Abstract
The etiology and pathogenesis of attention-deficit/hyperactivity disorder (ADHD) are unclear and a more valid diagnosis would certainly be welcomed. Starting from the literature, we built an hypothetical pyramid representing a putative set of biomarkers where, at the top, variants in DAT1 and DRD4 genes are the best candidates for their associations to neuropsychological tasks, activation in specific brain areas, methylphenidate response and gene expression levels. Interesting data come from the noradrenergic system (norepinephrine transporter, norepinephrine, 3-methoxy-4-hydroxyphenylglycol, monoamine oxidase, neuropeptide Y) for their altered peripheral levels, their association with neuropsychological tasks, symptomatology, drugs effect and brain function. Other minor putative genetic biomarkers could be dopamine beta hydroxylase and catechol-O-methyltransferase. In the bottom, we placed endophenotype biomarkers. A more deep integration of “omics” sciences along with more accurate clinical profiles and new high-throughput computational methods will allow us to identify a better list of biomarkers useful for diagnosis and therapies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The aetiology and pathogenesis of attention-deficit/hyperactivity disorder (ADHD) is not yet fully understood [1, 2]. ADHD is also a highly heritable disease, with estimated heritability rates of up to 80 % [3]. A recent review on genetics of ADHD summarized that all variants associated with the pathology explain only a small fraction of heritability: phenotype complexity and variants of small effect contribute to the missing heritability issue [4••]. To gain more insight into the mechanisms leading from a genetic/biological basis of the disease to the full clinical phenotype, endophenotypes are a promising strategy [5].
The DSM and ICD diagnoses of ADHD are based on a consensus about clusters of clinical symptoms. However, this diagnostic procedure has been criticized for not allowing sufficiently reliable and valid diagnoses [6]. NIMH recently has approved the Research Domain Criteria (RDoC) project where a set of assumptions permit to found a new classification system, by integrating genetics, imaging and cognitive information [6]. The RDoC approach suggests that a biomarker approach to diagnosis may be a more valid way to classify complex mental disorders such as ADHD. Biomarkers offer the opportunity to standardize and improve diagnostic assessment while providing insights into etiological mechanisms.
What is a Biomarker?
The United States food and drug administration (FDA) defines a biomarker as an objective measure of normal processes, pathological processes or pharmacological response [7]. For psychiatry, biomarkers could be used to screen for, diagnoses, or predict the development of, not only psychiatric disorders but also of personality or behavioral traits and RDoC domains. Biomarkers could potentially be used to inform treatment decisions.
Because no single biomarker candidate may be sufficient for accurate and reliable diagnosis, the current trend in psychiatry has shifted towards identifying sets of biomarkers [8, 9••, 10••, 11]. For example, Pies et al. defined a set of biomarkers for schizophrenia comprising:-neuregulin-1 mutations; -abnormal smooth-pursuit eye movements; -reduced anterior cingulate volumes, enlarged lateral and third ventricular volumes and white matters abnormalities [12].
Despite their promise, the search for biomarkers of psychiatric disorders has largely proven elusive. In a comprehensive review [10••], the authors concluded that according to their stringent criteria, no biomarker is available for diagnosing ADHD.
The main goal of this review is to present evidence for a putative signature set of biomarkers for ADHD using genomic, neurophysiological/neuropsychological, neuroimaging, pharmacogenomic and proteomic knowledge to diagnose ADHD. In Table 1 we report data from genomics and metabolomics classified according to neurophysiological/neuropsychological, neuroimaging, pharmacogenomic, biochemical and gene expression findings. Table 2 presents data from endophenotypes studies to define a further potential signature set of biomarkers useful for ADHD diagnosis.
Potential Dopaminergic Biomarkers for ADHD
Genomics
Deficiencies of dopaminergic and noradrenergic neurotransmitter systems have been associated with ADHD and the main symptoms of ADHD, but the dopamine system has been the main focus of genomic research [69].
Dopamine Transporter Gene (DAT1, SLC6A3)
There are at least four main reasons for a central role of DAT1 in ADHD genetics: 1) dopaminergic neurotransmission is controlled by the DAT1 protein; 2) the DAT1 protein is the main target for two ADHD medications: methylphenidate (MPH) and amphetamine (AMP); 3) knockout mice for DAT1 show hyperactivity and deficits in inhibitory behavior [69]; 4) DAT1 has been mapped near to a susceptibility locus for ADHD, 5p13 [70]. The most studied DAT1 variant is a variable number of tandem repeats (VNTR) of 40 base pairs located at the 3′-untranslated region (3′-UTR) of the gene. The ten repeat (10R) and nine repeat (9R) alleles are the most common. It has been demonstrated that the DAT density measured in vivo using neuroimaging was significantly higher in ADHD children with homozygosity for the 10R in basal ganglia, a brain area participating in inhibitory behaviors. This suggests a functional effect of the 10R allele [71]. A recent meta-analysis demonstrated an association between the 10R allele and ADHD [13], although a reverse association has been seen for ADHD adults [72].
Kebir and Joober and Barnes et al. summarized DAT1 studies of neuropsychological tasks [21, 22]. As regards the association between attention tasks and the 10R allele, some studies showed higher commission errors, more impulsive responses and greater reaction time variability (RTV) using the continuous performance test (CPT) and sustained attention to response test (SART). Other studies reported no association or opposite results [21]. Moreover boys scoring high on a teacher-rated report of ADHD symptoms and who were homozygous for the 10R allele displayed poorer response inhibition on the opposite word task. Concerning executive functions, the 10/10 genotype predicted better performance on the Tower of London (TOL), the self-order pointing task, and the Wechsler Intelligence Scale for Children-III (WISC-III) arithmetic and digit span subtests. Associations between a neurocognitive phenotype-spatial attentional bias and 10R allele was reported in children with ADHD [73, 74]. The attentional phenotype (left-sided inattention) was related to higher symptom severity [74].
Several studies have been conducted on neuroimaging and 3′UTR VNTR variant in ADHD. Genotype 10/10 individuals were found to generate more activations corresponding to inhibition tasks, showing also its interaction with diagnostic status (ADHD vs. controls) [15]. Moreover on non-ADHD siblings of ADHD probands and controls, an influence was observed between the variant and the activation in striatum and cerebellum vermis, with its interaction with familial risk for ADHD [18]. Similarly in another study, 10/10 individuals were more activated in left striatum, right dorsal premotor cortex, and bilateral temporoparietal junction [20]. In contrast, Congdon et al. observed greater neural activation associated with carriers of the 9R [23]. Finally caudate volumes were overall smaller in 10/10 than 9/10 children, particularly in the left than right hemisphere [14].
