Abstract
This paper is concerned with the optimization of effect factors for mycelial growth and exopolysaccharide production by Schizophyllum commune by one-factor-at-a-time and orthogonal methods. The one-factor-at-a-time method was adopted to investigate the effects of six different compounds (sodium carboxymethylcellulose, l-glutamic acid, VB1, naphthalene acetic acid, oleic acid, and Tween 80) on mycelial growth and exopolysaccharide production. Among these factors, oleic acid, VB1 and Tween 80 were identified to be the most important factors. Subsequently, the concentration of oleic acid, VB1 and Tween 80 were optimized using the orthogonal matrix method. The effects of the factors on the mycelial growth of S. commune were in the order of oleic acid > VB1 > Tween 80, and those on exopolysaccharide production were in the same order. The optimal concentration for mycelia and exopolysaccharide were determined as oleic acid 0.1% (v/v), VB1 0.5 mg/L, and Tween 80 6 mg/L. The subsequent verification experiments confirmed the validity of the models. Under this optimized conditions in shake flask culture, the mycelial yield and exo-biopolymer production were 25.93 and 2.79 g/L, respectively, which were considerably higher than those obtained in the preliminary studies. The result was further confirmed in a 7-L fermentor experiments.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Schizophyllum commune (Fr.) [1] is a species of basidiomycetes belonging to Schizophyllaceae of Agaricales. Its fruiting body is called “Baishen, Shuhua, Badanchai” in Chinese. For centuries, this mushroom has been regarded in the Orient as a popular healthy food and an effective medicine used to treat various diseases. This fungus has demonstrated that it can produce a variety of schizophyllan and hydrolytic enzymes, such as xylanases [2–3]. Schizophyllan produced by S. commune, an exopolysaccharide (EPS), is reportedly a homoglucan that possesses a β-1,3-linked backbone with single β-1,6-linked glucose side chains at approximately every third residue (Fig. 1) [4]. Schizophyllan has numerous potential applications. It can be used in petroleum recovery, as a thickener for cosmetic lotions, and as oxygen-impermeable films for food preservation. A great deal of efforts have been made on the pharmaceutical applications of the EPS. Schizophyllan acts as a biological response modifier and a non-specific stimulator of the immune system. By this action, the EPS has been used to work against a number of diseases, including AIDS, and to enhance the effect of vaccines and anti-cancer therapies. Currently, schizophyllan is commercially produced in China and Japan as an anti-tumor agent [3].
In 2007, we even found that S. commune fermentation broth (SCFB) had a significant effect of anti-anoxia by the hermetic experiment. The deposition by ethanol of SCFB, which mainly consisted of EPS is capable of significantly resisting anoxia (P < 0.01) [5].
To accelerate the mycelial growth and EPS formation, different factors have been proven to have stimulatory effect [6–8] in the previous researches. It was reported that fatty acids, oils, and surfactants could increase the production of fungal metabolites such as citric acid, aflatoxins, and carotenes as well as exocellular enzymes [9–10]. It was also reported that the production of microbial EPS was stimulated by some fatty acids [11]. Their effects may be due to depression, induction, or stimulation of secretion. The vegetable oils at certain levels did stimulate mycelial growth and polysaccharide formation by Ganoderma lucidum [12]; however, higher concentrations might also cause inhibition. The enhancement or inhibition might be dependent upon the types of fatty acids present in oil. Fan-Chiang Yang reported that oleic acid at the level of 0.15 g/100 mL led to a significant increase in cell concentration from 0.20 to 0.46 g/100 mL in a suspended culture of G. lucidum.
However, almost all reporters just tested one or two factors. This research investigated the effects of single and mixture of different factors on EPS formation and mycelial yield by S. commune.
