Abstract
Quality and shelf life of filleted hot smoked rainbow trout packaged in vacuum packaging (untreated) and with thyme oil (TO), and garlic oil (GO)-treated, after vacuum-packaged (VP) conditions stored at 2 °C, were investigated by measurement of microbiological, sensory and physicochemical analyses. Acceptability scores for appearance, odour, taste and texture of untreated and treated smoked trout decreased with storage time. The limit of sensory acceptance was reached after 5 weeks for the untreated samples, after 7 weeks for with thyme oil-treated samples (TO + VP) and after 6 weeks for with garlic oil-treated (GO + VP) samples. Total viable count evaluated that thyme oil- and garlic oil-treated vacuum-packaged samples had good results up to 7 weeks compared with 5 weeks in control vacuum packs. Psychrotrophic counts for untreated and treated samples exceeded 6 log cfu/g after 5 and 6 weeks of cold storage, respectively. Treated samples presented the lowest lactic acid bacteria counts. Total volatile basic nitrogen and trimethylamine nitrogen values gave acceptable results for up to 5 weeks for the untreated samples, 7 weeks for thyme- and garlic oil-treated samples. Treated with thyme oil samples presented the lowest thiobarbituric acid index values. The results obtained from this study showed that the shelf life of hot smoked rainbow trout stored in cold storage (2 °C), as determined by overall acceptability of all data, is 5 weeks for vacuum-packaged samples and 7 weeks for thyme- and garlic oil-treated vacuum-packaged smoked fish.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Smoked fish is a highly nutritious food containing highly unsaturated fatty acids, fat soluble vitamins, essential minerals as well as proteins containing the amino acids essentials for human beings (Bilgin et al. 2008). Smoked fish has characteristic flavour and colour. Hot smoked fish products made from white fish generally keep better than those made from fatty fish, although shelf life will vary considerably, depending on the amounts of salt and smoke present, the degree of drying and the storage temperature (Bannermann 2001). Generally, hot smoked fish stored in anaerobic conditions (vacuum-packaged) at chilled temperature is very sensitive to deterioration and, based on sensory evaluation, has a limited shelf life ranging from 3 to 4 weeks at refrigerator temperature (Rehbronn and Rutkowski 1982). Lowering of quality is manifested through changes in appearance, odour, colour or texture (Tülsner 1994; Kolsarıcı and Özkaya 1998; Cakli et al. 2006). Hot smoked fish, especially hot smoked rainbow trout, is of considerable economic importance worldwide, particularly in Europe. Improvements in the shelf life of hot smoked fish product can have an important economic feedback by reducing losses attributed to spoilage and by allowing the products to reach distant and new markets.
Spices, seeds, and their oils or extract application has proven to be an effective preservation method for the extension of shelf life of fresh fish (Harpaz et al. 2003; Quitral et al. 2009; Kykkidou et al. 2009). Thyme, oregano, sage, black seed, grape seed, garlic, rosemary oils have natural antimicrobial and antioxidant properties with the potential to extend the shelf life of foods (Burt 2004; Erkan and Bilen 2010; Erkan et al. 2010).
The effect of thyme and garlic oil (1%: v/w) in cold storage on vacuum-packaged hot smoked rainbow trout fillets’ microbiological status, sensory and chemical characteristics was studied using analysis.
Materials and Methods
Raw Material
Essential oils were purchased from the Ecem Natural Company (Istanbul, Turkey). Undiluted 100% oil of thyme and garlic oil was used. Hot smoked fish was prepared from rainbow trout (Oncorhynchus mykiss). Rainbow trout (212.5 ± 2.12 g and 26 ± 1.2 cm) were obtained from a freshwater fish farm of Bağcı in Turkey, in Mai 2008. The fresh fish samples were packed in polystyrene boxes with crushed ice and then transferred to the laboratory. The fish were harvested, beheaded, gutted manually and then washed. The samples (33 gutted fishes) were immersed in brine at a ratio of 1:1 (w/w) for 24 h at 2 °C. The brine contained 10% NaCl. After brining, the samples were briefly dipped in chilled tap water for 1 h.