Positron emission tomography (PET) and single photon emission tomography (SPECT) have been used to determine if ADHD patients show abnormal activity of the DAT in striatum. In a meta-analysis of these studies, Fusar-Poli et al. concluded that DAT activity was 14 % higher in ADHD patients compared with controls [24]. A meta-analysis of neuroimaging studies of humans found that the 9R allele was associated with increased DAT activity in striatum [75]. This study was conducted considering different neuropsychiatric illnesses and this strengths the issue that VNTR polymorphism regulates dopamine activity in the striatal brain regions not only in presence of ADHD but also of other disorders
A recent review on neuroimaging and pharmacogenetics reported some studies on DAT1 [25••]. In particular an association with 10R was found with higher regional cerebral blood flow in medial frontal and left basal ganglia regions, and with poorer treatment response, in children with ADHD receiving MPH for 4 days before SPECT imaging session. Moreover this allele was associated with greater DAT availability in basal ganglia following treatment and poorer treatment response in children with ADHD who completed 8 weeks of MPH treatment. In particular only a 28.6% of the subjects with 10/10 genotype showed good response to MPH treatment [71].
Pharmacogenetic studies have demonstrated increased response to MPH among homozygous 10R, but there are conflicting results [16•, 17••]. However a recent meta-analysis [19••] shows that there is no significant association between this VNTR and response to MPH treatment. The meta-analysis also found no effects on dimensions of hyperactivity/impulsivity and inattention. Thus this finding suggests that VNTR polymorphism is not a reliable predictor of MPH treatment success in ADHD, neither in relation to these specific symptom subscales.
A DAT1 VNTR at intron-8 that contains common 5-repeat (5R) and 6-repeat (6R) alleles has also been associated with ADHD susceptibility [13]. Recent evidence suggests that joint consideration in haplotypes may provide more information than can be inferred from the analyses of single genetic markers. There are some neurophysiological studies on the haplotypes of these two DAT1 variants: the 10/6 haplotype (formed by the 10R allele of the 3’UTR VNTR and the 6R allele of the intron 8 VNTR) is associated with dopaminergic reward-processing circuitry whose deficits are linked to ADHD [26]. The haplotype 10/3 (formed by the 10R allele of the 3’UTR VNTR and the 3R allele of the intron 8 VNTR) predicts spatial inattention suggesting a functional effect of this haplotype on cognitive performance in ADHD [27].
Other variants of DAT1 have been studied. rs6350 was associated to alerting and executive control performance using the attention network test [29]. Moreover a haplotype (rs403636 (G)/rs463379 (C)/rs393795 (C)/rs37020 (G)) was associated with spatial working memory in ADHD [28].
Dopamine D4 Receptor (DRD4)
The DRD4 gene is a good candidate for ADHD because of its high expression in brain regions implicated in attention and inhibition such as anterior cingulate cortex [76]. DRD4 was the first associated with novelty-seeking, a personality trait which is a common in ADHD. It has been demonstrated that mRNA expression levels of DRD4 were lower in ADHD [31].
DRD4 is located on chromosome 11p15.5. A highly polymorphic functional VNTR in the third exon has been frequently investigated in association studies. It comprises 11 copies of a 48-bp repeat sequence, where 4, 7, and 2R repeat alleles are the most prevalent. A meta-analysis showed that the 7R allele was associated with ADHD [13, 30••].
Recent reviews on neuropsychological endophenotypes [21, 22] concluded that, although more studies are needed, the 7R allele is associated with some cognitive markers such as speed of processing, set shifting, and cognitive impulsiveness, but not with response inhibition (the stop and go/no-go tasks). More robust evidence suggests that the absence of this allele is linked to high reaction time variability (RTV).
Neuroimaging studies found associations of 7R allele with thinner prefrontal and parietal cortex as well as with delay in cerebral cortical maturation [32, 33••].
Pharmacogenetic studies suggest an enhanced response to MPH among 7R carriers even though conflicting results have been obtained [16•, 17••]. Moreover some studies have suggested that different DRD4 genotypes exhibit different MPH dose–response curves [16•, 17••]. One study demonstrated that individuals with one or two copies of the 4R allele had positive linear improvement on mathematics test outcomes with increasing MPH dose, while those lacking a copy showed greater improvement at lower doses, and deterioration at higher doses. A second study found that those lacking any 4R allele had decreasing responses to higher MPH doses compared with other genotypes. Both these studies are consistent with the idea that the 7R codes for a defective dopamine receptor that responds less effectively at higher doses, an idea with some potential for clinical relevance if replicated in larger samples. One interesting finding suggests that DRD4 might play a role in predicting susceptibility to medication side effects. In the Preschool ADHD Treatment Study, MPH treated ADHD children homozygous for the 4R were three-times more likely to develop picking behaviors, while those with at least one copy of the 7R were four-times more likely to develop social withdrawal with increasing doses. Thus, 7R carriers appear to be a distinct subgroup of ADHD patients which experiences differential medication effects.
Other variants in the promoter region have also been studied, focusing on a 120-base pair duplication (120-bp dup), −521C/T (rs1800955), −616C/G (rs747302), −615A/G, and −376C/T, located in the 5′ untranslated region. However two meta-analyses conducted on 120-bp dup and −521C/T showed no association with ADHD [13, 30••].
Dopamine D2 receptor (DRD2)
The DRD2 gene is located at chromosome 11q23.1. The rationale for its involvement in ADHD is based on the evidence that it is expressed in those brain areas relevant for the pathology and it is implicated in the regulation of mesolimbic reward pathways [77]. A recent genome wide association study reported a nominal association of this gene with ADHD susceptibility [34]. The TaqIA (rs1800497) variant of DRD2 was believed to connect with urinary level of the DA metabolite homovanillic acid (HVA) and DRD2 expression levels. According to Wu et al.’s meta-analysis, an association of this polymorphism was reported [30••]. However, due to excessive heterogeneity, the authors concluded that this result was invalid. Pharmacogenetic studies have found no association of this variant with MPH response [35]. Two other DRD2 variants, rs2075654 and rs1079596, have been associated with greater commission error in ADHD [21].
Dopamine D5 Receptor (DRD5)
DRD5 maps to chromosome 4p15.3. In situ hybridization studies demonstrated that the expression of the gene is higher in hippocampus, a brain area involved in ADHD pathogenesis [78]. Moreover functional studies showed that DRD5 is implicated in synaptic strength in hippocampal memory formation [79]. These studies support the rationale on the involvement of DRD5 gene in ADHD. The association between ADHD and a highly polymorphic dinucleotide repeat of DRD5 ((CA)n), located in 18.5 kb at the end of the 5′ flanking region, has been the most studied. This variant comprises 12 alleles ranging from 134 to 156 bps in length, among which the 148-bp and 136-bp alleles are the most common. The 148-bp allele was a risk factor for ADHD according meta-analyses, while that of 136-bp allele was a protective factor for ADHD [13, 30••].
