Abstract
Background
Dislocation remains common after total hip arthroplasty. Efforts have been made to identify and minimize risk factors. One such factor, jump distance, or the distance the femoral head must travel before dislocating, has been poorly characterized with respect to three-dimensional kinematics.
Questions/purposes
We therefore determined: (1) the three-dimensional stability of four different component designs; (2) whether the degree of abduction and anteversion affects the stability; (3) whether pelvic inclination angles affected stability; and (4) which combination of these three factors had the greatest stability.
Methods
We created a positionable three-dimensional model of a THA. Acetabular components were modeled in various abduction and anteversion angles and in two different pelvic inclinations which simulate standing and chair-rising activities.
Results
The posterior horizontal dislocation distance increased as inclination angle and femoral head size increased. The 48-mm resurfacing typically had lower jump distances and was at risk of posterior edge loading at 30° inclination. The highest jump distance for all positions and activities occurred with the dual-mobility bearing.
Conclusion
These findings suggest that monoblock cups require extremely accurate positioning for low dislocation risk and that pelvic orientation may increase dislocation risks.
Clinical Relevance
As a result of the dual-mobility designs having the greatest resistance to dislocation, these cups may be appropriate for patients who are at risk for dislocation in difficult primary situations and in revision hip arthroplasty procedures in which proper component orientation may be less likely to be achieved.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dislocation remains a major complication after THA, often requiring revision surgery. In a recent study of 51,345 revision hip arthroplasties from a Medicare database, dislocation was the leading cause of revision (22%), which was higher than both infection and aseptic loosening [10]. In addition, the revision burden is expected to increase over the next 20 years from 10% presently to over 20% of all THAs by the year 2030 [25]. Dislocation causes are multifactorial and are associated with femoral stem design [1, 48], acetabular component orientation [2, 7, 28], surgical approach [7, 31, 34], soft tissue laxity [40], femoral head size [12, 21, 23, 34], and other patient-specific factors [7, 18, 34].
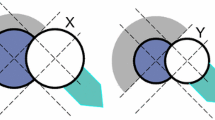
Mechanisms described for the characterization of dislocation using these testing conditions include levering the femoral head out of the acetabulum secondary to impingement [2, 33] as well as femoral head translation resulting from muscle weakness [6, 33] or soft tissue laxity [11]. Regardless of the mechanism of dislocation, the femoral head must travel a certain distance, typically termed the jump distance, before it can be unseated from the acetabular component. Prior studies to quantify these measurements did so by using simplified two-dimensional geometry (Fig. 1) to compare the effect of varying femoral head sizes on the jump distance required for dislocation. Additionally, these studies used a range of acetabular cup positions (varying through the extremes of 0° to 90° of abduction), which can generate results that might never be realized in an in vivo setting. However, the continued prevalence of dislocation after THA suggests that although jump distance may be a valid indicator for dislocation risk, the way this value is determined may require a more sophisticated approach and that for any given bearing size, there may be multiple other factors that affect it [16]. In this study, our approach will analyze various clinical relevant designs and component orientations to further clarify our understanding of dislocation risk.
We therefore evaluated: (1) the three-dimensional stability of four different component designs; (2) whether various clinically relevant positions of abduction and anteversion affect the stability; (3) whether varying pelvic inclination angles as measured during standing and during rising from a seated position affect stability; and (4) which combinations of implant type, component orientation, and pelvic tilt had the greatest stability.
Materials and Methods
We first defined a three-dimensional posterior horizontal dislocation distance (PHDD) or three-dimensional jump distance. The PHDD was determined using a three-dimensional solid model, whereby the following various factors were varied for four different prosthetic designs: acetabular inclination angle, acetabular anteversion, and forward pelvic tilt. The dependent output of the model was the distance traveled by the femoral head just before dislocation (PHDD), which was then compared between the different implants as well as the other variables. The definition of PHDD and the model characteristics are described subsequently.
