Abstract
Background
Limb salvage surgery has evolved to become the standard method of treating sarcomas of the extremities with acceptable oncologic results. However, little information exists relative to the activity level or ability to participate in sports after tumor reconstructions.
Questions/purposes
The aims of the study were to answer the following questions: (1) Which sports activity levels and what types of sports can be expected in the long term after tumor reconstruction? (2) Which frequency durations are patients with Ewing’s sarcoma able to perform in long-term followup after local control? (3) Do surgical complications affect sports activity level?
Methods
Thirty patients (13 females, 17 males; mean age, 18 ± 8 years; range, 2–36 years at diagnosis; mean followup 16 ± 6 years [minimum, 5 years]) were included. Tumors were located in the pelvis, femur, tibia, and fibula. Surgical procedures included surgical resections alone (n = 8), surgical resection with biological reconstruction (n = 9), or endoprosthetic reconstruction (n = 13). We assessed UCLA sports activity levels, kinds of sports as well as the frequency per week and the duration of each training unit at long term (minimum followup, 5 years).
Results
In long-term followup 83% patients (25 of 30) were performing athletic activity regularly. The hours/week of sports depended on type of surgery and were highest after resections in the pelvis and femur (5.8) and were lowest after megaprosthetic reconstruction of the pelvis (1.0). Patients undergoing biologic reconstructions were able to perform high-impact sports. UCLA sports activity levels were high after joint-preserving vascularized fibula for tibia reconstruction (7.4) and after megaprosthetic reconstruction of the lower extremity (6.3–6.4) and were low after tumors located in the fibula (4.2). Complications during followup did not significantly influence sports activity in long-term survivors.
Conclusions
Long-term survivors can achieve high levels of sports activity in many instances. Tumor sites are associated with the postoperative sports activity levels. This information can help surgeons counsel patients in terms of athletic expectations after limb salvage reconstruction for patients with Ewing’s sarcoma.
Level of Evidence
Level III, therapeutic study. See Guidelines for Authors for a complete description of levels of evidence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ewing’s sarcoma is one of the most common malignant bone tumors in adolescents and young adults [2, 25, 30] and is located primarily in the proximal upper and lower extremities as well as in the pelvis. Ewing’s sarcoma differs from osteosarcoma in some respects: the mean age of the patients is younger than those with osteosarcoma and seldom occurs beyond the age of 30 years, local control can be achieved with radiation as well as resection, and the drugs used to treat Ewing’s sarcoma are different than those used to treat osteosarcoma [3, 20, 31]. In addition to adequate local control, the use of aggressive polychemotherapy has remarkably improved the survival of patients with Ewing’s sarcoma [10, 25, 32, 37] since adjuvant chemotherapy was first introduced in the 1980s and has led to a growing number of long-term survivors [8, 20, 23]. However, chemotherapy can result in long-term late effects on the cardiac, pulmonary, and metabolic systems as well as on skeletal muscle [16]. Moreover, extensive tumor resections of the extremities can lead to functional impairment and therefore to restrictions and limitations in activities of daily living [11, 28].
Physical activity has well-documented positive effects in cancer survivors. Exercise leads to improvements in pulmonary function and cardiovascular endurance but also in muscle strength [29] and muscle weakness in cancer survivors can be influenced by regular exercise [4, 6, 29, 33]. Therefore, postcancer rehabilitation is of high value comprising a long-term benefit [3] and physical activity constitutes an important component to posttreatment rehabilitation. In addition, sport is an important part of most children’s and adolescents’ lifestyles and longitudinal studies show that children and adolescents who are active continue to be active throughout their lives [34, 35].
Although the functional status of patients with Ewing’s sarcoma after limb salvage treatment has been assessed using basic musculoskeletal scores like the Musculoskeletal Tumor Society (MSTS) score in previous studies [14, 16, 24, 26, 31, 37] and limitations in sports have been recently described [27], no detailed data are available on the sports activity levels of longer-term survivors.