Smoking Process
Smoke was produced from oak sawdust with combustion. The processing time in the kiln was divided into two stages: (1) a preliminary drying and cooking period (45 min) at 75 °C; (2) a smoking and partial cooking period at 80 °C (60 min). After cooling at room temperature (60 min), the smoked fish were filleted and divided into three lots.
Essential Oil Treatment, Packaging and Storage of Hot Smoked Rainbow Trout
The following lots of samples were prepared: The first lot of samples comprised the controls (samples in the control treatment had no essential oils added). Thyme oil (TO; second lot) and garlic oil (GO; third lot) were added onto the surface (two sides) of each fillet using a micropipette so as to achieve a final 1% (v/w) concentration. The volume of thyme and garlic oil was chosen based on information in the literature regarding storage of bluefish and chub mackerel (Erkan and Bilen 2010; Erkan et al. 2010). The control (VP) and oil-treated (TO + VP and GO + VP) hot smoked rainbow trout (three filets for each group of each analysis week) were placed in high-barrier plastic film bags and packaged under vacuum-packaged. Samples were stored in a refrigerator (2 ± 1 °C) and analysed once a week during 7 weeks to determine the shelf life.
Packaging Material and Apparatus
Plastic film bags were obtained from UPM-Kymmene, Walki-Pack, Plastic Films Factory (Valkeakoski, Finland). The characteristics of the plastic film bags were as follows: transmission rate (mL/m2): O2, 6.89; CO2, 5.42; N2, 2.48, at 4 °C; vapour permeability, 7.86 mL/m2 at 37.8 ± 1 °C, 90 ± 2% RHg/m2 days atm. Samples were packed in a Komet vacuum-packaging machine PAXX model (Germany).
Analyses
Microbiological Analyses
Sample Preparation
Smoked fish (25 g) obtained from each smoked fish fillet were transferred aseptically to a Stomacher bag (Seward Medical, London, UK) containing 225 mL of 0.1% peptone water (Merck, Cat No. 107228) and homogenised for 60 s using a Lab Blender 400, Stomacher at high speed (Stomacher, IUL Instrument, Spain).
Microbiological Media and Count
For microbial count, 0.1-mL samples of serial dilutions (1:10, diluents, 0.1% peptone water; Merck, 107228) of fish homogenates were spread on the surface of agar plates. Total viable counts (TVC) were determined using plate count agar (Merck, 105463) after incubation for 24–48 h at 37 °C. Plate count agar was used for psychrotrophic bacteria and incubated at 7 °C for 10 days. Lactic acid bacteria were enumerated on MRS medium (Merck, 110660) and incubated at 37 °C for 48 h (Baumgart 1986). Results are expressed as a logarithm of colony forming units (log cfu) per gram of sample.
Sensory Analysis
The attributes of hot smoked fish were evaluated by a panel of five experienced judges on each week of sampling. Sensory evaluation was conducted in individual booths under controlled conditions of light, temperature and humidity. Sensory analysis was performed using the methods of Amerina et al. (1965). Smoked fish were assessed on the basis of appearance, odour, taste and texture characteristics using a nine-point descriptive scale. A score of 9–7 indicated “very good” quality; a score of 6.9–4.0, “good or acceptable quality”; and a score of 3.9–1.0, “unacceptable quality”.
Physicochemical Analysis
The skin of smoked fish were removed for the physicochemical analysis and was homogenised using foot processor.
Chemical Analysis
One gram of each groups (three fillet) were mixed with 10 mL distilled water. The pH of the fish homogenate was measured using a digital pH meter Hanna pH 211 Microprocessor pH meter (HANNA Instruments, Michigan, USA), standardised at pH 4.0 and 7.0 (Erkan 2007). Total volatile basic nitrogen (TVB-N, mg/100 g fish flesh) was determined according to the method described by Antonacopoulos and Vyncke (1989). Trimethylamine nitrogen (TMA-N, mg/100 g fish flesh) was measured using the method of AOAC (1998), while the method used by Erkan and Özden (2008) was followed for the thiobarbituric acid (TBA) index values were expressed as mg of malondialdehyde (MDA)/kg of sample. The concentration of malondialdehyde was calculated from a standard curve using solutions of the MDA precursor (same molecular weight) 1,1,3,3-tetraethoxy-propane into distilled water after the addition of a quantity of TBA solution.