Interestingly, the 148 bp allele associated to commission errors, omission errors, RTs, and RTV [36], whereas no association was detected with cognitive test scores [38]. Pharmacogenetic studies found no association of MPH response with the 148-bp allele, whereas the 151-bp allele has been linked to a favorable MPH response [37].
Metabolomics
Phenylethylamine (PEA)
In dopaminergic neurons of the nigrostriatal system, the dopamine (DA) receptor agonist PEA is synthesized by the decarboxylation of phenylalanine. PEA stimulates the release of DA and, according to a meta-analysis [39••], urinary levels of PEA are significantly lower in patients with ADHD compared with controls. Interestingly decreased levels of PEA have also been associated with symptoms of inattentiveness and administration of AMP and MPH markedly increased urinary excretion of PEA [39••].
DA and HVA
DA and its main metabolite HVA showed no difference in urine excretion in ADHD patients compared with controls [39••].
Potential Noradrenergic Biomarkers for ADHD
Genomics
Norepinephrine Transporter Gene (NET1, SLC6A2)
The SLC6A2 gene codes for the norepinephrine transporter, which is responsible for the reuptake of norepinephrine (NE) from the synaptic cleft back into the presynaptic neuron and is targeted by atomoxetine, a medication for ADHD. NET1 is most highly expressed in the frontal lobes where it plays a role in noradrenergic and dopaminergic reuptake [80]. A frequently studied SNP (rs5569 or G1287A) located in exon 9 was not significant in a meta-analysis of candidate gene studies [13], but a nominal association was observed in a genome-wide association study [34]. No association was found between this variant and CPT performance [21]. A recent review on pharmacogenetics concluded that NET1 influence on response to MPH is small [17••], although a recent genome-wide association study found two SNPs in SLC6A2 gene associated with MPH response [81]. In a study rs3785143 was associated with atomoxetine response [47].
Moreover children with the G/G genotype at G1287A showed a greater decrease in mean omission error scores after MPH administration compared to A allele carriers. Another polymorphism A-3081T at 5’flank, demonstrated to be functional, was associated with T allele with greater decrease in mean commission errors scores, meaning improved impulsive behavior [40, 41]. Other variants such as rs998424 and rs3785157 were linked to high performance on the similarities subtest of the WISCIII, but not with the cerebral volume or thickness of the cortex [42]. Another SNP rs3785155 was associated to RTV [43].
Metabolomics
Norepinephrine (NE)
Concerning NE levels, a meta-analysis [39••] indicated higher urinary levels in ADHD compared with controls, and a normalization of NE concentrations has been observed after treatment with polyphenol complex and with fenfluramine and AMP [39••].
3-Methoxy-4-Hydroxyphenylglycol (MHPG)
MHPG is the main metabolite of NE. Its urinary levels were lower in ADHD patients and stimulant trials showed that decreases in ADHD symptoms with treatment were associated with greater reductions in urinary MHPG excretion [39••].
Neuropeptide Y (NPY)
NPY frequently colocalizes with catecholamine systems. It participates in the regulation of feeding, circadian rhythms, reproduction, and thermoregulation. Oades et al. found increased plasma NPY concentrations in ADHD children compared with controls [82]. Moreover a recent genome-wide copy number variation analysis [44] found that NPY was included in a rare 3 Mb duplication on chromosome 7p15.2 to 15.3 and an association of this duplication was found with increased NPY plasma concentrations. Moreover an association was observed between gene dose-dependent increases in NPY and emotion processing [44].
Potential Adrenergic Biomarkers for ADHD
Genomics
Alpha-2A-adrenergic receptor (ADRA2A)
ADRA2A in the prefrontal cortex influences executive functions impaired in ADHD [83]. It is also a target for two ADHD medications: guanfacine and clonidine. A-1291 C>G creates an MspI site in the promoter region and it is a functional polymorphism. Two meta-analyses confirmed no association with ADHD [13, 45]. However the C allele was associated with TOL, trail making test (TMT), and RTV on the stop-signal task as well as the CC genotype was associated with high RTV on the CPT test [21]. A recent pharmacogenetic review [17••] summarized some studies reporting that G allele is associated with greater reduction of inattentive symptoms over time. Froehlich et al. also found a main effect of this genetic variant on MPH response [46]. However, the G allele was associated with hyperactive-impulsive symptoms on placebo and across doses.
Another variant rs553668, for which the disorder meta-analysis was negative [13], was associated with the performance on TMT suggesting an involvement of this gene on executive functions [21]. Yang et al. described an association between GG haplotype (rs1800544/rs553668) and non-remission of ADHD symptoms with atomoxetine treatment [47].
Metabolomics
Epinephrine (EPI) and Metanephrine (M)
EPI and its main metabolite M levels, according to a recent meta-analysis [39••], were respectively no different or higher in ADHD patients. The difference for M was lost after Bonferroni correction.
Potential Metabolism Enzymes as Biomarkers for ADHD
Genomics/Metabolomics
Catechol-O-Methyltransferase (COMT)
COMT is an enzyme responsible for the degradation of DA and NE. It is highly expressed in frontal lobe where it regulates synaptic DA levels [84]. Studies examining the association between COMT and ADHD have largely focused on a functional SNP in exon 4 that creates an amino acid substitution (valine/methionine). This variant affects COMT enzyme activity, such that homozygotes for the valine allele shows 3–4 times greater activity than homozygotes for the methionine allele. However meta-analysis [48••] indicates no association between ADHD and the 158Val/Met.
A review summarized studies on 158Val/Met and neuropsychological endophenotypes [21]. While no association was observed between this variant and performance on cognitive tasks, a significant finding was reported between the Met allele and impairment in sustained attention and fewer commission errors. In the neurophysiological studies, this variant was associated with antisocial and aggressive behaviors of ADHD [50, 51]. Pharmacogenetic studies reported a positive association between Val allele and response to MPH [52, 53].