Therefore, the aim of this study was to assess sports activity levels after limb salvage surgical procedures in patients with Ewing’s sarcoma with a minimum of 5 years followup. We specifically evaluated the following: (1) Which sports activity levels and what types of sports can be expected in the long term after tumor reconstruction? (2) Which frequency durations are patients with Ewing’s sarcoma able to perform in long-term followup after local control? (3) Do surgical complications affect sports activity level?
Patients and Methods
The study was approved by the local ethics committee and was done in accordance with the Declaration of Helsinki.
Patients
Inclusion criteria for this study were survivors of Ewing’s sarcoma treated at our institution who underwent a limb salvage procedure and multimodal treatment with a minimum of 5 years followup. Data from the institution’s tumor registry were retrieved between 1984 and 2005 and were complemented by thorough chart reviews. We identified 123 patients who had been diagnosed and treated with Ewing’s sarcoma of the pelvis, lower extremity, and who were treated by tumor resection and limb salvage surgery as well as polychemotherapy at our institution. At the time of this examination, 68 had died of Ewing’s sarcoma. The inclusion criteria were met by 55 patients. Four patients were lost to followup and four were not able to participate in the study because of major illness unrelated to Ewing’s sarcoma (eg, stroke, asthmatic disease, heart failure). Moreover, 17 patients with no evidence of disease and in good overall condition did not want to participate in the study, leaving a study cohort of 30 patients (55% of the potentially eligible 55).
Those 30 patients (Table 1) had gone through multidisciplinary treatment with polychemotherapy and surgery including resection and reconstruction. Histological analysis showed 27 Ewing’s sarcomas and three peripheral neuroendocrine tumors. Eleven (37%) of these primary tumors were located in the femur, nine (30%) in the pelvis, six (20%) in the tibia, four (13%) in the fibula. (Neo)adjuvant chemotherapy was given according to recommended protocols (Table 1) [10, 20, 23].
Fourteen (47%) patients were treated by local radiation therapy as a definitive local control or in case of one patient as an adjuvant for a marginal resection. Patients with megaprostheses or fibula per tibia reconstruction were not treated by radiation therapy unless resection margins were found to be marginal.
Surgical Procedures
Limb salvage surgery including resections only, resections, and endoprosthetic or biological reconstruction methods were performed in all patients. Wide resection margins were achieved in 29 patients. Marginal resection was performed in one patient with proximal femur resection.
Resections only were done in the innominate bone in Enneking-Dunham Zones I to IV [12] and parts of the femur diaphysis. In tumors located in the proximal and distal fibula, these were resected with adjacent parts of the proximal or distal tibia to achieve wide margins.
Endoprosthetic reconstructions were done in 10 patients who received Kotz modular femorotibial replacements (Howmedica Modular Resec-tion System®; Stryker, Mahwah, NJ, USA) of the proximal femur (n = 7), distal femur (n = 2), and proximal tibia (n = 1). In three patients, reconstruction of the pelvis was carried out by a custom-made three-dimensional pelvis endoprosthesis.
Biological reconstruction was performed in five patients who received a fibula for tibia reconstruction as a joint-preserving intercalary resection, in two patients with resection in the periacetabular region close to the pubic bone, and in two patients with reconstruction of diametaphyseal parts of the femur.
Metastases were evident in three patients; no local relapse was noted in the followup period.
Assessment
The frequency per week and the duration of each training unit, the types of the performed athletic activities, and the sports activity levels by use of the UCLA score [39] were assessed at their latest followup (minimum of 5-year followup).
The UCLA activity score evaluates the general activity level [39]. It is recommended to evaluate the patient’s athletic activity level with regard to intensity and frequency. This score is useful for monitoring physical activity of lower extremity osteoarthritic populations [36] and patients after joint arthroplasty [22].
Oncological and nononcological complications according to the ISOLS failure mode classification [18] were assessed.
Statistical Analysis
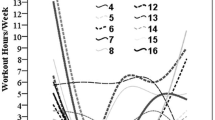
Data were coded and analyzed using the SPSS software package (SPSS, IBM, Ehningen, Germany). Nonparametric Wilcoxon signed-rank test was used to assess statistical significance when comparing long-term results of UCLA score and hours/week performance between surgical methods. Repeated-measures two-way analysis of variance was used to assess the effects of the presence and absence of complications on long-term UCLA score and on hour/week sports performance. Spearman rank correlation was used to assess the correlation between the UCLA scores at different time points (Fig. 1). A p value < 0.05 was considered statistically significant.