In the horizontal plane, the PHDD is the minimum posterior-directed distance the femoral head must travel to be located tangential to the edge of the acetabular component in a direction directly posterior. The femoral component was initially seated within the acetabular component. The axes origin was located at the center of the femoral head. The x-axis was pointed anteriorly, the y-axis superiorly, and the z-axis laterally from this origin. For a right hip, we then established a point at 270° (9 o’clock) as the location where a posterior dislocation would occur. The head of the femoral component was then placed tangential to this point and the distance between the current center of the femoral (Fig. 2C) head and the previously defined origin was calculated as the PHDD (denoted as distance X in Fig. 2). We imported a CT scan of an adult pelvis into Pro/Engineer computer-aided design software (Parametric Technology Corporation, Needham, MA, USA). The scan used was a thin slice, high resolution that can create a three-dimensional solid model of the pelvis. This was used for visualization purposes only and did not affect the analysis. Four different combinations of acetabular and femoral components were modeled in Pro/Engineer and were used to calculate PHDD.
We studied four implant designs. The first two implants (Trident Alumina Ceramic Bearing; Stryker Orthopaedics, Mahwah, NJ, USA) had 28- and 36-mm inner acetabular diameters and were internally hemispheric. The third design was a resurfacing-type cup (Cormet Hip Resurfacing System; Corin, Gloucestershire, UK) with a bearing that was lateralized by 3.5 mm and had a 48-mm bearing diameter. This bearing had approximately 163° of coverage of the femoral head. The fourth design was a monoblock dual-mobility cup (Anatomic Dual Mobility™; Stryker Orthopaedics) with an anatomic-shaped rim with a 48-mm diameter head (Fig. 3). The anatomic shape allows the cup to be deeper than a hemisphere in areas. For the dual-mobility design, the three-dimensional measurement was taken with the cup implanted in the recommended orientation (as illustrated in Fig. 2C).
We used various acetabular inclination and abduction angles to determine their effect on the PHDD for two different pelvic tilt angles. The acetabular component was placed at varying positions, angles of inclination of 30°, 45°, and 60°, and anteversion angles of 0°, 10°, and 20°, leading to a total of nine experimental acetabular positions.
Values for pelvic inclination angles were obtained from gait analysis laboratory measurements taken from healthy volunteers and have been reported previously. We modeled two typical positions: a standing (5°) and an extreme position with the greatest value of pelvic tilt, which occurred when rising from a low, seated position (26°). We hypothesized that the increased pelvic tilt that results from the rising from a seated position may be a risk factor for posterior dislocation [35]. When combined with the various inclination and anteversion angles, the PHDD was measured in 18 different orientations for each specific implant.
All data were initially generated by measurements taken from Pro/Engineer and were compiled in an Excel spreadsheet (Microsoft, Redmond, WA, USA). We used these data for comparative analyses among all four component designs and across all acetabular and pelvic orientations tested.
Results
Within the three standard THA designs, the PHDD increased as the femoral head size increased. There was minimal difference in PHDD between the 28- and 36-mm femoral heads at lower acetabular inclination angles, but the difference became more pronounced at 60° of acetabular inclination (Fig. 4). The resurfacing 48-mm prosthesis demonstrated a more complicated relationship. At 30° of acetabular inclination, the resurfacing had the lowest PHDD and was in fact negative for all three anteversion angles. This means that the posterior edge of the cup was actually medial to the head center and this may therefore indicate a risk of edge loading. When positioned at 45° of acetabular inclination, the resurfacing cup was comparable to the 28- and 36-mm hemispherical heads; and at 60° of inclination, the resurfacing had a larger PHDD than the 28- and 36-mm heads. The 48-mm head of the dual-mobility cup consistently had the largest PHDD in all positions and both pelvic tilt angles.
This figure illustrates the effect of PHDD as a function of pelvic inclination and acetabular anteversion for each of the three acetabular inclination angles. Note the largest values are found in the dual-mobility bearing surface, regardless of inclination angle, and that the hip resurfacing had negative values at lower inclination angles, suggesting the presence of an edge-loading phenomenon.