Materials and Methods
Microorganism and Culture Conditions
S. commune (Fr.) (screened by this research group and reserved at the Tianjin Key Lab of Industrial Microbiology, Tianjin University of Science & Technology) was used through out this study. The stock culture was maintained on potato dextrose agar slant (g/L): potato juice 200, glucose 20, MgSO4·7H2O 1.5, KH2PO4 3.0, VB1 0.01, and agar 20 and subcultured once a month. Slants were incubated at 26°C for 6 d and then stored at 4°C. The medium for inoculum preparation (seed medium) contained the following components (g/L): glucose 30, soybean powder 10, yeast extract 1, MgSO4·7H2O 0.5, KH2PO4 1.0, and VB1 0.01, pH 6.0. Flask cultures were carried out in 500-mL Erlenmeyer flasks containing 150 mL medium. The fermentation medium for flask culture consisted of the following components (g/L): 5.0 NH4Cl, 12° Brix malt extract.
Inoculum Preparation, Flask Culture, and Bioreactor Fermentation
The seed culture by transferring approximately 1.0 cm2 of the slant culture into a 500-mL Erlenmeyer flask containing 150 mL seed medium at 26°C on a rotary shaker at 150 rpm for 3 d. The flask culture experiments were performed in 500-mL flasks containing 150 mL of the fermentation medium after inoculation with 10% (v/v) of the inoculum at 26°C on a rotary shaker incubator at 150 rpm for 6 d. The bioreactor fermentation was performed in a 7-L bioreactor (East Biotech, GBCS-5W, China) with an initial volume of 4 L at 26°C, and the agitation speed was set at 150 rpm.
Analytical Methods
Biomass Concentration
The mycelia in freely suspended cultures were filtered through reweighed filter paper by suction, washed for three times with water, dried to constant weight at 60°C, and determined for biomass.
Exopolysaccharide Concentration
The filtrate obtained in the above was used for the determination of polysaccharide concentration. Adding absolute ethanol (V ethanol/V filtrate = 4:1) to filtrate and stirring simultaneously at room temperature, the crude polysaccharide precipitated after 12 h. The precipitated polysaccharide was collected by centrifugation at 3, 000×g for 10 min and then dried at 60°C to remove the residual ethanol. Total polysaccharide in the culture medium was determined by phenol-sulfuric acid assay according to the procedure reported by Dubois et al. [13].
Residual Sugar Concentration
Residual sugars were estimated by the dinitrosalicylic acid method of Miller [14].
Photo
To record the morphology, biomass was transferred to a 9-cm Petri dish and photographed using a digital camera (DX7630, Kodak, Japan).
Results and Discussions
One-Factor-at-a-Time Method
Effect of Sodium CMC on EPS Production and Mycelial Growth
The rheological change of broth by adding surface-active reagents such as carboxymethylcellulose (CMC) affects bubble formation, flow pattern, and mass transfer coefficient of oxygen [15]. Many researchers have also reported that gas hold-up increased in the presence of CMC in the airlift fermentor [15–17]. In this study, the effect of CMC on EPS production and mycelial growth by the addition of CMC was investigated.
Figure 2 showed the result with the addition of CMC at different concentrations (0.2–1.0%) (w/v). The maximum EPS and mycelial dry weight were obtained at 0.6% CMC (w/v), achieving 2.25 and 21.35 g/L, respectively. Compared with the control, the EPS production and mycelial growth with the addition of CMC at 0.6% were enhanced by 17.25% and 19.14%, respectively.
Naohiko Taga reported that the viscosity of broth and the volumetric mass transfer coefficient (KLa) of the fermentor were affected by the addition of CMC. The P(3HB) production rate was about twice of the control when 0.05% CMC was added in the medium. Some kinds of reagents, such as surface-active reagents, were also known to increase oxygen transfer rate and viscosity of the culture medium [18].Hence, the addition of CMC may also increase oxygen transfer rate; as a result, the EPS production and mycelial growth were also increased.