Colour Measurement
The colour of the fish samples was determined with the help of a Konica Minolta chromo meter (model CR 400/410; Minolta, Osaka, Japan). L* (brightness), a* (+a, red; −a, green) and b* (+b, yellow; −b, blue) values were measured. The colorimeter was calibrated using white references (CR-A44). The fish muscles (three fillets) were homogenised and placed in glass petri dishes (12 cm diameter), and the colour measurement was repeated three times using different parts of the surface. The average of these values for L*, a* and b* parameter was calculated (Gerdes and Santos Valdez 1991).
Statistical Analysis
All analytical determinations were at weeks 1, 2, 3, 4, 5, 6 and 7. Experiments were replicated twice on different occasions with different fish samples. The mean each sample for each group was analysed three times (\( n = {\hbox{replicate}} \times {\hbox{group}} \times {\hbox{three}}\;{\hbox{times}} \); \( n = 2 \times 3 \times 3 \)). Results are reported as mean values ± standard deviation (SD). Data were subjected to analysis of variance. The Tukey’s honestly significant difference procedure was used to test for differences between means (P < 0.05) (Sümbüloğlu and Sümbüloğlu 2002).
Results and Discussion
Assessment of Microbiological Parameters
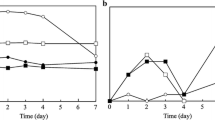
Changes in TVC of refrigerated hot smoked rainbow trout fillets during storage under vacuum packaging with or without thyme and garlic oil are shown in Fig. 1a. Low initial (week 1) TVC of hot smoked rainbow trout fillets of approximately 3.00 cfu/g (VP), 2.78 cfu/g (TO + VP) and 2.88 log cfu/g (GO + VP) indicates good fish quality, in agreement with results (3.3 log cfu/g) reported by Cakli et al. (2006) for vacuum-packaged rainbow trout fillets. At the end of shelf life after 7 weeks at 2 °C, respectively, levels of 6.65, 5.40 and 5.10 log cfu/g were reached. TVC is an important criterion for quality evaluation; the maximum recommended bacterial count for good-quality products is 5.7 log cfu/g, and the maximum recommended bacterial count for marginally acceptable quality products is 7 log cfu/g (ICMSF 1986). Considering these values, it is possible to say that TVCs of vacuum-packaged smoked rainbow trout (untreated) exceeded the microbiological limits of acceptability after 4 weeks of storage. This limit was not exceeded throughout storage in TO + VP and GO + VP samples. Thus, compared with the control samples (VP), a shelf life extension of 3 weeks was achieved for samples TO + VP and GO + VP, respectively, as determined by microbiological parameters (TVC). The thyme and garlic oil can be considered effectively inhibitory on the total aerobic flora.
Initial populations of psychrotrophic bacteria of VP, TO + VP and GO + VP samples were 2.88, 2.78 and 2.66 log cfu/g, respectively. Psychrotrophic bacteria count reached final counts of 7.47, 7.18 and 6.90 log cfu/g, respectively, for VP, TO + VP and GO + VP hot smoked trout samples (Fig. 1b). Interestingly, TO + VP and GO + VP smoked samples, on 5 weeks of storage, had significantly lower (p < 0.05) counts than had the control samples. VP and TO + VP/GO + VP smoked trout samples exceeded the value of 6 log cfu/g for psychrotrophic bacteria count, considered as the upper acceptability limit for fish and fish products (Erkan 2007), after weeks 4 and 6 of storage, respectively. Thus, compared with the control samples (VP), a shelf life extension of 2 weeks was achieved for samples TO + VP and GO + VP, respectively, as determined by psychrotrophic bacteria counts.