Monoamine Oxidase A (MAOA)
The MAOA gene encodes a protein involved in the metabolism of DA, 5-HT, and NE. It has been associated with impulsive, aggressive behavior [85]. Moreover an MAOA knockout mouse showed increased levels of aggressive behavior and monoaminergic neurotransmitter levels [86]. Recent studies have focused largely on a functional 30-bp VNTR 1.2 kb upstream of the gene, previously associated with impulsivity and aggression. According the classification system [87], the 2- and 3R are considered ‘low-activity’ alleles, the others are high-activity’ alleles. A recent meta-analysis reported no association with ADHD [13]. According to neuropsychological studies, the 4 and 5R alleles are associated with commission errors on the TOVA test, and a MPH administration attenuated this association [54].
Moreover a gender-specific association has been reported with the ATT haplotype (rs12843268/rs3027400/rs1137070) [55]. In boys, the ATT haplotype was associated with motor control; in girls with visuo-spatial working memory. This suggests that MAOA genotype influences cognitive and motor functioning in ADHD and sex moderates these effects. This may be related to biological differences between males and females in serotonin neurotransmission. Finally Nymberg et al. reported a sex-specific association between ADHD symptomatology and reward deficiency or insufficient response inhibition depending on rs12843268 genotype [56]. Concerning blood levels, a recent meta-analysis indicated reduced levels in ADHD compared with controls [39••]. MAO levels were associated with impulsiveness and MPH treatment normalized the reduced levels.
Dopamine Beta Hydroxylase (DBH)
The DBH gene encodes an enzyme that catalyzes the conversion of DA into NE which is particularly expressed in the prefrontal cortex [88]. A–1021C/T variant (rs1611115) accounts for up to 50 % of the variation of plasma DBH activity, but it is the intron 5 TaqI variant (rs2519152) that has been most often tested. A meta-analyses of these variants reported no association with ADHD [13].
Two recent reviews of neuropsychological endophenotypes summarized studies of DBH [21, 22]. In particular the A2/T allele was found associated with poorer performances on a temporal order judgment task, more commission and omission errors and greater RTV on the SART, more errors on the Wisconsin Card Sorting Test (WCST) (problem-solving) and the Matching Familiar Figures Test (MFFT) (cognitive impulsiveness).
Finally an association between neuropsychological measures of executive function in children with ADHD and the –1021C/T variant has been reported [57].
A meta-analysis [39••] found that ADHD patients showed lower activities of DBH in serum and urine and it was suggested that decreased DBH levels correlate with ADHD symptoms [39••, 89, 90••].
Potential CNS Development Biomarkers for ADHD
Genomics/Metabolomics
Brain Derived Neurotrophic Factor (BDNF)
BDNF has been mapped to chromosome 11p13. It belongs to a family of neurotrophins, involved in promoting neurogenesis, neuronal survival, and synaptic plasticity [91]. A genome wide study supported a nominal association with ADHD susceptibility [34]. A common variant resulting in a valine to methionine amino acid substitution at codon 66 (Val66Met; rs6265) regulates the intracellular tracking and activity-dependent secretion of BDNF in brain. A meta-analysis found no association of this variant with ADHD [13]. This variant and two others, rs2049046 and rs11030104, were not associated with the Digit Span test [21]. Moreover better MPH response in ADHD children who are Val/Val homozygous was reported [59].
Concerning peripheral levels, higher plasma concentrations were observed in ADHD patients as compared with controls [58], whereas no alterations were observed in serum levels [60].
Synaptosomal-Associated Protein 25 (SNAP-25)
SNAP-25 codes for a protein involved in axonal growth, synaptic plasticity, and in the docking and fusion of synaptic vesicles in presynaptic neurons necessary for the regulation of neurotransmitter release [92]. The major variants studied were rs362987 (intron 4), rs363006 (intron 6), rs3746544 (3′UTR), and rs1051312 (3′UTR). A main effect on ADHD susceptibility was observed only with rs3746544 [13].
Concerning pharmacogenetic studies, homozygotes for the T allele of T1065G had moderately improved MPH responses, whereas homozygotes for the G allele developed sleep difficulties and irritability. Moreover, homozygotes for the T allele at T1069C exhibited poorer MPH response, whereas homozygotes for the C allele developed tics and other abnormal movements [37].
Potential Biomarkers for Environmental Risk factors
Metabolomics
Iron
It is a coenzyme of tyrosine hydroxylase and MAO, which are critical in the synthesis and degradation, respectively. Ferritin is a major intracellular iron storage protein and serum ferritin levels are an indication of iron stores in the body and brain. Children with iron deficiency exhibited ADHD symptoms, such as inattention, hyperactivity, and/or impulsivity [review 90••]. A meta-analysis indicated that serum ferritin levels were lower for ADHD patients compared with controls, although the finding was lost after Bonferroni correction [39••].
Zinc
Zinc is another essential cofactor for neurotransmitter metabolism which affects DA metabolism. Zinc deficiency is associated with behavior problems in children with ADHD [review 90••]. Although a systematic review of randomized controlled clinical trials demonstrated that using zinc, either alone or in combination with stimulants, did not improve ADHD symptoms [93], a meta-analysis reported reduced serum/plasma/urine levels in ADHD [39••].
Oxidative Stress
The normal oxidation-reduction reactions that create energy in the cell create toxic oxidants or reactive oxygen species. These by-products of normal oxidation-reduction reactions are highly unstable. Antioxidants counteract the effects of oxidants. When antioxidants are not sufficient, oxidative stress occurs and damages cellular proteins, lipids, carbohydrates, and nucleic acids. Brain tissue is especially susceptible to oxidative stress. Indirect evidence for oxidative stress in ADHD comes from studies showing some treatment efficacy for anti-oxidant compounds such as omega-3 fatty acids, pycnogenol and N-acetylcysteine [94–97].
A meta-analysis of oxidative stress in ADHD suggested that patients with ADHD have normal levels of anti-oxidant production but that their response to oxidative stress is insufficient, leading to oxidative damage [61••].
Potential Endophenotypes as Biomarkers for ADHD
Besides indentifying a genetic marker involved in neurophysiological/neuropsychological, neuroimaging, pharmacogenetics, biochemical features, a putative signature set of biomarkers could be integrated with a research on endophenotypes (Table 2).
EEG Theta/Beta Ratio (TBR)
TBR is a measurement of the power (wave amplitude squared) produced by brain waves altering between 3.5 and 28.0 Hz in children with ADHD [98]. A genetic overlap between ADHD symptoms and EEG theta power has been reported [99] and this supports a recent review suggesting TBR as a potential biomarker for ADHD [63]. However a recent meta-analysis concluded that excessive TBR cannot be considered a reliable diagnostic measure of ADHD but it could have a prognostic value [62••]. In June 2013 FDA approved Neuropsychiatric EEG-Based Assessment Aid (NEBA) System, a test recording the type and number of brain waves that nerve cells give off each second.
