et al. [17].
Heterologous Expression of the GOD Gene in P. pastoris SMD1168
The GOD gene without its native signal sequence was inserted into the vector pPICZαA immediately after the Kex2 cleavage site and the construct was correctly fused to the α-factor signal. About 80 transformants were generated on YPDS plate with 100 µg/mL zeocin, and six transformants were selected for high resistance to zeocin (2,000 µg/mL). PCR analysis was performed to determinate whether the GOD gene was integrated into the P. pastoris genome at the AOX1 locus. The resulting PCR product lengths of 2,100 bp (300 + 1,800) revealed that the GOD gene was correctly inserted into the yeast genome (Fig. 2). The P. pastoris recombinant strain (SMD1168/GOD) was ultimately screened for the sake of yielding the highest GOD activity (4.10 U/mL) in shake flask after methanol induction of 72 h. The culture of control strain SMD1168/pPICZαA had no detectable GOD activity.
Optimization of Expression Conditions in Shake Flask
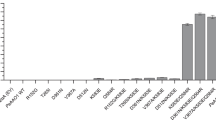
The influence of cultivation conditions on GOD production was investigated in shake flask. Amid the investigated parameters, methanol concentration was the most significant. After 72 h, the GOD activity induced by 2.0% methanol was roughly 1.4 times of that induced by 0.5% methanol. On the other hand, initial pH values resulted in less effect than methanol concentration. The levels of both cell growth and GOD yield were a bit higher at pH 5.0 than at pH 6.0. The recombinant P. pastoris strain propagated as effectively at 15 °C as at 30 °C. In contrast, the production declined seriously with the depressed temperature. And the highest GOD activity was attained at 240 rpm though the maximal OD600 was yielded at 280 rpm.
The profiles of cell growth and GOD production were studied under the optimal conditions (addition of 2.0% methanol daily, an initial pH value of 5.0 in BMMY medium, an induction temperature of 30 °C, and incubation at 240 rpm) in shake flask (Fig. 3). It was indicated that the GOD activity rose steadily as the induction went on. The maximal GOD activity of 39.37 U/mL was achieved after induction of 168 h, which was 20-fold higher than that (1.92 U/mL) obtained from A. niger Z-25.
Purification of Recombinant GOD from P. pastoris
rGOD was purified from P. pastoris culture supernatant by one-step procedure (Table 1). After ammonium sulfate precipitate, 153.46 U/mg of rGOD was pooled from the culture supernatant after 3.34-fold purification with a final yield of 95.6%. SDS-PAGE of the purified enzyme showed that rGOD exhibited a single band with a molecular weight of 94 kDa (Fig. 4a). We predicted the mature peptide of GOD with a molecular mass of 64 kDa and seven possible putative N-glycosylation sites at conserved positions as described in the “Cloning and Sequence Analysis of GOD Gene from Aspergillus niger Z-25” section. Figure 4 shows the subunit molecular weight of the purified GODs from P. pastoris (94 kDa) and A. niger (76 kDa). Consequently, the carbohydrate content was confirmed to be 31.9% for P. pastoris and 15.8 % for A. niger.
Properties of Recombinant GOD
The effects of temperature on rGOD and the thermal stability of the recombinant enzyme are present in Fig. 5a. rGOD had an optimum reaction temperature at 40 °C. Interestingly, the activity of rGOD increased slightly at the reaction temperature ranging from 20 to 40 °C, and then fall moderately till 60 °C. The recombinant enzyme retained more than 90% of activity within 40 °C. Over 30% of activity was lost at 50 °C, whereas almost no activity was detected above 60 °C.
Figure 5b demonstrates the effects of pH on rGOD and the pH stability of the recombinant enzyme. The activity of rGOD peaked at pH 6.0. More than 88% of the maximum activity was observed between pH 4.0 and 8.0. Outside this range, the activity decreased dramatically.
The K m and K cat values for rGOD were 16.95 mM and 484.26 s−1, respectively.
Discussion
Protease-deficient cells like SMD1168 (his4, pep4) are known to possess a lower viability and are more difficult to transform compared with the wild-type strains [12]. Therefore, an efficient transformation was fulfilled by electroporation of P. pastoris SMD1168 pretreated with LiAc and DTT [14], creating sufficient transformants for selection. The GOD activity of 4.1 U/mL was achieved with induction by methanol in shake flask for 3 days with unoptimized conditions, which was a higher yield compared with previous reports on heterologous expression of two GOD genes from P. variable (0.33 U/mL, 11 days) [9] and A. niger (1.23 U/mL, 2 weeks) [10]. Furthermore, about 40 U/mL of GOD activity was yielded by shake flask culture under optimal conditions for 7 days. Usually, a tenfold increase in the yield of recombinant protein has been observed between shake flask and high-density fermentation cultures [18]. More fermentation strategies will be investigated with fermenter in subsequent studies to further improve the GOD yield. This would result in a significant influence in food engineering and biotechnology fields.