In general, the PHDD increased as acetabular inclination angle increased. All implants demonstrated linearly increasing PHDD values as inclination angles increased from 30° to 60°. At 30° of inclination and 26° of pelvic tilt, negative values for PHDD were calculated for the resurfacing prosthesis. Depending on acetabular anteversion and pelvic tilt, the mean increase in PHDD as inclination angle increased was 8 and 10 mm for the 28- and 36-mm heads and 11 mm for the dual-mobility cup. The 48-mm resurfacing had the highest increase in PHDD with increasing inclination angle with a mean 15-mm increase, although its PHDD value was still always lower than the dual-mobility design.
For any given anteversion, an inclination angle of 30° at a standing pelvic inclination resulted in a smaller PHDD than an inclination angle of 60° when rising from a seated position (Fig. 5). Of note, the PHDD was lower (greater dislocation risk) for any given anteversion when there was a shallow inclination angle at a standing pelvic tilt (30° inclination and 5° pelvic tilt) when compared with a more severe inclination angle at the seated pelvic tilt (60° inclination and 26° pelvic tilt).
The smallest PHDD values were determined for 26° pelvic tilt and 30° of acetabular inclination. In the analysis of the effect of pelvic tilt in relation to acetabular inclination and/or anteversion, in a general sense, there was more stability with 5° of pelvic tilt than 26° for all of the prostheses tested. All implants had an overall lower PHDD for the 26° pelvic inclination angle than the 5° angle. For both pelvic inclination angles, each implant showed a linear increase in PHDD with respect to both acetabular inclination angle and acetabular anteversion.
Discussion
Jump distance has been related to THA dislocation rates and has commonly been studied in two-dimensional models. However, to the best of our knowledge, there has been little published work on the effect of bearing design, acetabular orientation, or pelvic inclination on jump distance. Because of continued problems with hip dislocation after THA, we created a model that determined that the following factors influenced PHDD: specific acetabular design, acetabular orientation, and pelvic inclination.
There were several limitations of the current study. First, this model was purely based on prosthetic characteristics and designs, and future models could incorporate bony anatomy as well as soft tissues. Despite this limitation, our model has shown that the interplay among acetabular orientation, pelvic position, femoral head size, and prosthetic design is more complex than previously understood. These data further our understanding of the sensitivity of some implant designs to component positioning but assume a purely posterior dislocation and do not account for soft tissue stabilizers. Bench validation could further validate the model, assess the effect of soft tissues and kinetic factors on stability, and determine if the prosthetic head dislocates in a purely posterior fashion (as modeled here, with dislocation modeled at 270° for simplicity of calculation) or if it follows more of a path of least resistance in the generalized posterior direction. However, we believe holding all factors constant, as we did in this model, allows us to identify general trends that can later be refined with more sophisticated cadaveric or dynamic models. Second, this model considers only a translational dislocation mechanism and does not take into account the levering mechanism that occurs from bony or prosthetic impingement. In vivo, dislocation mechanisms are more likely to be a combination of these two mechanisms (levering from impingement with translational movement of the femoral head) with additional stability provided by the soft tissues. The load provided by the patient’s body weight will also affect the in vivo mechanism of dislocation with this load possibly conferring some protective benefits when the pelvis is in certain orientations (eg, standing) and may contribute to amplify the dislocation risk when in other positions (eg, rising from a seated position). All studies such as this one may be limited by application to each patient’s unique anatomy (dysplastic hips, extraarticular femoral deformities, prior acetabular or femoral trauma, etc), which may influence component positioning and cannot be accounted for here.
Many basic science [3, 11, 14, 16, 41] and clinical studies [8, 13–15, 29, 37, 42–45] support the notion that larger head sizes correlate with increased jump distance and therefore decrease THA dislocation risk. For example, in one of the first cadaver studies on this topic by the Harris Orthopaedic Laboratory, 36-mm heads had decreased dislocation rates compared with 26- and 32-mm heads, which was attributed solely to jump distance [11]. The present study was consistent with the literature because as head size increased (28, 36, to 48 mm), jump distance correspondingly increased.