Lactic acid bacteria (LAB) were dominant in vacuum-packed foods. Initial counts were approximately 1.50 log cfu/g (LAB), indicating a good hygiene of the marine environment from which the fish were caught, as well as good fishing practices and handling. Final populations of LAB (6.32, 5.94 and 5.24 log cfu/g) were recorded for treatments V, TO + VP and GO + VP, respectively (Fig. 1c). Giatrakou et al. (2008) and Kykkidou et al. (2009) reported that thyme–oregano oil treatment was effective in eliminating the growth of TVC and LAB in modified atmosphere packaged fish under refrigerated storage. Likewise, Kostaki et al. (2009) reported that the treatment of fresh fish fillets (sea bass) with a mixture of thyme oil reduced the final TVC and LAB counts by approximately 1 log, compared with control. They are also in agreement with those reported by Chouliara et al. (2007), who reported a reduction in growth of aerobic bacteria by 5 log cfu/g in chicken meat after 9 days of storage under 1% oregano oil and air packaging.
Assessment of Sensory Acceptance
The results of the sensory evaluation (overall appearance, texture, odour and taste) of smoked rainbow trout samples are presented in Fig. 2a–d. Overall appearance, texture, odour and taste scores for hot smoked samples stored under vacuum conditions, with thyme and garlic essential oil, showed a similar pattern of decreasing acceptability. The spoilage patterns described by the panelists were as follows: softening of texture before off-odours developed and presence of bitter and rancid off-flavours. Appearance score decreased as colour discolouration increased.
The limit of overall appearance was reached after 5 weeks for the control and after 7 weeks for GO + VP samples. The limit of acceptability for texture and odour was reached after 6 and 7 weeks of storage for control and thyme–garlic oil-treated samples, respectively. The limit of acceptability for taste was reached after 4 weeks for VP samples, while this limit was reached after 6 weeks for GO + VP and after 7 weeks for TO + VP samples. Based on appearance, texture, odour and taste scores, a shelf life of approximately 5 weeks for the VP samples, 7 weeks TO + VP samples and 6 weeks GO + VP may be expected. Autolytic enzymes can have a major impact on the loss of textural quality of smoked fish during the early stages of deterioration, but they did not produce the characteristic off-flavours and off-odours, which are typical of microbiological activity (Truelstrup et al. 1996). Lactic acid bacteria have been reported to be potential spoilage organisms in smoked fish products, and lactic acid bacteria are responsible off-flavours and off-odours (Leroi et al. 1998). Yanar et al. (2006) reported a shelf life for hot smoked catfish of 5 weeks, while Yanar (2007) reported a shelf life for wrapped hot smoked catfish stored at refrigerator temperature of 3.5 weeks. The shelf life of hot smoked vacuum-packaged rainbow trout stored in a refrigerator condition was found to be 5 weeks (Cakli et al. 2006).
The combination of vacuum and garlic–thyme oil resulted in a shelf life extension of approximately 1–2 weeks, attributed to the antimicrobial effects of the thyme oil phenolic components thymol and carvacrol, and garlic oil components, allicin, known to exert antimicrobial activity (Holley and Patel 2005). Likewise, Giatrakou et al. (2008), Kostaki et al. (2009), and Kykkidou et al. (2009) reported that the treatment of fresh fish (sword fish and sea bass) fillets with a mixture of oregano and thyme oil extended the shelf lives by approximately 5 days, compared with the control.
Assessment of Physicochemical Parameters
At the beginning of the storage period, pH values of hot smoked rainbow trout were determined as 5.83. pH values of control and treated samples showed increase and decrease during the storage. At the end of the storage period of 7 weeks, pH values of samples were found to be 5.58, 5.91 and 5.48, respectively (Fig. 3a). Similar results have been reported by Kolsarıcı and Özkaya (1998) for hot smoked trout under vacuum conditions. The pH first increases after decreases are in agreement with the findings of Erkan et al. (2007), for chub mackerel stored in vacuum.