Methanol is the sole carbon and energy source for cell growth and protein biosynthesis during the induction phase [19]. Thus, the methanol concentration of the medium is a crucial parameter in shake flask cultivation. Low levels of methanol (0.1–1.0%) had been numerously recommended to induce the expression of foreign genes in P. pastoris [20–22]. Nevertheless, our study observed that 2.0% methanol was exactly suitable to induce GOD expression. Khatri and Hoffmann [23] reported that high methanol concentration was required to compensate for the lack of oxygen, probably accounting for the demand of high levels of methanol to induce GOD expression in shake flask where dissolve oxygen was limited.
One-step procedure with ammonium sulfate precipitate was adequate for purification of rGOD. We attributed it to utilizing the host strain SMD1168. The secreted rGOD constituted the majority of total proteins in the culture supernatant, facilitating the process of purification.
Table 2 illustrates the comparison of biochemical characterizations amid recombinant and native GODs from A. niger and Penicillum species. The molecular weight and subunit molecular weight of GODs ranged from about 130 to 180 kDa and 60 to 90 kDa, respectively. And the optimum temperature for GODs varied from 25 to 55 °C. The enzymes obtained from A. niger and P. variabile P16 displayed optimum pH between 5.0 and 6.0, while GODs from P. amagasakiense ATCC 28686 and P. DSM 11428 showed weakly acid pH optima; GODs from P. funiculosum 433 and P. canescens Tt42 had slightly alkaline pH optima. The thermal and pH stability of GODs were reported to be 25–60 °C and 3.0–8.6, respectively. The kinetic parameters of GODs from different sources altered dramatically.
There was no substantial discrepancy of properties among recombinant and native GODs from A. niger Z-25 and GOD from A. niger (Sigma Type VII), except that native enzyme from A. niger Z-25 had a much narrower pH range of stability and the one from Sigma displayed a relatively higher optimum temperature. It was noteworthy that recombinant and native GODs from A. niger Z-25 had different K m and K cat values from those of the enzyme from Sigma. Though a lower K m value contributed to a higher affinity for β-d-glucose of this rGOD, yet a lower K cat value accounted for a slower catalysis velocity of the recombinant enzyme.
References
Banks, J. G., Board, R. G., & Sparks, N. H. C. (1986). Natural antimicrobial systems and their potential in food preservation of the future. Biotechnology and Applied Biochemistry, 8, 103–147.
Richter, G. (1983). Glucose oxidase. In T. Godfrey & J. Reichelt (Eds.), Industrial enzymology: the application of enzymes in industry (pp. 428–436). New York: MacMillan.
Kapat, A., Jung, J., & Park, Y. (1998). Improvement of extracellular recombinant glucose oxidase production in fed-batch culture of Saccharomyces cerevisiae: Effect of different feeding strategies. Biotechnology Letters, 20, 319–323.
Witt, S., Singh, M., & Kalisz, H. M. (1998). Structural and kinetic properties of nonglycosylated recombinant Penicillium amagasakiense glucose oxidase expressed in Escherichia coli. Applied and Environmental Microbiology, 64, 1405–1411.
Hodgkins, M., Mead, D., Balance, D. J., Goodey, A., & Sudbery, P. (1993). Expression of the glucose oxidase gene from Aspergillus niger in Hansenula polymorpha and its use as a reporter gene to isolate regulatory mutations. Yeast, 9, 625–635.
Frederick, K. R., Tung, J., Emerick, R. S., Masiarz, F. R., Chamberlain, S. H., Vasavada, A., et al. (1990). Glucose oxidase from Aspergillus niger. Journal of Biological Chemistry, 265, 3793–3802.
Whittington, H., Williams, S. K., Bidgood, K., Dodsworth, N., Peberdy, J., Dobson, M., et al. (1990). Expression of the Aspergillus niger glucose oxidase gene in A. niger, A. nidulans and Saccharomyces cerevisiae. Current Genetics, 18, 531–536.
Park, E. H., Shin, Y. M., Lim, Y. Y., Kwon, T. H., Kim, D. H., & Yang, M. S. (2000). Expression of glucose oxidase by using recombinant yeast. Journal of Biotechnology, 81, 35–44.
Crognale, S., Pulci, V., Brozzoli, V., Petruccioli, M., & Federici, F. (2006). Expression of Penicillium variabile P16 glucose oxidase gene in Pichia pastoris and characterization of the recombinant enzyme. Enzyme and Microbial Technology, 39, 1230–1235.