Mean UCLA score of patients after different surgical procedures at long-term followup highlighted in black boxes (main study aim). Patients after resections of pelvis and femur diaphyses only and after joint-preserving fibula for tibia reconstructions represent the highest UCLA ratings followed by patients after megaendoprosthetic reconstructions of the proximal and distal femur and proximal tibia with moderate levels, patients after resections around the fibula with pour, and patients after megaendoprosthetic reconstructions of the pelvis with inferior levels of sports activity. Boxes highlighted in light gray show retrospectively assessed UCLA scores of patients at certain time points prediagnosis of Ewing’s sarcoma and 1, 3, and 5 years postoperatively, although bearing recall bias, these boxes show a significant decrease 1 year postoperatively followed by increasing levels of sports activity in the followup. pT = proximal tibia modular endoprostheses; dF = distal femur modular endoprostheses; pF = proximal femur modular endoprostheses; Resection Only = resections on the innominate bone Dunham I-IV and parts of the femoral diaphysis.
Results
Sports Activity Levels and Types of Sports
The mean sports activity level measured by UCLA score was 5.7 (range, 2–9) and appeared similar to workout frequencies. Interestingly, UCLA scored highest in fibula for tibia reconstructions at 7.4 (range, 5–9) followed by patients after resections and biological reconstructions of the pelvis at 5.6 (range, 3–7) followed by megaendoprosthetic reconstruction of the knee at 6.3 (range, 5–7), megaendoprosthetic reconstructions of the proximal femur at 6.4 (range, 3–7), and were lowest after resections of the fibula resections at 4.25 (range, 2–8) and after pelvis megaendoprosthesis at 4.0 (range, 3–6). Types of sports depended on surgical procedure. High-impact sports including jogging and skiing were possible after megaendoprostheses of the knee and proximal femur. Tennis in addition to these types of sports was possible after joint-preserving fibula for tibia reconstruction. None of these sports was possible after resections of tumors located in the fibula (Table 2).
Frequencies
In long-term followup, 25 of 30 (83%) patients were able to perform regular sports; the mean workout was 2.8 (range, 0–15) hours/week. Depending on the surgical methods, the mean sports activity differed (Fig. 1). Patients after resections and biological reconstructions of the pelvis had a mean workout of 5.8 (range, 1–15) hours per week after megaendoprosthetic reconstruction of the knee at 2.0 (range, 1–3), after megaendoprosthetic reconstructions of the proximal femur at 2.1 (range, 0–8), and after fibula for tibia reconstructions at 2.0 (range, 0.5–4) hours per week, whereas the workout after resections of the fibula was 1.7 (range, 0–4) and after pelvis megaendoprosthesis at 1.3 in one patient.
Complications
As a result of the achievement of wide resection in primary surgery after tumors localized in the proximal fibula, the superficial branch of the peroneal nerve was resected in one patient. Another had complete peroneal nerve resection and no success in a suralis nerve transplantation. In one patient, an upper ankle arthrodeses was performed because of tumor localized in the distal fibula.
Twelve (40%) patients had at least one complication, which led to revision surgery. Complications according to the ISOLS failure mode classification [18] were seen in 10 of 13 patients (77%) with megaendoprostheses. Soft tissue failure (Type 1) was observed in seven (54%) patients, aseptic loosing (Type 2) in four patients (11%), structural failure (Type 3) in five patients (38%), infection (Type 4) in three patients (9%), and local recurrence (Type 5) in none of the patients. In patients after biological reconstructions and resections, five of 17 patients (30%) had complications that led to revisions. Lung metastases occurred in three (10%) patients and one metastasis in the femur in one patient; all were resected. No significant differences in sports activity levels between patients with or without complications were seen.
Discussion
The development of limb salvage surgery [2, 13, 32] was a major advance in treatment of primary bone sarcomas [14, 16, 31], allowing patients potentially to return to sport in ways not possible when amputations were the mainstay of treatment. Currently, limb salvage procedures are still the first choice in primary bone sarcoma surgery, although developmental changes in technical orthopaedics have led to high levels of sports activity in amputees [7, 9].