The TVB-N may be considered as a quality index for fish. Its increase is related to the activity of spoilage bacteria and endogenous enzymes (Erkan et al. 2007). At the beginning of storage, the TVB-N value was 23.22 mg/100 g flesh, which was approximately similar to the values reported for hot smoked rainbow (18.55 mg/100 g), for cold smoked rainbow trout (20.00 mg/100 g) stored in a refrigerator (Kolsarıcı and Özkaya 1998) and for hot smoked catfish (17.67 mg/100 g) stored in a refrigerator, reported by Yanar (2007). As shown in Fig. 3b, the TVB-N value of samples untreated and treated with thyme and garlic essential oil vacuum-packaged trout rose to 33.82, 24.16 and 26.74 mg/100 g flesh by the end of the storage period. With regard to data on the limit of highest acceptable level of TVB-N for fresh fish, Huss (1988) proposed a TVB-N value of 30–35 mg/100 g whilst, recently, Giménez et al. (2002) proposed a value of 25 mg/100 g as the onset of spoilage for rainbow trout. In the present study, the TVB-N level showed fluctuations during storage, indicating that TVB-N could not be a good indicator of rainbow trout quality, as also proposed by Chytiri et al. (2004) and Rezaei et al. (2008) for rainbow trout. On the basis of the present data (total viable count and sensory evaluation), a similar TVB-N limit value of 32.09 mg/100 g may be proposed for the initiation of vacuum-packaged hot smoked trout spoilage. Hot smoked trout samples VP, TO + VP and GO + VP exceeded the proposed (in our study) upper TVB-N acceptability limit on weeks 5, 7 and 7, respectively, values that could be assigned as the physicochemical (using TVB-N as a parameter) shelf life of hot smoked trout samples under the experimental conditions of the present study. The inhibition of TVB-N production in second and third groups smoked rainbow trout samples may be attributed to the effects of VP and/or with the combined synergistic effect of thyme and garlic oil, the latter known to possess antibacterial properties due to its phenolic (carvacrol and thymol) and organosulphur (allicin) constituents (Burt 2004).
TMA is produced by the decomposition of trimethylamine oxide due to bacterial spoilage and enzymatic activity (Tülsner 1994). The initial TMA-N values of samples were found 0.96 mg/100 g. TMA-N values of control and TO + VP/GO + VP samples increased significantly (p < 0.05) with storage time; by the end of the storage period (week 7), however, samples vacuum-packaged with thyme and garlic oil reached a significantly (p < 0.05) lower TMA-N value of 1.28 and 1.04 mg/100 g, respectively, in comparison with the samples of controls, which attained higher levels of 1.99 mg/100 g (Fig. 3b). Lower production of TMA-N in TO + VP and GO + VP hot smoked samples may be due to the antibacterial properties of thyme and garlic oil. TMA-N is not a particularly useful indicator of fresh water fish (trout) freshness (Tülsner 1994). Given the great variation in TMA-N acceptability limits for various fish species reported, and TMA-N values shown in Fig. 3, as well as data based on sensory (overall acceptance) scores (5 weeks) and microbiological (TVC) data (5 weeks), a more realistic TMA-N limit of acceptability for hot smoked rainbow trout, of ca. 2.00 mg/100 g, may be proposed. Similarly, low TMA-N values have been reported for hot smoked rainbow trout Cakli et al. (2006). On the basis of this limit, VP, TO + VP and GO + VP hot smoked rainbow trout fillets exceeded this value on weeks 5, 7 and 7, respectively, which could be used to mark the shelf life of hot smoked trout fillets. Lower production of TMA-N in TO + VP and GO + VP smoked fish samples may be due to the antibacterial properties of thyme and garlic oil. Interestingly, vacuum packaging thyme–garlic oil treatment (combination of packaging and oil) effectively controlled the population of TVC to less than 7 log cfu/g during refrigerated storage of smoked fish (Fig. 1a).