The effects of acetabular positioning and pelvic tilt demonstrated more complex, but not unsurprising, interrelationships. For example, PHDD was lower at the more provocative pelvic tilt of 26°; however, as cup inclination angle increased, anteversion played more of a protective role when comparing the two pelvic tilt values. This is in agreement with what should be intuitively concluded in that a more anteverted cup will provide greater stability against a posterior dislocation. This also suggests that, although increased amounts of anteversion and inclination may not always be optimal, they may provide protection against dislocation when performing provocative maneuvers. Interestingly, the dual-mobility design was more stable at all component positions when compared with both hemispherical cups with smaller femoral heads as well as the resurfacing-type implant with a similar, anatomically sized femoral head.
In this study, the dual-mobility hip had the highest PHDD of any design regardless of head size, which held for all acetabular orientations and pelvic inclinations. This correlates with the clinical studies with this type of device that have demonstrated almost no dislocations in the primary or revision hip arthroplasty setting [4, 9, 19, 20, 22, 26, 38, 39, 46, 47]. For example, in a study by Philippot and coauthors [39] of 384 primary THAs using a dual-mobility cup, which was similar in design to the one presently evaluated, there was no early or late instability at a mean followup of greater than 15 years (range, 12–20 years). Multiple other reports have confirmed these low dislocation rates with these designs in both the primary and revision setting [9, 19, 20, 22, 26, 39, 46, 47].
Multiple studies have reported the theoretical advantages of hip resurfacing for reducing dislocation rates [1, 22, 36]. However, in a recent systematic review comparing resurfacing versus standard THA in young active patients, the authors found no differences in dislocation rates [22]. This may be partly the result of the less than hemispherical head coverage and/or because of the low head-neck ratio that might lead to earlier prosthetic and bony impingement [5, 24]. In the present study, resurfacing showed increased posterior dislocation distances when compared with 28- and 32-mm heads but these were lower than the dual-mobility design. The negative PHDD values indicate that the posterior wall of the resurfacing cup was medial to the head center, implying a risk for edge loading during chair rise with low inclination angles. There have been several reports of high cup inclination angles leading to edge loading and increased wear of metal-on-metal hip arthroplasties [17, 27, 30, 32, 41, 49]. Based on the present study, it also appears that excessively closed cup positions may also lead to negative wear characteristics.
When evaluating jump distance, most studies have used purely mathematical techniques for assessments in either two or three dimensions coupled with characterization of typically one other factor such as acetabular abduction angle or head size [14, 41]. Recently, Sariali et al. [41] evaluated lateral jump distance according to implant characteristics, increasing head size, and head offset as well as a function of cup anteversion and abduction angle. They found that the jump distance decreased as cup abduction angles increased and increased with increasing anteversion angles. Additionally, there were increases in jump distance with increased head diameters, an improvement that decreased with increases in abduction angles. They also found head offset was the most important parameter influencing jump distance (increased offset reduced jump distance). However, similar to the Crowninshield et al. [14] study, their model ultimately resulted in a vertical dislocation mechanism. One must question the results obtained from these studies because dislocations clinically occur in either the anterior or, more commonly, the posterior direction.
In summary, we have described a new model to assess three-dimensional posterior jump distance (posterior horizontal dislocation distance). We assessed various acetabular designs and found that the dual-mobility socket with anatomic rim had the greatest jump distance. Increasing head size, decreasing acetabular inclination, and decreasing acetabular anteversion had positive influences on increased jump distance. Pelvic orientation of 26° reduced jump distance for all prostheses and acetabular orientations, which supports the hypothesis that chair rise is a high-risk activity for dislocation. The clinical importance of our findings is that newer designs may be useful in preventing hip dislocation and may be appropriate for patients who are at increased risk for dislocation in difficult primary situations and in revision procedures.