Effect of l-Glutamic Acid on EPS Production and Mycelial Growth
The cultivation with the addition of l-glutamic acid at different concentrations (0.5–2.0 mg/L) was shown in Fig. 3. When 1.0 mg/L l-glutamic acid was added to the medium, the maximum EPS and mycelial concentrations were 2.33 and 20.51 g/L, respectively. Compared with the control, EPS production and mycelial growth with the addition of 1.0 mg/L l-glutamic acid were enhanced by 21.62% and 10.21%, respectively. Nampoothiri et al. [19] reported the increased yield of gellan gum with the addition of amino acids, and Survase et al. [20] also reported that the yield of acleroglucan increased when added with l-lysine. Survase et al. [20] found that amino acids may serve as the metabolic precursors for the scleroglucan production indicating that l-glutamic acid may also be the metabolic precursor for the EPS production.
Effect of Thiamin (VB1) on the EPS Production and Mycelial Growth
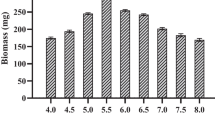
Vitamins are co-enzymes in cells, which are important factors in most mushroom growth. Thiamin, an important composition of cocarboxylase in the cells, is necessary in mushroom carbon metabolism. The effect of thiamin concentration on EPS production was studied in the range of 0.5–2.5 mg/L. Considering both mycelial growth and EPS production, the optimum concentration of thiamin was 0.5 mg/L. Accordingly, the concentrations of EPS and mycelia achieved by the addition of 0.5 mg/L thiamin were 1.16 and 14.33 g/L, respectively, corresponding to 38.16% and 29.62% enhancement compared with the control (Fig. 4).
Effect of NAA on EPS Production and Mycelial Growth
In sulfated polysaccharide production by a marine microalga of Gyrodinium impudicum strain KG03, with the addition of naphthalene acetic acid (NAA) in the f/2 medium, the yield was 46.7 mg/L, which was 2.7 times of the original medium [8]. To examine the effect of NAA on mycelial growth and EPS production, NAA was added into the culture medium at different concentrations. The highest EPS and mycelial concentration were observed at 0.2 mg/L NAA and reached 1.03 and 16.44 g/L, respectively, which were 28.75% and 56.04% higher than those obtained in the control (Fig. 5).
Effect of Oleic Acid on the EPS Production and Mycelial Growth
Fan-Chiang Yang reported the effects of oleic acid on mycelial growth and EPS formation by G. lucidum in flask culture. The stimulatory effect of olive oil might be attributed to the fact that the main composition of the oil is oleic acid [12]. In this study, the effects of oleic acid concentration on EPS production and mycelial growth were examined in the range of 0.1–0.4% (w/v) (Fig. 6). At the optimum concentration of 0.1% oleic acid, EPS and mycelial concentrations reached 1.71 and 17.98 g/L, respectively, corresponding to the respective 58.33% and 56.89% enhancement compared with the control. Results presented in this paper indicated that the synthesis of EPS and mycelial growth by S. commune could substantially increase in the presence of oleic acid. Although it was reported that fatty acid could stimulate the production of other fungal metabolites, their effect on polysaccharide formation has only been examined in few studies.
The mechanism of stimulatory effect has been proposed that oils or factty acids modified membrane composition and increase the membrane permeability or directly affected the level of synthesis of the enzymes involved in polysaccharide production [21].
It was also suggested that the lipids may be partially incorporated in the cell membrane, thereby facilitating immediate uptake of nutrients from the culture medium. According to the paper, the major fatty acid in some fungus was a di-unsaturated 18 carbon acid. The effect of linoleic acid (C18) on mycelial growth and EPS production would be of interest and deserve further study [12].
Effect of Tween 80 on the EPS Production and Mycelial Growth
Tween 80 is the most commonly used amendment in the production of enzymes by solid-state fermentation [22]. However, very little information is available on the effects of Tween 80 on the EPS production and mycelial growth of mushroom.
The effect of Tween 80 on mycelial growth and EPS production was examined by adding Tween 80 into the medium at different concentrations (Fig. 7). At 6 mL/L Tween 80, the highest achievement of EPS and mycelia were 2.65 and 23.46 g/L, respectively, exhibiting the respective 47.92% and 43.24% increase compared with those of the control.