Lipid oxidation is a major quality problem especially in fatty marine species. It leads to the development of off-odours and off-tastes in edible oils and fat containing foods, known as oxidative rancidity. The TBA index value is an index of lipid oxidation measuring MDA content. MDA is formed through hydroperoxides, which are the initial reaction products of polyunsaturated fatty acids with oxygen (Rezaei et al. 2008). Changes in TBA values for VP, TO + VP and GO + VP hot smoked trout samples during storage are shown in Fig. 3d. The initial TBA index value for hot smoked rainbow trout fillets was 0.77 mg MDA/kg in agreement with Yanar (2007) and Cakli et al. (2006) who reported a TBA index value for hot smoked tilapia and rainbow trouts of 0.84 and 1.4 mg MDA/kg. In the case of vacuum-packaged smoked fish, the TBA index value increased to a maximum during storage up to the third week and decreased. Similar results were determined in TO + VP and GO + VP samples. Lower production of TBA index in TO + VP hot smoked samples may be due to the antioxidant properties of thyme oil. The decrease in TBA index values may represent the breakdown of the malondialdehyde to tertiary degradation. Similar results have been obtained in hot smoked tilapia (Yanar et al. 2006) and hot smoked trout Cakli et al. (2006). The TBA values for TO + VP and GO + VP were significantly lower (P < 0.05) than those for VP, indicating that the TO and GO treatments used in TO + VP and GO + VP inhibited lipid oxidation. The use of thyme oil to protect muscle foods against oxidation has been reported in the literature. Mariutti et al. (2008) and Erkan and Bilen (2010) observed that a garlic oil and thyme oil was an effective means of controlling lipid oxidation in chicken and fish meat, as reflected in thiobarbituric acid reactive substance values.
Colour is an important parameter (Du and Sun 2008). Colour values of the samples can be seen in Fig. 4a–c. At the beginning of the storage, L* values of VP, TO + VP and GO + VP hot smoked samples were determined as 72.89 ± 2.85. In the TO + VP and GO + VP samples, L* values increased according to time of storage. At the end of the storage period of 7 weeks, L* values reached 78.98 ± 0.79 and 76.78 ± 2.77 for TO + VP and GO + VP, respectively. According to the results on the seventh week of storage, vacuum-packaged hot smoked fish flesh became much lighter. At the beginning of the storage period, a* values for samples were determined as 6.84 ± 1.58. At the end of the storage period of 7 weeks, a* values of VP samples increased to 10.35 ± 0.73 and a* values of TO + VP and GO + VP samples decreased to 2.78 ± 0.31 and 0.27 ± 0.22, respectively. b* values of VP and TO + VP samples were increased at the end of the storage, whereas GO + VP were decreased at the end of the storage. Chouliara et al. (2007) reported similar in L*, a* and b* values for oregano essential oil and modified atmosphere packaging in fresh chicken meat during 12 days of storage.
Conclusions
In the present study, a shelf life of 7 weeks was recorded for hot smoked trout treated with thyme and garlic oil in cold storage, compared with a shelf life of 5 weeks for the untreated hot smoked rainbow trout based on sensory, chemical and microbiological evaluation.
References
Amerina, M. A., Pangborn, R. V., & Roessler, E. B. (1965). Principles of sensory evaluation of food. New York: Academic.
Antonacopoulos, N., & Vyncke, W. (1989). Determination of volatile basic nitrogen in fish. Zeitschrift fur Lebensmittel-Untersuchung und -Forschung, 189, 309–316.
AOAC. (1998). AOAC Official Method 971.14, trimethylamine nitrogen in seafood colorimetric method. In J.M. Hungerford (Chapter Ed.) Fish and other marine products. In P. Cunniff (Ed.) Official methods of analysis of AOAC International, chapter 35 (p. 7). Gaithersburg: AOAC International.
Bannermann, Mc K. A. (2001). Hot smoking of fish. Ministry of Agriculture, Fisheries and Food Torry Research Station. Torry Advisory Note No: 82 (revised).
Baumgart, J. (1986). Lebensmittel tierischer Herkunft, Feinkosterzeugnisse, gefrorene, tiefgefrorene und getrocknete lebensmittel, Fertiggerichte, hitzekonservierte Lebensmittel, Speiseeis, Zucker, Kakao, Zuckerwaren, Rohmassen. Mikrobiologische Untersuchung von Lebensmitteln. Edt: Jürgen Baumgart, unter Mitarbeit von Jürgen Firnhaber, Gottfried Spicher. p. 207, Behr’s Verlag. ISBN: 3-922528-91-0 Hamburg.