In summary, as also reported in Thome et al. the neurophysiological markers must be viewed as promising candidates for biomarkers of ADHD but further studies are needed [10••].
Reaction Time Variability (RTV)
Individuals with ADHD are described frequently as ubiquitously slower and more variable than their unaffected peers, and ADHD-related RTV is considered by many to reflect a unique, stable, and etiologically important characteristic of the disorder. Results of a recent meta-analysis indicate that, contrary to contemporary characterizations of individuals with ADHD as slower and more variable, ADHD individuals may be better characterized as more variable but not slower after accounting for their increased response variability. However RTV lacks specificity among clinical disorders, and thus is not a viable diagnostic marker of ADHD [100••].
Selective Attention and Vigilance/Sustained Attention
A prominent multidimensional model of attention differentiates between alertness, divided attention, selective attention, vigilance/sustained attention and shifting. According to Thome et al. the most robust findings indicated that children with ADHD displayed difficulties in measures of selective attention and vigilance/sustained attention [10••]. Impairments of vigilance/sustained attention are the most replicated neuropsychological finding in ADHD. However a considerable number of investigations were unable to find difference between ADHD patients and controls [64–66]. In addition effect sizes indicated only small to moderate differences between ADHD patients and controls.
Executive Functions
They are an umbrella term encompassing various functions of higher cognitive functioning including planning, problem solving, concept formation, fluency, cognitive flexibility, and working memory. The most consistent finding in the domain of executive functions is that patients with ADHD display impairments in working memory. Working memory measures appear therefore to be the most sensitive indicator of executive dysfunctioning in ADHD [10••]. Additional studies support this finding [67, 68]. However, as for vigilance/sustained attention, no difference was found between patients and controls with small to moderate effect sizes [64–66].
In summary, although promising results were obtained for selective attention and vigilance/sustained attention and working memory, no sensitive or specific profile based on neuropsychological tests assessing cognition in ADHD has as yet been found [101••].
Brain Differences in ADHD
Kasparek et al. reported alterations in several areas of the brain, particularly the anterior cingulum, the dorsolateral as well as ventrolateral prefrontal cortex, the orbitofrontal cortex, the superior parietal regions, the caudate nucleus, the thalamus, the amygdala and the cerebellum. Imaging studies point to the persistence of changes in both brain structure and function into adulthood, although there might be a tendency for improvement of caudate nucleus pathology. Growing evidence suggests that MPH treatment can lead to improvement of brain changes seen in neuroimaging by its positive effect on neuroplasticity [102••].
Conclusion
The study of psychiatric biomarkers is very complex because of the heterogeneous nature of psychiatric disorders which are often referred to as syndromes with several subtypes, not uniform problems. Therefore, a single biomarker is very unlikely to provide enough information to identify cellular and metabolic pathways involved in a particular individual. Identification of a signature set of biomarkers for disorder subtypes each based on their underlying biological pathways will be the most effective for diagnosis and treatment selection.
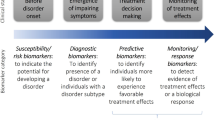
Starting from the literature, we have built an hypothetical pyramid representing a putative set of biomarkers where at the top there are potentially useful biomarkers for ADHD and at the bottom those that are probably not useful (Fig. 1). Following Pies et al.’s [12] example, a potential genetic-biomarker could be represented by the variants in DAT1 and DRD4 genes as the best candidates as biomarkers for ADHD. Indeed the 10R allele at the 3′UTR of SLC6A3, is associated with specific neuropsychological tasks [21, 22], generates more activation in specific brain areas [33••], and is associated to MPH response [16•, 17••]. Similarly the 7R allele at exon 3 of DRD4 is involved in specific neuropsychological tasks [21, 22], brain structure [32, 33••], MPH response [16•, 17••], and expression levels of DRD4 [31].
Hypothetical pyramid representation of possible signature sets of biomarkers for ADHD diagnosis. Starting from the literature, we built an hypothetical pyramid describing a putative set of biomarkers where, at the top, variants in DAT1 and DRD4 genes are the best candidates as useful biomarkers, for their associations to neuropsychological tasks, activation in specific brain areas, methylphenidate response and gene expression levels. A further level is represented by the noradrenergic system (Norepinephrine transporter, Norepinephrine, 3-methoxy-4-hydroxyphenylglycol, Monoamine Oxidase, Neuropeptide Y) for their altered peripheral levels, their association with neuropsychological tasks, symptomatology, drugs effect and brain function. Other minor putative genetic biomarkers could be Dopamine Beta Hydroxylase and Catechol-O-methyltransferase. In the bottom, we placed endophenotype biomarkers. In particular working memory, selective attention vigilance/sustained attention, Theta/beta ratio, Reaction Time Variability and neuropsychological endophenotypes are, in this order, probably not useful biomarkers for ADHD diagnosis. Notes: Dopamine Transporter gene (DAT1, SLC6A3); Dopamine D4 receptor (DRD4); Norepinephrine transporter (NET1, SLC6A2); Norepinephrine (NE); 3-methoxy-4-hydroxyphenylglycol (MHPG); Monoamine oxidase (MAO); Neuropeptide Y (NPY); Dopamine beta hydroxylase (DBH); Catechol-O-methyltransferase (COMT); Theta/beta ratio (TBR); Reaction time variability (RTV)
Interesting data also come from the noradrenergic system. In fact, according to metabolomic studies, NET1, NE, its metabolite MHPG and MAO along with NPY could represent good candidates as biomarkers for ADHD, based on their peripheral levels altered in ADHD [39••], their association with neuropsychological tasks [21, 40–43, 54–56], symptomatology, drugs effect [39••], and brain function [44].
Other minor putative genetic biomarkers could be the two enzymes DBH and COMT. Although the meta-analyses of Taq1 and -1021 T/C in DBH gene and 158Val/Met in COMT gene have not identified susceptibility alleles, variants of these genes are associated with neuropsychological performance [21, 22, 57], neurophysiological features [50, 51], brain function [103], peripheral levels [39••, 89], and MPH response [52, 53].
Concerning the identification of endophenotype biomarkers, to date it is not possible to characterize an accurate biomarker for ADHD. As described in [10••, 101••, 104], no biomarker has sufficiently high specificity and sensitivity for the ADHD diagnosis. Currently proposed biomarkers are limited by age, gender, and drug effects and studies have been limited by small sample sizes and poor control for multiple testing.