The Orthogonal Matrix Method
To investigate the relationship between factors and optimize their concentrations for mycelial growth and EPS production, the orthogonal matrix L9(34) method was used [23]. To each the same results as those of the orthogonal matrix method, 34, that is, 81 experiments are necessary to achieve experimental goals for full-factors experimental projects. Orthogonal projects, as a result of the suitable design of factors, can give effective responses. They have been successfully applied to improve culture media for the production of primary and secondary metabolites in fermentation process [23]. According to preliminary experiments, with only nine experiments of L9(34) orthogonal projects, we selected and varied three levels as shown in Table 1. The experimental conditions for each project are listed in Table 2, and experiment results are included in the last two columns. The fermentation conditions of temperature, agitation rate, and growth period were fixed at 26°C, 150 rpm, and 6 d, respectively.
Order of Effects of Factors
According to the orthogonal method [23], the effects of those factors on mycelial growth and EPS production were calculated, and the results are shown in Table 3. According to the magnitude order of R (Max Dif), the effect of factors on mycelial growth could be ordered as oleic acid > VB1 > Tween 80; those on EPS production were in the same order. This result pointed out that the effect of oleic acid was more important than that of other factors.
Optimum Levels of Each Factor
The results in Table 3 showed the following: (1) To obtain a high EPS production, the optimum composition were oleic acid 0.1% (v/v), VB1 0.5 mg/L, and Tween 80 6 mg/L. Under this condition, EPS was increased by 67.07% compared with the control. (2) To obtain a high mycelial growth, the optimum compositions were oleic acid 0.1% (v/v), VB1 0.5 mg/L, and Tween 80 6 mg/L. Under this condition, mycelia could increase 63.18% compared with the control.
The Verification Experiments in 7-L Bioreactor
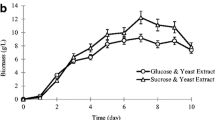
Figure 8 showed the typical time courses of mycelial growth and EPS production in a 7-L stirred-tank bioreactor under optimal culture condition (oleic acid 0.1% (v/v), VB1 0.5 mg/L, and Tween 80 6 mg/L) for EPS production. The maximum EPS production indicated was achieved at 2.35 g/L after 6 d of fermentation, while the maximum mycelial yield was 19.83 g/L after 6 d. This also corresponded to 38.96% and 22.16% enhancement in EPS production and mycelial dry weight, respectively, compared with the control.
However, the EPS and mycelial dry weight under the optimal conditions (oleic acid 0.1% (v/v), VB1 0.5 mg/L, and Tween 80 6 mg/L) in 7-L bioreactor were lower than those in shaking flasks, which might be due to fact that mycelia were damaged by the mechanical action of agitator. Figure 9a and b showed that, compared with mycelia in the flask, mycelia in reactor had more mycelial fragment, indicating that mycelia had been damaged by the mechanical action of agitator and further effected the EPS production.
Conclusions
S. commune (Fr.) has been regarded as a popular food in the Orient and an effective medicine used to treat various diseases with numerous potential applications. In 2007, we even found that SCFB had a significant effect of anti-anoxia by the hermetic experiment. The deposition by ethanol of SCFB, which mainly consist of EPS, is capable of significantly resisting anoxia (P < 0.01) [5].
In order to attain a higher mycelial and EPS production, six different factors (sodium carboxymethylcellulose, l-glutamic acid, VB1, NAA, oleic acid, and Tween 80) were determined on their effect on the EPS production and mycelial growth. Using the one-factor-at-a-time method and orthogonal matrix method, the optimal conditions (oleic acid 0.1% (v/v), VB1 0.5 mg/L, and Tween 80 6 mg/L) was obtained with a high EPS yield in S. commune. This optimized conditions in shake flask culture led to a mycelial yield of 25.93 g/L and EPS production of 2.79 g/L, respectively, which were considerably higher than those obtained in preliminary studies. The subsequent experiments in 7-L fermentor confirmed the results.
References
Watling, R., & Sweeney, J. (1972). Research Communications in Chemical Pathology and Pharmacology, 3, 214–226.
Desrochers, M., Jurasek, L., & Paice, M. G. (1981). Applied and Environmental Microbiology, 41, 222–228.