Bilgin, Ş., Ünlüsayın, M., İzci, L., & Günlü, A. (2008). The determination of the shelf life and some nutritional components of gilthead sea bream (Sparus aurata L., 1758) after cold and hot smoking. Turkish Journal of Veterinary and Animal Sciences, 32(1), 49–56.
Burt, S. (2004). Essential oils: Their antibacterial properties and potential applications in foods—A review. International Journal of Food Microbiology, 94, 223–253.
Cakli, Ş., Kılınc, B., Dincer, T., & Tolasa, S. (2006). Comparison of the shelf lifes of map and vacuum packaged hot smoked rainbow trout (Onchoryncus mykiss). European Food Research and Technology, 224(1), 19–26.
Chouliara, E., Karatapanis, A., Savvaidis, I. N., & Kontominas, M. G. (2007). Combined effect of oregano essential oil and modified atmosphere packaging on shelf-life extension of fresh chicken breast meat, stored at 4 °C. Food Microbiology, 24, 607–617.
Chytiri, S., Chouliara, I., Savvaidis, I. N., & Kontominas, M. G. (2004). Microbiological, chemical and sensory assessment of iced whole and filleted aquacultured rainbow trout. Food Microbiology, 21, 157–165.
Du, C. J., & Sun, D. W. (2008). Retrospective shading correlation of confocal laser scanning microscopy beef images for three-dimensional visualization. Food and Bioprocess Technology, 2, 167–176.
Erkan, N. (2007). Freshness and quality of aquacultured sea bass (Dicentrarchus labrax) and sea bream (Sparus aurata) stored in ice. Archiv für Lebensmittelhygiene, 58(3), 98–106.
Erkan, N., & Bilen, G. (2010). Effect of essential oils treatment on the frozen storage stability of chub mackerel fillets. Journal für Verbraucherschutz und Lebensmittelsicherheit, 5, 101–110.
Erkan, N., & Özden, Ö. (2008). Quality assessment of whole and gutted sardine (Sardine pilchardus) stored in ice. International Journal of Food Science & Technology, 43(9), 1549–1559.
Erkan, N., Özden, Ö., & İnuğur, M. (2007). The effects of modified atmosphere and vacuum packaging on quality of chub mackerel. International Journal of Food Science & Technology, 42(11), 1297–1304.
Erkan, N., Tosun, Ş. Y., Ulusoy, Ş., & Üretener, G. (2010). The use of a thyme and laurel essential oil treatments to extend the shelf life of bluefish (Pomatomus saltatrix) during storage in ice. Journal für Verbraucherschutz und Lebensmittelsicherheit. doi:10.1007/s00003-010-0587-x.
Gerdes, D. L., & Santos Valdez, C. (1991). Modified atmosphere packaging of commercial Pacific red snapper (Sebastes entomelas, Sebastes flavidus or Sebastes godei). Lebensmittel. -Wissenschaft und Technology, 24, 256–258.
Giatrakou, V., Kykkidou, S., Papavergou, A., Kontominas, M. G., & Savvaidis, I. N. (2008). Potential of oregano essential oil and MAP to extend the shelf life of fresh swordfish: A comparative study with ice storage. Journal of Food Science, 73(4), M167–M173.
Giménez, B., Roncalés, P., & Beltrán, J. A. (2002). Modified atmosphere packaging of filleted rainbow trout. Journal of Science and Food Agricultural, 84, 1154–1159.
Harpaz, S., Glatman, L., Drabkin, V., & Gelman, A. (2003). Effects of herbal essential oils used to extend the shelf-life of freshwater reared Asian sea bass fish (Lates calcarifer). Journal of Food Protection, 66, 410–417.
Holley, R. A., & Patel, D. (2005). Improvement in shelf-life and safety of perishable foods by plant essential oils and smoke antimicrobials. Food Microbiology, 22, 273–292.