References
Barrack RL. Dislocation after total hip arthroplasty: implant design and orientation. J Am Acad Orthop Surg. 2003;11:89–99.
Barrack RL, Butler RA, Laster DR, Andrews P. Stem design and dislocation after revision total hip arthroplasty: clinical results and computer modeling. J Arthroplasty. 2001;16:8–12.
Bartz RL, Nobel PC, Kadakia NR, Tullos HS. The effect of femoral component head size on posterior dislocation of the artificial hip joint. J Bone Joint Surg Am. 2000;82:1300–1307.
Bauchu P, Bonnard O, Cypres A, Fiquet A, Girardin P, Noyer D. The dual-mobility POLARCUP: first results from a multicenter study. Orthopedics. 2008;31. pii: orthosupersite.com/view.asp?rID=37180.
Beaule PE, Harvey N, Zaragoza E, Le Duff MJ, Dorey FJ. The femoral head/neck offset and hip resurfacing. J Bone Joint Surg Br. 2007;89:9–15.
Beaule PE, Schmalzried TP, Udomkiat P, Amstutz HC. Jumbo femoral head for the treatment of recurrent dislocation following total hip replacement. J Bone Joint Surg Am. 2002;84:256–263.
Berry DJ. Unstable total hip arthroplasty: detailed overview. Instr Course Lect. 2001;50:265–274.
Berry DJ, von Knoch M, Schleck CD, Harmsen WS. Effect of femoral head diameter and operative approach on risk of dislocation after primary total hip arthroplasty. J Bone Joint Surg Am. 2005;87:2456–2463.
Bouchet R, Mercier N, Saragaglia D. Posterior approach and dislocation rate: A 213 total hip replacements case-control study comparing the dual mobility cup with a conventional 28-mm metal head/polyethylene prosthesis. Orthop Traumatol Surg Res. 2011;97:2–7.
Bozic KJ, Kurtz SM, Lau E, Ong K, Vail TP, Berry DJ. The epidemiology of revision total hip arthroplasty in the United States. J Bone Joint Surg Am. 2009;91:128–133.
Burroughs BR, Rubash HE, Harris WH. Femoral head sizes larger than 32 mm against highly cross-linked polyethylene. Clin Orthop Relat Res. 2002;405:150–157.
Bystrom S, Espehaug B, Furnes O, Havelin LI. Femoral head size is a risk factor for total hip luxation: a study of 42,987 primary hip arthroplasties from the Norwegian Arthroplasty Register. Acta Orthop Scand. 2003;74:514–524.
Conroy JL, Whitehouse SL, Graves SE, Pratt NL, Ryan P, Crawford RW. Risk factors for revision for early dislocation in total hip arthroplasty. J Arthroplasty. 2008;23:867–872.
Crowninshield RD, Maloney WJ, Wentz DH, Humphrey SM, Blanchard CR. Biomechanics of large femoral heads: what they do and don’t do. Clin Orthop Relat Res. 2004;429:102–107.
Cuckler JM, Moore KD, Lombardi AV Jr, McPherson E, Emerson R. Large versus small femoral heads in metal-on-metal total hip arthroplasty. J Arthroplasty. 2004;19:41–44.
D’Lima DD, Urquhart AG, Buehler KO, Walker RH, Colwell CW Jr. The effect of the orientation of the acetabular and femoral components on the range of motion of the hip at different head-neck ratios. J Bone Joint Surg Am. 2000;82:315–321.
De Haan R, Pattyn C, Gill HS, Murray DW, Campbell PA, De Smet K. Correlation between inclination of the acetabular component and metal ion levels in metal-on-metal hip resurfacing replacement. J Bone Joint Surg Br. 2008;90:1291–1297.
DeWal H, Su E, DiCesare PE. Instability following total hip arthroplasty. Am J Orthop (Belle Mead NJ). 2003;32:377–382.
Guyen O, Pibarot V, Vaz G, Chevillotte C, Bejui-Hugues J. Use of a dual mobility socket to manage total hip arthroplasty instability. Clin Orthop Relat Res. 2009;467:465–472.