This work supports the necessity to take into account the deep integration of “omics” sciences such as “pharmacogenomics”, “phenomics”, “epigenomics,” “proteomics”, “transcriptomics”, and “metabolomics”. In fact, a better understanding of the interaction network of genes, proteins, and biochemical processes in relation to more accurate clinical profiles, by using new high-throughput computational methods, will allow us to identify a list of biomarkers both for the optimization of diagnostic assessment as well as for the personalization of therapies.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Biederman J, Faraone SV. Attention-deficit hyperactivity disorder. Lancet. 2005;366(9481):237–48.
Faraone SV. Attention-deficit hyperactivity disorder and the shifting sands of psychiatric nosology. Br J Psychiatr. 2013;203(2):81–3.
Faraone SV, Mick E. Molecular genetics of attention deficit hyperactivity disorder. Psychiatr Clin N Am. 2010;33(1):159–80.
Akutagava-Martins GC, Salatino-Oliveira A, Kieling CC, Rohde LA, Hutz MH. Genetics of attention-deficit/hyperactivity disorder: current findings and future directions. Expert Rev Neurother. 2013;13(4):435–45. This is an exhaustive review on the genetics studies in ADHD. It summarized that all variants associated with the pathology explain only a small fraction of heritability: phenotype complexity and variants of small effect contribute to the missing heritability issue.
Gould TD, Manji HK. The molecular medicine revolution and psychiatry: bridging the gap between basic neuroscience research and clinical psychiatry. J Clin Psychiatry. 2004;65(5):598–604.
Insel T (2013) Director’s Blog: transforming diagnosis. http://www.nimh.nih.gov/about/director/2013/transforming-diagnosis.shtml
Biomarkers Definitions Working Group. Biomarkers and surrogate endpoints preferred definitions and conceptual framework. Clin Pharmacol Ther. 2001;69:89–95.
Singh I, Rose N. Biomarkers in psychiatry. Nature. 2009;460(7252):202–7.
Sokolowska I, Ngounou Wetie AG, Wormwood K, Thome J, Darie CC, Woods AG. The potential of biomarkers in psychiatry: focus on proteomics. J Neural Transm. 2013 Dec 20. doi:10.1007/s00702-013-1134-6. This is a review on the importance to use the proteomics to identify biomarkers for different psychiatric pathologies including ADHD.
Thome J, Ehlis AC, Fallgatter AJ, Krauel K, Lange KW, Riederer P, et al. Biomarkers for attention-deficit/hyperactivity disorder (ADHD). A consensus report of the WFSBP task force on biological markers and the World Federation of ADHD. World J Biol Psychiatry. 2012;13(5):379–400. This is a comprehensive review on clinical, imaging, genetic and proteomic markers and ADHD. The review concluded that, although some promising candidates such as olfactory sensitivity, substantial echogenicity, no biomarkers for ADHD are available.
Lakhan SE, Vieira K, Hamlat E. Biomarkers in psychiatry: drawbacks and potential for misuse. Int Arch Med. 2010;3:1.
Pies R. Beyond reliability: biomarkers and validity in psychiatry. Psychiatry (Edgmont). 2008;5(1):48–52.
Gizer IR, Ficks C, Waldman ID. Candidate gene studies of ADHD: a meta-analytic review. Hum Genet. 2009;126(1):51–90.
Shook D, Brady C, Lee PS, Kenealy L, Murphy ER, Gaillard WD, et al. Effect of dopamine transporter genotype on caudate volume in childhood ADHD and controls. Am J Med Genet B. 2011;156B(1):28–35.
Braet W, Johnson KA, Tobin CT, Acheson R, McDonnell C, Hawi Z, et al. fMRI activation during response inhibition and error processing: the role of the DAT1 gene in typically developing adolescents and those diagnosed with ADHD. Neuropsychologia. 2011;49(7):1641–50.
McGough JJ. Attention deficit hyperactivity disorder pharmacogenetics: the dopamine transporter and D4 receptor. Pharmacogenomics. 2012;13(4):365–8. This is a review on pharmacogenetics and ADHD focusing on DAT1 and DRD4 genes. The author concludes that there is no evidence at present to suggest any role for pharmacogenetics in the clinical management of ADHD for SLA6A3, DRD4 or any other genetic polymorphism.
Bruxel EM, Akutagava-Martins GC, Salatino-Oliveira A, Contini V, Kieling C, Hutz MH, et al. ADHD pharmacogenetics across the life cycle: new findings and perspectives. Am J Med Genet B Neuropsychiatr Genet. 2014. doi:10.1002/ajmg.b.32240. This is a systematic review of the literature on ADHD pharmacogenetics to provide a critical discussion of the existent findings, new approaches, limitations, and recommendations for future research. It focuses on the number of studies continues to grow, and the heterogeneity in methodological strategies employed by different studies.
Durston S, Fossella JA, Mulder MJ, Casey BJ, Ziermans TB, Vessaz MN, et al. Dopamine transporter genotype conveys familial risk of attention-deficit/hyperactivity disorder through striatal activation. J Am Acad Child Adolesc Psychiatry. 2008;47(1):61–7.
Kambeitz J, Romanos M, Ettinger U. Meta-analysis of the association between dopamine transporter genotype and response to methylphenidate treatment in ADHD. Pharmacogenomics J. 2014;14(1):77–84. This is a a meta-analysis of studies investigating the moderating effect of the SLC6A3 VNTR on response to methylphenidate treatment in subjects with ADHD. The results indicate that SLC6A3 VNTR is not a reliable predictor of methylphenidate treatment success in ADHD.
Bedard AC, Schulz KP, Cook EH, Fan J, Clerkin SM, Ivanov I, et al. Dopamine transporter gene variation modulates activation of striatum in youth with ADHD. Neuroimage. 2010;53(3):935–42.
Kebir O, Joober R. Neuropsychological endophenotypes in attention-deficit/hyperactivity disorder: a review of genetic association studies. Eur Arch Psychiatry Clin Neurosci. 2011;261(8):583–94.
Barnes JJM, Dean AJ, Nandam LS, O’Connell RG, Bellgrove MA. The molecular genetics of executive function: role of monoamine system genes. Biol Psychiatry. 2011;69(12):E127–43.
Congdon E, Constable RT, Lesch KP, Canli T. Influence of SLC6A3 and COMT variation on neural activation during response inhibition. Biol Psychol. 2009;81(3):144–52.
Fusar-Poli P, Rubia K, Rossi G, Sartori G, Balottin U. Striatal dopamine transporter alterations in ADHD: pathophysiology or adaptation to psychostimulants? A meta-analysis. Am J Psychiatr. 2012;169(3):264–72.