However, as a result of the lack of data, clinicians currently cannot anticipate if sports will be possible after surgery and which types of sports can be pursued. Therefore, it is difficult to advise young patients and their parents regarding what they can expect. Realistic and positive expectations lead to better health outcomes after surgical procedures [21], and so better data on what patients achieve in terms of sports performance after tumor surgery would be important. Furthermore, survivors of bone tumors pose a poorer health status compared with healthy control subjects [1] and physical training may provide beneficial effects on health like seen in cancer survivors in general [29, 33]. In this study, we found moderate sports activity levels in patients after megaendoprosthetic reconstructions in the proximal femur, distal femur, and proximal tibia in the long term. These and especially patients after joint-preserving fibula for tibia reconstructions were able to practice even high-impact sports like jogging and skiing. In general long-term sports activity was not affected by complications associated with the disease.
There are several limitations to this study. Types of sports may be different in other parts of the world. This study reviews sports of patients living in central Europe. We are aware of the selection bias resulting from our focus on long-term survivors who represent a healthier group of patients with Ewing´s sarcoma and also as a result of 30% of the patients who wished not to take part in the study. This suggests that our results should be construed as a “best-case analysis” of what might be expected after resection and reconstruction for Ewing’s sarcoma. The reason why apparently healthy survivors elected not to participate in sports may be because it forced them to recall their limitations resulting from their disease in some study participations and/or patients were too busy; alternatively, because of their age (mean, 36 years), they may be highly involved in their profession and business and do not have the time to fill out a questionnaire. There is a great age distribution in our study cohort; however, differences between patients at 30 years of age and patients at 40 years of age may not be too different. In fact, longitudinal studies show that children and adolescents who are active are going to be active throughout their lives [35, 36]. Concerning the retrospective preoperative sports data (Fig. 1), there is a certain recall bias, although we noted that the patients remembered their physical activities precisely, indicating the importance of sports in their personal history after tumor treatment.
To date in young patients after bone tumor treatment, no sports activity levels have been assessed yet. In our study, we found that 83% patients performed sports at long-term followup, indicating the importance of sports in the life of adolescents and young adults. There are several studies on sport activities of patients after elective joint replacement. The current trend of expert opinion favors more liberal recommendations in sports activity resulting from a growing number of young patients undergoing total joint arthroplasty [17, 38]. Depending on surgical methods, survivors reached different post-Ewing’s sarcoma sports activity levels with overwhelming scoring after fibula for tibia reconstruction. This may point out the importance of joint-preserving surgical procedures. Not significantly worse were the levels of megaprosthetic methods of the knee and hip. These findings were in line with previous studies about sports activity and elective joint arthroplasty of the hip and the knee [5], in which patients returned to moderate sports levels. Some patients after implantation of megaprostheses of the proximal femur, distal femur, and proximal tibia were able to practice sports not even recommended by the Hip and Knee Society Survey 2005 (skiing, jogging) for THA and TKA [17]. However, patients after megaprosthetic reconstruction of the pelvis as well as resection of tumors located in the proximal and distal fibula showed low activity levels. This was surprising, but a closer look at tumors located around the fibula revealed collateral damage of adjacent structures. Reasons for that were complete and incomplete peroneal nerve resections, mobilized with a lower leg orthosis as well as loss of ankle function resulting from arthrodeses in one patient with tumor site in the distal fibula. Many patients were practicing low-impact sports like cycling, hiking/Nordic walking, swimming, and working out in a fitness center. High-impact sports were hardly practiced at all.
Depending on different surgical methods, we found different workout frequencies. Patients after resections and biological reconstructions of the pelvis had a high workout of 5.8 hours per week followed by megaprosthetic reconstruction of the proximal femur and of the knee ranging between 2.1 and 2.0 hours per week. Patients after fibula for tibia reconstructions reached 2.0 hours per week in the long term as well. Interestingly, the sports performance of patients after fibula resections was poor (1.7) and not unexpectedly poor after receiving a pelvis megaprosthesis (1.3). Garber et. al. reported that 3.75 hours/week of sports activity are recommended for healthy adults [15]. According to this, patients after Ewing’s sarcoma with megaprostheses achieved 53% to 80% of recommended sports activity and fibula resections and megaprostheses of the pelvis failed to achieve these recommended sports activity by more than 100%.