Hamadouche M, Biau DJ, Huten D, Musset T, Gaucher F. The use of a cemented dual mobility socket to treat recurrent dislocation. Clin Orthop Relat Res. 2010;468:3248–3254.
Hedlundh U, Ahnfelt L, Hybbinette CH, Wallinder L, Weckstrom J, Fredin H. Dislocations and the femoral head size in primary total hip arthroplasty. Clin Orthop Relat Res. 1996;333:226–233.
Jiang Y, Zhang K, Die J, Shi Z, Zhao H, Wang K. A Systematic review of modern metal-on-metal total hip resurfacing vs standard total hip arthroplasty in active young patients. J Arthroplasty. 2011;26:419–426.
Kelley SS, Lachiewicz PF, Hickman JM, Paterno SM. Relationship of femoral head and acetabular size to the prevalence of dislocation. Clin Orthop Relat Res. 1998;355:163–170.
Kluess D, Zietz C, Lindner T, Mittelmeier W, Schmitz KP, Bader R. Limited range of motion of hip resurfacing arthroplasty due to unfavorable ratio of prosthetic head size and femoral neck diameter. Acta Orthop. 2008;79:748–754.
Kurtz SM, Ong KL, Schmier J, Mowat F, Saleh K, Dybvik E, Karrholm J, Garellick G, Havelin LI, Furnes O, Malchau H, Lau E. Future clinical and economic impact of revision total hip and knee arthroplasty. J Bone Joint Surg Am. 2007;89(Suppl 3):144–151.
Lautridou C, Lebel B, Burdin G, Vielpeau C. [Survival of the cementless Bousquet dual mobility cup: Minimum 15-year follow-up of 437 total hip arthroplasties] [in French]. Rev Chir Orthop Reparatrice Appar Mot. 2008;94:731–739.
Lee RK, Essner A, Longaray J, Wang A. Metal-on-metal bearings: the problem is edge-loading wear. Surg Technol Int. 2010;20:303–308.
Lewinnek GE, Lewis JL, Tarr R, Compere CL, Zimmerman JR. Dislocations after total hip-replacement arthroplasties. J Bone Joint Surg Am. 1978;60:217–220.
Lombardi AV Jr, Skeels MD, Berend KR, Adams JB, Franchi OJ. Do large heads enhance stability and restore native anatomy in primary total hip arthroplasty? Clin Orthop Relat Res. 2011;469:1547–1553.
Mak M, Jin Z, Fisher J, Stewart TD. Influence of acetabular cup rim design on the contact stress during edge loading in ceramic-on-ceramic hip prostheses. J Arthroplasty. 2011;26:131–136.
Masonis JL, Bourne RB. Surgical approach, abductor function, and total hip arthroplasty dislocation. Clin Orthop Relat Res. 2002;405:46–53.
Matthies A, Underwood R, Cann P, Ilo K, Nawaz Z, Skinner J, Hart AJ. Retrieval analysis of 240 metal-on-metal hip components, comparing modular total hip replacement with hip resurfacing. J Bone Joint Surg Br. 2011;93:307–314.
McGrory BJ, Morrey BF, Cahalan TD, An KN, Cabanela ME. Effect of femoral offset on range of motion and abductor muscle strength after total hip arthroplasty. J Bone Joint Surg Br. 1995;77:865–869.
Morrey BF. Instability after total hip arthroplasty. Orthop Clin North Am. 1992;23:237–248.
Nadzadi ME, Pedersen DR, Yack HJ, Callaghan JJ, Brown TD. Kinematics, kinetics, and finite element analysis of commonplace maneuvers at risk for total hip dislocation. J Biomech. 2003;36:577–591.
Nikolaou V, Bergeron SG, Huk OL, Zukor DJ, Antoniou J. Evaluation of persistent pain after hip resurfacing. Bull NYU Hosp Jt Dis. 2009;67:168–172.