Huss, H. H. (1988). Fresh fish: Quality and quality changes. Rome: Food and Agriculture Organization (FAO) of the United Nations. 132 p.
International Commission on Microbiological Specifications for Foods (ICMSF). (1986). Sampling plans for fish and shellfish. In: Microorganisms in foods. Sampling for microbiological analysis: Principles and scientific applications, vol. 2, 2nd edition (pp. 181–196). Toronto: University of Toronto Press.
Kolsarıcı, N., & Özkaya, Ö. (1998). Gökkusağı Alabalıgı (Salmo gairdneri)’nın Raf Ömrü Üzerine Tütsüleme Yöntemleri ve Depolama Sıcaklığının Etkisi. Turkish Journal of Veterinary and Animal Science, 22, 273–284.
Kostaki, M., Giatrakou, V., Savvaidis, I. N., & Kontominas, M. G. (2009). Combined effect of MAP and thyme essential oil on the microbiological, chemical and sensory attributes of organically aquacultured sea bass (Dicentrarchus labrax) fillets. Food Microbiology, 26, 475–482.
Kykkidou, S., Giatrakov, V., Papavergou, A., Kontominas, M. G., & Savvaidis, I. N. (2009). Effect of thyme essential oil and packaging treatments on fresh Mediterranean swordfish fillets during storage at 4 °C. Food Chemistry, 115, 169–175.
Leroi, F., Joffraud, J. J., Chevailer, F., & Cardinal, M. (1998). Study of the microbial ecology of cold-smoked salmon during storage at 8 °C. International Journal of Food Microbiology, 39, 111–121.
Mariutti, L. R. B., Orlien, V., Bragagnolo, N., & Skibsted, L. H. (2008). Effect of sage and garlic on lipid oxidation in high-pressure processed chicken meat. European Food Research and Technology, 227(2), 337–344.
Quitral, V., Donoso, M. L., Ortiz, J., Herrera, M. V., Araya, H., & Aubourg, S. P. (2009). Chemical changes during the chilled storage of Chilean jack mackerel (Trachurus murphyi): Effect of a plant-extract icing system. LWT Food Science and Technology, 42, 1450–1454.
Rehbronn, E., & Rutkowski, F. (1982). Das Räuchern von Fischen. Berlin: Verlag Paul Parey. ISBN:3-490-26614-5.
Rezaei, M., Hosseini, S. F., Langrudi, H. E., Safari, R., & Hosseini, S. V. (2008). Effect of delayed icing on quality changes of iced rainbow trout (Onchorynchus mykiss). Food Chemistry, 106, 1161–1165.
Sümbüloğlu, K., & Sümbüloğlu, V. (2002). Biyoistatistik. 10. Baskı, p.275, Hatipoğlu Basım ve Yayım San. Tic. Ltd. Sti. ISBN: 975-7527-12-2. Ankara.
Truelstrup, L., Gill, T., Drewes, S., & Huss, H. (1996). Importance of autolysis and microbiological activity on quality of cold-smoked salmon. Food Research International, 29(2), 181–188.
Tülsner, M. (1994). Fischverarbeitung. Bd.1 –Rohstoffeigenschaften von Fisch und Grundlagen der Verarbeitungsprozesse. Behr’s Verlag Hamburg. ISBN: 3-86022-196-5.
Yanar, Y. (2007). Quality changes of hot smoked catfish (Clarias gariepinus) during refrigerated storage. Journal of Muscle Foods, 18, 391–400.
Yanar, Y., Çelik, M., & Akamca, E. (2006). Effects of brine concentration on shelf-life of hot-smoked tilapia (Oreochromis niloticus) stored at 4 °C. Food Chemistry, 97, 244–247.
Acknowledgment
This work was supported by the Research Fund of Istanbul University with the projects 1208 and BYP 7341.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Erkan, N. The Effect of Thyme and Garlic Oil on the Preservation of Vacuum-Packaged Hot Smoked Rainbow Trout (Oncorhynchus mykiss). Food Bioprocess Technol 5, 1246–1254 (2012). https://doi.org/10.1007/s11947-010-0412-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-010-0412-7