Forty percent of patients had complications requiring revision surgery. Taking primary surgery into consideration, they had an impact on the immediate sports outcome. In the recall data (Fig. 1), sports activity 1 year postoperatively was significantly worse. The same would be expected after major revision surgery after complications, but statistical significance cannot be proved as a result of different time to complications intervals at a certain time point of investigation. However, in the long term, there was no objective evidence that complications affect sports activity. We consider that, after all, recovery may be possible from both oncologic as well as nononcologic complications. Furthermore, no complications occurred that were caused by practicing sports.
Healthy long-term Ewing’s sarcoma survivors can achieve high levels of sports activity after Ewing’s sarcoma. The site of the tumor influenced the performance levels achieved. Patients with tumors located in the fibula are at high risk to develop low sports activity levels because of peroneal nerve resections or arthrodeses of the upper ankle. Preoperative sports activity levels may be an indicator for postoperative sports activity levels. This information can help surgeons counsel patients in terms of athletic expectations after limb salvage reconstruction for patients with Ewing’s sarcoma. Our study strongly advocates specific training programs during long-term rehabilitation based on the knowledge of what is possible in young bone sarcoma survivors. This could hypothetically improve quality of life in these patients.
References
Aksnes LH, Bauer HCF, Dahl AA, Fosså SD, Hjorth L, Jebsen N, Lernedal H, Hall KS. Health status at long-term follow-up in patients treated for extremity localized Ewing sarcoma or osteosarcoma: a Scandinavian sarcoma group study. Pediatr Blood Cancer. 2009;53:84–89.
Arndt CAS, Rose PS, Folpe AL, Laack NN. Common musculoskeletal tumors of childhood and adolescence. Mayo Clin Proc. 2012;87:475–487.
Bielack S, Jürgens H, Jundt G, Kevric M, Kühne T, Reichardt P, Zoubek A, Werner M, Winkelmann W, Kotz R. Osteosarcoma: the COSS experience. Cancer Treat Res. 2009;152:289–308.
Burnham TR, Wilcox A. Effects of exercise on physiological and psychological variables in cancer survivors. Med Sci Sports Exerc. 2002;34:1863–1867.
Chatterji U, Ashworth MJ, Lewis PL, Dobson PJ. Effect of total knee arthroplasty on recreational and sporting activity. ANZ J Surg. 2005;75:405–408.
De Backer IC, Vreugdenhil G, Nijziel MR, Kester AD, van Breda E, Schep G. Long-term follow-up after cancer rehabilitation using high-intensity resistance training: persistent improvement of physical performance and quality of life. Br J Cancer. 2008;99:30–36.
DiCaprio MR, Friedlaender GE. Malignant bone tumors: limb sparing versus amputation. J Am Acad Orthop Surg. 2003;11:25–37.
Dirksen U, Jürgens H. Approaching Ewing sarcoma. Future Oncol. 2010;6:1155–1162.
Doukas WC, Hayda RA, Frisch HM, Andersen RC, Mazurek MT, Ficke JR, Keeling JJ, Pasquina PF, Wain HJ, Carlini AR, MacKenzie EJ. The Military Extremity Trauma Amputation/Limb Salvage (METALS) study: outcomes of amputation versus limb salvage following major lower-extremity trauma. J Bone Joint Surg Am. 2013;95:138–145.
Dunst J, Jürgens H, Sauer R, Pape H, Paulussen M, Winkelmann W, Rübe C. Radiation therapy in Ewing’s sarcoma: an update of the CESS 86 trial. Int J Radiat Oncol Biol Phys. 1995;32:919–930.
Eiser C. Assessment of health-related quality of life after bone cancer in young people: easier said than done. Eur J Cancer. 2009;45:1744–1747.
Enneking WF, Dunham WK. Resection and reconstruction for primary neoplasms involving the innominate bone. J Bone Joint Surg Am. 1978;60:731–746.