Peters CL, McPherson E, Jackson JD, Erickson JA. Reduction in early dislocation rate with large-diameter femoral heads in primary total hip arthroplasty. J Arthroplasty. 2007;22:140–144.
Philippot R, Adam P, Reckhaus M, Delangle F, Verdot FX, Curvale G, Farizon F. Prevention of dislocation in total hip revision surgery using a dual mobility design. Orthop Traumatol Surg Res. 2009;95:407–413.
Philippot R, Camilleri JP, Boyer B, Adam P, Farizon F. The use of a dual-articulation acetabular cup system to prevent dislocation after primary total hip arthroplasty: analysis of 384 cases at a mean follow-up of 15 years. Int Orthop. 2009;33:927–932.
Robbins GM, Masri BA, Garbuz DS, Greidanus N, Duncan CP. Treatment of hip instability. Orthop Clin North Am. 2001;32:593–610, viii.
Sariali E, Stewart T, Jin Z, Fisher J. In vitro investigation of friction under edge-loading conditions for ceramic-on-ceramic total hip prosthesis. J Orthop Res. 2010;28:979–985.
Seyler TM, Etienne G, Plate JF, Fisher P, Mont MA. Use of modular large femoral heads without liners in hip arthroplasty. Surg Technol Int. 2006;15:217–220.
Skeels MD, Berend KR, Lombardi AV Jr. The dislocator, early and late: the role of large heads. Orthopedics. 2009;32. pii: orthosupersite.com/view.asp?rID=42837.
Smith TM, Berend KR, Lombardi AV Jr, Emerson RH Jr, Mallory TH. Metal-on-metal total hip arthroplasty with large heads may prevent early dislocation. Clin Orthop Relat Res. 2005;441:137–142.
Stuchin SA. Anatomic diameter femoral heads in total hip arthroplasty: a preliminary report. J Bone Joint Surg Am. 2008;90(Suppl 3):52–56.
Tarasevicius S, Busevicius M, Robertsson O, Wingstrand H. Dual mobility cup reduces dislocation rate after arthroplasty for femoral neck fracture. BMC Musculoskelet Disord. 2010;11:175.
Vielpeau C, Lebel B, Ardouin L, Burdin G, Lautridou C. The dual mobility socket concept: experience with 668 cases. Int Orthop. 2011;35:225–230.
von Knoch M, Berry DJ, Harmsen WS, Morrey BF. Late dislocation after total hip arthroplasty. J Bone Joint Surg Am. 2002;84:1949–1953.
Walter WL, Insley GM, Walter WK, Tuke MA. Edge loading in third generation alumina ceramic-on-ceramic bearings: stripe wear. J Arthroplasty. 2004;19:402–413.
Author information
Authors and Affiliations
Corresponding author
Additional information
Three of the authors (JN, CH, JM) certify that they are employees of Stryker Orthopaedics (Mahwah, NJ, USA). Two of the authors (MAM, DCM) certify that they have or may receive payments or benefits, in any one year, an amount in excess of $10,000 from Stryker Orthopaedics in connection with the submitted article. The remaining author (AJJ) certifies that he, or a member of his immediate family, has no commercial associations that might pose a conflict of interest in connection with the submitted article. No institutions received funding from any commercial entity in support of this work.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research editors and board members are on file with the publication and can be viewed on request.
Clinical Orthopaedics and Related Research neither advocates nor endorses the use of any treatment, drug, or device. Readers are encouraged to always seek additional information, including FDA-approval status, of any drug or device prior to clinical use.
This work was performed at either Stryker Orthopaedics, Mahwah, NJ, USA, or at Sinai Hospital of Baltimore, Baltimore, MD, USA
About this article
Cite this article
Nevelos, J., Johnson, A., Heffernan, C. et al. What Factors Affect Posterior Dislocation Distance in THA?. Clin Orthop Relat Res 471, 519–526 (2013). https://doi.org/10.1007/s11999-012-2559-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-012-2559-1