Falcone M, Smith RM, Chenoweth MJ, Bhattacharjee AK, Kelsoe JR, Tyndale RF, et al. Neuroimaging in psychiatric pharmacogenetics research: the promise and pitfalls. Neuropsychopharmacology. 2013;38(12):2327–37. This review summarizes progress in efforts to incorporate neuroimaging into genetics and treatment research on major psychiatric disorders, such as schizophrenia, major depressive disorder, bipolar disorder, attention-deficit/hyperactivity disorder, and addiction.
Paloyelis Y, Mehta MA, Faraone SV, Asherson P, Kuntsi J. Striatal sensitivity during reward processing in attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2012;51(7):722–32.
Bellgrove MA, Johnson KA, Barry E, Mulligan A, Hawi Z, Gill M, et al. Dopaminergic haplotype as a predictor of spatial inattention in children with attention-deficit/hyperactivity disorder. Arch Gen Psychiatr. 2009;66(10):1135–42.
Shang CY, Gau SSF. Association between the DAT1 gene and spatial working memory in attention deficit hyperactivity disorder. Int J Neuropsychopharmacol. 2014;17(1):9–21.
Konrad K, Dempfle A, Friedel S, Heiser P, Holtkamp K, Walitza S, et al. Familiality and molecular genetics of attention networks in ADHD. Am J Med Genet B Neuropsychiatr Genet. 2010;153B(1):148–58.
Wu J, Xiao HF, Sun HJ, Zou L, Zhu LQ. Role of dopamine receptors in ADHD: a systematic meta-analysis. Mol Neurobiol. 2012;45(3):605–20. This review summarized the ongoing research of DA receptor genes in ADHD pathogenesis and gathered the past published data with meta-analysis and revealed the high risk of DRD5, DRD2, and DRD4 polymorphisms in ADHD.
Taurines R, Grunblatt E, Schecklmann M, Schwenck C, Albantakis L, Reefschlager L, et al. Altered mRNA expression of monoaminergic candidate genes in the blood of children with attention deficit hyperactivity disorder and autism spectrum disorder. World J Biol Psychiatry. 2011;12:104–8.
Shaw P, Eckstrand K, Sharp W, Blumenthal J, Lerch JP, Greenstein D, et al. Attention-deficit/hyperactivity disorder is characterized by a delay in cortical maturation. Proc Natl Acad Sci U S A. 2007;104(49):19649–54.
Wu Z, Yang L, Wang Y. Applying Imaging Genetics to ADHD: the Promises and the Challenges. Mol Neurobiol. 2014 Apr 1. doi:10.1007/s12035-014-8683-z. This is an exhaustive review on neuroimaging genetics in ADHD. The authors conclude that imaging genetics has somewhat showed its potential to provide a more precise understanding of how the genes shape the brain variations and further the clinical features of ADHD.
Lasky-Su J, Neale BM, Franke B, Anney RJL, Zhou KX, Maller JB, et al. Genome-wide association scan of quantitative traits for attention deficit hyperactivity disorder identifies novel associations and confirms candidate gene associations. Am J Med Genet B. 2008;147B(8):1345–54.
Winsberg BG, Comings DE. Association of the dopamine transporter gene (DAT1) with poor methylphenidate response. J Am Acad Child Adolesc Psychiatry. 1999;38(12):1474–7.
Manor I, Corbex M, Eisenberg J, Gritsenkso I, Bachner-Melman R, Tyano S, et al. Association of the dopamine D5 receptor with attention deficit hyperactivity disorder (ADHD) and scores on a continuous performance test (TOVA). Am J Med Genet B. 2004;127B(1):73–7.
Froehlich TE, McGough JJ, Stein MA. Progress and promise of attention-deficit hyperactivity disorder pharmacogenetics. CNS Drugs. 2010;24(2):99–117.
Loo SK, Rich EC, Ishii J, McGough J, McCracken J, Nelson S, et al. Cognitive functioning in affected sibling pairs with ADHD: familial clustering and dopamine genes. J Child Psychol Psychiatry. 2008;49(9):950–7.
Scassellati C, Bonvicini C, Faraone SV, Gennarelli M. Biomarkers and attention-deficit/hyperactivity disorder: a systematic review and meta-analyses. J Am Acad Child Adolesc Psychiatry. 2012;51(10):1003–19. This is an exhaustive meta-analysis providing evidence for several peripheral biomarkers as being associated with ADHD both in diagnosis and in treatment efficacy.
Park MH, Kim JW, Yang YH, Hong SB, Park S, Kang H, et al. Regional brain perfusion before and after treatment with methylphenidate may be associated with the G1287A polymorphism of the norepinephrine transporter gene in children with attention-deficit/hyperactivity disorder. Neurosci Lett. 2012;514(2):159–63.
Kim BN, Kim JW, Cummins TDR, Bellgrove MA, Hawi Z, Hong SB, et al. Norepinephrine genes predict response time variability and methylphenidate-induced changes in neuropsychological function in attention deficit hyperactivity disorder. J Clin Psychopharmacol. 2013;33(3):356–62.
Bobb AJ, Addington AM, Sidransky E, Gornick MC, Lerch JP, Greenstein DK, et al. Support for association between ADHD and two candidate genes: NET1 and DRD1. Am J Med Genet B. 2005;134B(1):67–72.
Kollins SH, Anastopoulos AD, Lachiewicz AM, FitzGerald D, Morrissey-Kane E, Garrett ME, et al. SNPs in dopamine D2 receptor gene (DRD2) and norepinephrine transporter gene (NET) are associated with continuous performance task (CPT) phenotypes in ADHD children and their families. Am J Med Genet B. 2008;147B(8):1580–8.
Lesch KP, Selch S, Renner TJ, Jacob C, Nguyen TT, Hahn T, et al. Genome-wide copy number variation analysis in attention-deficit/hyperactivity disorder: association with neuropeptide Y gene dosage in an extended pedigree. Mol Psychiatry. 2011;16(5):491–503.
Shiffrin ND, Gruber J, Glatt SJ, Faraone SV. No association between MspI allele of the ADRA2A polymorphism and ADHD: meta-analysis of family-based studies. Psychiatr Genet. 2013;23(4):174–5.
Froehlich TE, Epstein JN, Nick TG, Castro MSM, Stein MA, Brinkman WB, et al. Pharmacogenetic predictors of methylphenidate dose-response in attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2011;50(11):1129–39.
Yang L, Qian Q, Liu L, Li H, Faraone SV, Wang Y. Adrenergic neurotransmitter system transporter and receptor genes associated with atomoxetine response in attention-deficit hyperactivity disorder children. J Neural Transm. 2013;120(7):1127–33.