Enneking WF, Spanier SS, Goodman MA. A system for the surgical staging of musculoskeletal sarcoma. Clin Orthop Relat Res. 1980;153:106–120.
Fuchs B, Valenzuela RG, Inwards C, Sim FH, Rock MG. Complications in long-term survivors of Ewing sarcoma. Cancer. 2003;98:2687–2692.
Garber CE, Blissmer B, Deschenes MR, Franklin BA, Lamonte MJ, Lee I-M, Nieman DC, Swain DP, American College of Sports Medicine. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc. 2011;43:1334–1359.
Ginsberg JP, Goodman P, Leisenring W, Ness KK, Meyers PA, Wolden SL, Smith SM, Stovall M, Hammond S, Robison LL, Oeffinger KC. Long-term survivors of childhood Ewing sarcoma: report from the childhood cancer survivor study. J Natl Cancer Inst. 2010;102:1272–1283.
Healy WL, Sharma S, Schwartz B, Iorio R. Athletic activity after total joint arthroplasty. J Bone Joint Surg Am. 2008;90:2245–2252.
Henderson ER, Groundland JS, Pala E, Dennis JA, Wooten R, Cheong D, Windhager R, Kotz RI, Mercuri M, Funovics PT, Hornicek FJ, Temple HT, Ruggieri P, Letson GD. Failure mode classification for tumor endoprostheses: retrospective review of five institutions and a literature review. J Bone Joint Surg Am. 2011;93:418–429.
Juergens C, Weston C, Lewis I, Whelan J, Paulussen M, Oberlin O, Michon J, Zoubek A, Juergens H, Craft A. Safety assessment of intensive induction with vincristine, ifosfamide, doxorubicin, and etoposide (VIDE) in the treatment of Ewing tumors in the EURO-EWING 99 clinical trial. Pediatr Blood Cancer. 2006;47:22-29.
Jürgens H, Bier V, Dunst J, Harms D, Jobke A, Kotz R, Kühl J, Müller-Weihrich S, Ritter J, Salzer-Kuntschik M. [The German Society of Pediatric Oncology Cooperative Ewing Sarcoma Studies CESS 81/86: report after 6 1/2 years] [in German]. Klin Padiatr. 1988;200:243–252.
Mondloch MV, Cole DC, Frank JW. Does how you do depend on how you think you’ll do? A systematic review of the evidence for a relation between patients’ recovery expectations and health outcomes. CMAJ. 2001;165:174–179.
Naal FD, Impellizzeri FM, Leunig M. Which is the best activity rating scale for patients undergoing total joint arthroplasty? Clin Orthop Relat Res. 2009;467:958–965.
Paulussen M, Craft AW, Lewis I, Hackshaw A, Douglas C, Dunst J, Schuck A, Winkelmann W, Köhler G, Poremba C, Zoubek A, Ladenstein R, van den Berg H, Hunold A, Cassoni A, Spooner D, Grimer R, Whelan J, McTiernan A, Jürgens H. Results of the EICESS-92 Study: two randomized trials of Ewing’s sarcoma treatment–cyclophosphamide compared with ifosfamide in standard-risk patients and assessment of benefit of etoposide added to standard treatment in high-risk patients. J Clin Oncol. 2008;26:4385–4393.
Porsch M, Kornhuber B, Hovy L. Functional results after partial pelvic resection in Ewing’s sarcoma of the ilium. Arch Orthop Trauma Surg. 1999;119:199–204.
Potratz J, Dirksen U, Jürgens H, Craft A. Ewing sarcoma: clinical state-of-the-art. Pediatr Hematol Oncol. 2012;29:1–11.
Rödl RW, Hoffmann C, Gosheger G, Leidinger B, Jürgens H, Winkelmann W. Ewing’s sarcoma of the pelvis: combined surgery and radiotherapy treatment. J Surg Oncol. 2003;83:154–160.
Rueegg CS, Michel G, Wengenroth L, von der Weid NX, Bergstraesser E, Kuehni CE. Physical performance limitations in adolescent and adult survivors of childhood cancer and their siblings. PLoS One. 2012;7. Available at: www.ncbi.nlm.nih.gov/pmc/articles/PMC3474773/. Accessed March 12, 2013.