Yamaguchi, M., Tahara, Y., Nakano, A., & Taniyama, T. (2007). Secretory and continuous expression of Aspergillus niger glucose oxidase gene in Pichia pastoris. Protein Expression and Purification, 55, 273–278.
Romanos, M. A., Scorer, C. A., & Clare, J. J. (1992). Foreign gene expression in Yeast: A review. Yeast, 8, 423–488.
Cereghino, J. L., & Cregg, J. M. (2000). Heterologous protein expression in the methylotrophic yeast Pichia pastoris. FEMS Microbiology Reviews, 24, 45–66.
Zhang, Y., Lu, Z., & Zou, X. (2005). 15 keV N+ implantation to induce mutation of protopectinase-producing Aspergillus sp. Journal of Radiation Research and Radiation Processing, 33, 140–144. (in Chinese).
Wu, S., & Letchworth, G. J. (2004). High efficiency transformation by electroporation of Pichia pastoris pretreated with lithium acetate and dithiothreitol. BioTechniques, 36, 152–154.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254.
Kriechbaum, M., Heilmann, H. J., Wientjes, F. J., Hahn, M., Jany, K. D., Gassen, H. G., et al. (1989). Cloning and DNA sequence analysis of the glucose oxidase gene from Aspergillus niger NRRL-3. FEBS Letters, 255, 63–66.
Hecht, H. J., Kalisz, H. M., Hendle, J., Schmid, R. D., & Schomburg, D. (1993). Crystal structure of glucose oxidase from Aspergillus niger refined at 2.3 Å resolution. Journal of Molecular Biology, 229, 153–172.
Li, P., Anumanthan, A., Gao, X., Ilangovan, K., Suzara, V., Düzgünes, N., et al. (2007). Expression of recombinant proteins in Pichia pastoris. Applied Biochemistry and Biotechnology, 142, 105–124.
Macauley-Patrick, S., Fazenda, M. L., McNeil, B., & Harvey, L. M. (2005). Heterologous protein production using the Pichia pastoris expression system. Yeast, 22, 249–270.
Guarna, M. M., Lesnicki, G. J., Tam, B. M., Robinson, J., Radziminski, C. Z., Hasenwinkle, D., et al. (1997). On-line monitoring and control of methanol concentration in shake-flask cultures of Pichia pastoris. Biotechnology and Bioengineering, 56, 279–286.
Boettener, M., Prinz, B., Holz, C., Stahl, U., & Lang, C. (2002). High-throughput screening for expression of heterologous proteins in the yeast Pichia pastoris. Journal of Biotechnology, 99, 51–62.
Damasceno, L. M., Pla, I., Chang, H., Cohen, L., Ritter, G., Old, L. J., et al. (2004). An optimized fermentation process for high-level production of a single-chain Fv antibody fragment in Pichia pastoris.Protein Expression and Purification, 37, 18–26.
Khatri, N. K., & Hoffmann, F. (2005). Impact of methanol concentration on secreted protein production in oxygen-limited cultures of recombinant Pichia pastoris. Biotechnology and Bioengineering, 93, 871–879.
Kalisz, H. M., Hecht, H., Schomburg, D., & Schmid, R. D. (1991). Effects of carbohydrate depletion on the structure, stability and activity of glucose oxidase from Aspergillus niger. Biochimica et Biophysica Acta, 1080, 138–142.
Kalisz, H. M., Hendle, J., & Schmid, R. D. (1997). Structural and biochemical properties of glycosylated and deglycosylated glucose oxidase from Penicillium amagasakiense. Applied Microbiology and Biotechnology, 47, 502–507.
Rando, D., Kohring, G.-W., & Giffhorn, F. (1997). Production, purification and characterization of glucose oxidase from a newly isolated strain of Penicillium pinophilum. Applied Microbiology and Biotechnology, 48, 34–40.
Sukhacheva, M. V., Davydova, M. E., & Netrusov, A. I. (2004). Isolation and properties of glucose oxidase from Penicillum funiculosum 433. Applied Biochemistry and Microbiology, 40, 32–36.
Simpson, C. (2005). Isolation, purification and characterization of a novel glucose oxidase from Penicillium canescens. Master's thesis, Rhodes University, Grahamstown, South Africa.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, Y., Lu, F., Zhao, H. et al. Cloning and Heterologous Expression of Glucose Oxidase Gene from Aspergillus niger Z-25 in Pichia pastoris . Appl Biochem Biotechnol 162, 498–509 (2010). https://doi.org/10.1007/s12010-009-8778-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-009-8778-6