Leathers, T., Nunnally, M., & Price, N. (2006). Biotechnology Letters, 28, 623–626. doi:10.1007/s10529-006-0028-1.
Rau, U. (1999). Production of schizophyllan. In C. Bucke (Ed.), Methods in biotechnology, Vol. 10. Carbohydrate biotechnology protocols. Totowa, NJ: Humana Press.
Hao, L.-M., Xing, X.-H., Zhang, Y.-K., Li, Z., Mu, J., & Wu, T.-Y. (2007). Food Science, 28, 320–323.
Schisler, L. C., & Volkoff, O. (1977). Mycologia, 69, 118–125. doi:10.2307/3758625.
Survase, S. A., Saudagar, P. S., & Singhal, R. S. (2007). Bioresource Technology, 98, 1509–1512. doi:10.1016/j.biortech.2006.05.022.
Yim, J. H., Kim, S. J., Ahn, S. H., & Lee, H. K. (2003). Biomolecular Engineering, 20, 273–280. doi:10.1016/S1389-0344(03)00070-4.
Fukushima, Y., Itoh, H., Fukase, T., & Motai, H. (1991). Applied Microbiology and Biotechnology, 34, 586–590. doi:10.1007/BF00167904.
Reese, E. T., & Maguire, A. (1969). Applied Microbiology, 17, 242–245.
Stasinopoulos, S. J., & Seviour, R. J. (1990). Biotechnology and Bioengineering, 36, 778–782.
Yang, F. C., Ke, Y. F., & Kuo, S. S. (2000). Enzyme and Microbial Technology, 27, 295–301. doi:10.1016/S0141-0229(00)00213-1.
DuBois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A., & Smith, F. (1956). Analytical Chemistry, 28, 350–356. doi:10.1021/ac60111a017.
Miller, G. L. (1959). Analytical Chemistry, 31, 426–428. doi:10.1021/ac60147a030.
Malcolm, K. M. (1991). Biotechnology and Bioengineering, 38, 1261–1270. doi:10.1002/bit.260381102.
Deckwer, W. D., & Schumpe, A. (1993). Chemical Engineering Science, 48, 889–911. doi:10.1016/0009-2509(93)80328-N.
Schumpe, A., & Deckwer, W. D. (1982). Industrial & Engineering Chemistry Process Design and Development, 21, 706–711. doi:10.1021/i200019a028.
Taga, N., Tanaka, K., & Ishizaki, A. (1997). Biotechnology and Bioengineering, 53, 529–533. doi:10.1002/(SICI)1097-0290(19970305)53:5<529::AID-BIT11>3.0.CO;2-B.
Nampoothiri, K. M., Singhania, R. R., Sabarinath, C., & Pandey, A. (2003). Process Biochemistry, 38, 1513–1519. doi:10.1016/S0032-9592(02)00321-7.
Survase, S. A., Saudagar, P. S., & Singhal, R. S. (2007). Bioresource Technology, 98, 410–415. doi:10.1016/j.biortech.2005.12.013.
Stasinopoulos, S. J., & Seviour, R. J. (1990). Biotechnology and Bioengineering, 36, 778–782. doi:10.1002/bit.260360804.
Zeng, G. M., Shi, J. G., Yuan, X. Z., Liu, J., Zhang, Z. B., Huang, G. H., et al. (2006). Enzyme and Microbial Technology, 39, 1451–1456. doi:10.1016/j.enzmictec.2006.03.035.
Xu, C. P., Kim, S. W., Hwang, H. J., Choi, J. W., & Yun, J. W. (2003). Process Biochemistry, 38, 1025–1030. doi:10.1016/S0032-9592(02)00224-8.
Acknowledgments
We thank Mrs. Guifang Deng for her kindly help in fermentation experiments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hao, Lm., Xing, Xh., Li, Z. et al. Optimization of Effect Factors for Mycelial Growth and Exopolysaccharide Production by Schizophyllum commune . Appl Biochem Biotechnol 160, 621–631 (2010). https://doi.org/10.1007/s12010-008-8507-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-008-8507-6