Ruggieri P, Mavrogenis AF, Mercuri M. Quality of life following limb-salvage surgery for bone sarcomas. Expert Rev Pharmacoecon Outcomes Res. 2011;11:59–73.
Schneider CM, Hsieh CC, Sprod LK, Carter SD, Hayward R. Cancer treatment-induced alterations in muscular fitness and quality of life: the role of exercise training. Ann Oncol. 2007;18:1957–1962.
Schrager J, Patzer RE, Mink PJ, Ward KC, Goodman M. Survival outcomes of pediatric osteosarcoma and Ewing’s sarcoma: a comparison of surgery type within the SEER database, 1988-2007. J Registry Manag. 2011;38:153–161.
Schuck A, Ahrens S, Paulussen M, Kuhlen M, Könemann S, Rübe C, Winkelmann W, Kotz R, Dunst J, Willich N, Jürgens H. Local therapy in localized Ewing tumors: results of 1058 patients treated in the CESS 81, CESS 86, and EICESS 92 trials. Int J Radiat Oncol Biol Phys. 2003;55:168–177.
Sluga M, Windhager R, Lang S, Heinzl H, Bielack S, Kotz R. Local and systemic control after ablative and limb sparing surgery in patients with osteosarcoma. Clin Orthop Relat Res. 1999;358:120–127.
Sprod LK, Hsieh CC, Hayward R, Schneider CM. Three versus six months of exercise training in breast cancer survivors. Breast Cancer Res Treat. 2010;121:413–419.
Tammelin T, Näyhä S, Hills AP, Järvelin MR. Adolescent participation in sports and adult physical activity. Am J Prev Med. 2003;24:22–28.
Telama R, Yang X, Laakso L, Viikari J. Physical activity in childhood and adolescence as predictor of physical activity in young adulthood. Am J Prev Med. 1997;13:317–323.
Terwee CB, Bouwmeester W, van Elsland SL, de Vet HCW, Dekker J. Instruments to assess physical activity in patients with osteoarthritis of the hip or knee: a systematic review of measurement properties. Osteoarthritis Cartilage. 2011;19:620–633.
Thomas IH, Cole WG, Waters KD, Menelaus MB. Function after partial pelvic resection for Ewing’s sarcoma. J Bone Joint Surg Br. 1987;69:271–275.
Vogel LA, Carotenuto G, Basti JJ, Levine WN. Physical activity after total joint arthroplasty. Sports Health. 2011;3:441–450.
Zahiri CA, Schmalzried TP, Szuszczewicz ES, Amstutz HC. Assessing activity in joint replacement patients. J Arthroplasty. 1998;13:890–895.
Acknowledgments
We thank Prof M. Dominkus for being in charge of the local tumor registry for many years providing necessary basic information. Furthermore, we thank the former and present secretaries of the tumor registry, Mag. T. Zettl and O. Wallner, respectively.
Author information
Authors and Affiliations
Corresponding author
Additional information
Each author certifies that he or she, or a member of his or her immediate family, has no funding or commercial associations (eg, consultancies, stock ownership, equity interest, patent/licensing arrangements, etc) that might pose a conflict of interest in connection with the submitted article.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research editors and board members are on file with the publication and can be viewed on request.
Clinical Orthopaedics and Related Research neither advocates nor endorses the use of any treatment, drug, or device. Readers are encouraged to always seek additional information, including FDA-approval status, of any drug or device prior to clinical use.
Each author certifies that his or her institution approved the human protocol for this investigation, that all investigations were conducted in conformity with ethical principles of research, and that informed consent for participation in the study was obtained.
This paper was in part presented at the ISOLS meeting 2013 in Bologna, Italy, and was awarded a Poster Award.
About this article
Cite this article
Hobusch, G.M., Lang, N., Schuh, R. et al. Do Patients With Ewing’s Sarcoma Continue With Sports Activities After Limb Salvage Surgery of the Lower Extremity?. Clin Orthop Relat Res 473, 839–846 (2015). https://doi.org/10.1007/s11999-014-3622-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-014-3622-x