Sun HJ, Yuan FF, Shen XM, Xiong GL, Wu J. Role of COMT in ADHD: a systematic meta-analysis. Mol Neurobiol. 2014;49(1):251–61. This is a meta-analysis on findings investigating associations between COMT gene and ADHD. The results indicated no association.
Qian QJ, Yang L, Wang YF, Zhang HB, Guan LL, Chen Y, et al. Gene-gene interaction between COMT and MAOA potentially predicts the intelligence of attention-deficit hyperactivity disorder boys in China. Behav Genet. 2010;40(3):357–65.
Thapar A, Langley K, Fowler T, Rice F, Turic D, Whittinger N, et al. Catechol O-methyltransferase gene variant and birth weight predict early-onset antisocial behavior in children with attention-deficit/hyperactivity disorder. Arch Gen Psychiatr. 2005;62(11):1275–8.
Caspi A, Langley K, Milne B, Moffitt TE, O’Donovan M, Owen MJ, et al. A replicated molecular genetic basis for subtyping antisocial behavior in children with attention-deficit/hyperactivity disorder. Arch Gen Psychiatr. 2008;65(2):203–10.
Cheon KA, Jun JY, Cho DY. Association of the catechol-O-methyltransferase polymorphism with methylphenidate response in a classroom setting in children with attention-deficit hyperactivity disorder. Int Clin Psychopharmacol. 2008;23(5):291–8.
Kereszturi E, Tarnok Z, Bognar E, Lakatos K, Farkas L, Gadoros J, et al. Catechol-O-methyltransferase Val158Met polymorphism is associated with methylphenidate response in ADHD children. Am J Med Genet B. 2008;147B(8):1431–5.
Manor I, Tyano S, Mel E, Eisenberg J, Bachner-Melman R, Kotler M, et al. Family-based and association studies of monoamine oxidase A and attention deficit hyperactivity disorder (ADHD): preferential transmission of the long promoter-region repeat and its association with impaired performance on a continuous performance test (TOVA). Mol Psychiatry. 2002;7(6):626–32.
Rommelse NNJ, Altink ME, Arias-Vasquez A, Buschgens CJM, Fliers E, Faraone SV, et al. Differential association between MAOA, ADHD and neuropsychological functioning in boys and girls. Am J Med Genet B. 2008;147B(8):1524–30.
Nymberg C, Jia T, Lubbe S, Ruggeri B, Desrivieres S, Barker G, et al. Neural mechanisms of attention-deficit/hyperactivity disorder symptoms are stratified by MAOA genotype. Biol Psychiatry. 2013;74(8):607–14.
Kieling C, Genro JP, Hutz MH, Rohde LA. The -1021 C/T DBH polymorphism is associated with neuropsychological performance among children and adolescents with ADHD. Am J Med Genet B Neuropsychiatr Genet. 2008;147B(4):485–90.
Shim SH, Hwangbo Y, Kwon YJ, Jeong HY, Lee BH, Lee HJ, et al. Increased levels of plasma brain-derived neurotrophic factor (BDNF) in children with attention deficit-hyperactivity disorder (ADHD). Prog Neuro-Psychopharmacol Biol Psychiatry. 2008;32(8):1824–8.
Kim H, Heo HI, Kim DH, Ko IG, Lee SS, Kim SE, et al. Treadmill exercise and methylphenidate ameliorate symptoms of attention deficit/hyperactivity disorder through enhancing dopamine synthesis and brain-derived neurotrophic factor expression in spontaneous hypertensive rats. Neurosci Lett. 2011;504(1):35–9.
Scassellati C, Zanardini R, Tiberti A, Pezzani M, Valenti V, Effedri P, et al. Serum brain-derived neurotrophic factor (BDNF) levels in attention deficit-hyperactivity disorder (ADHD). Eur Child Adolesc Psichiatry. 2014;23(3):173–7.
Joseph N, Zhang-James Y, Perl A, Faraone SV. Oxidative Stress and ADHD: A Meta-Analysis. J Atten Disord. 2013;20(10):1–10 doi:10.1177/1087054713510354. The authors performed a meta-analysis of studies on oxidative stress and antioxidant status in medication naive patients with ADHD and controls. The results suggest that patients with ADHD have normal levels of antioxidant production, but that their response to oxidative stress is insufficient, leading to oxidative damage.
Arns M, Conners CK, Kraemer HC. A decade of EEG theta/beta ratio research in ADHD: a meta-analysis. J Atten Disord. 2013;17(5):374–83. This is a meta-analysis on the TBR in ADHD. The results indicated that excessive TBR cannot be considered a reliable diagnostic measure of ADHD, however a substantial sub-group of ADHD patients do deviate on this measure and TBR has prognostic value in this sub-group, warranting its use as a prognostic measure rather than a diagnostic measure.
Yener GG, Basar E. Brain oscillations as biomarkers in neuropsychiatric disorders: following an interactive panel discussion and synopsis. Suppl Clin Neurophysiol. 2013;62:343–63.
Frazier TW, Demaree HA, Youngstrom EA. Meta-analysis of intellectual and neuropsychological test performance in attention-deficit/hyperactivity disorder. Neuropsychology. 2004;18(3):543–55.
Martinussen R, Hayden J, Hogg-Johnson S, Tannock R. A meta-analysis of working memory impairments in children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2005;44(4):377–84.
Willcutt EG, Doyle AE, Nigg JT, Faraone SV, Pennington BF. Validity of the executive function theory of attention-deficit/hyperactivity disorder: a meta-analytic review. Biol Psychiatry. 2005;57(11):1336–46.
Kasper LJ, Alderson RM, Hudec KL. Moderators of working memory deficits in children with attention-deficit/hyperactivity disorder (ADHD): A meta-analytic review. Clin Psychol Rev. 2012;32(7):605–17.
van Ewijk H, Heslenfeld DJ, Luman M, Rommelse NN, Hartman CA, Hoekstra P, et al. Visuospatial working memory in ADHD patients, unaffected siblings, and healthy controls. J Atten Disord. 2014;18(4):369–78.
Cortese S. The neurobiology and genetics of attention-deficit/hyperactivity disorder (ADHD): what every clinician should know. Eur J Paediatr Neurol. 2012;16(5):422–33.
Friedel S, Saar K, Sauer S, Dempfle A, Walitza S, Renner T, et al. Association and linkage of allelic variants of the dopamine transporter gene in ADHD. Mol Psychiatry. 2007;12(10):923–33.
Cheon KA, Ryu YH, Kim JW, Cho DY. The homozygosity for 10-repeat allele at dopamine transporter gene and dopamine transporter density in Korean children with attention deficit hyperactivity disorder: relating to treatment response to methylphenidate. Eur Neuropsychopharmacol. 2005;15(1):95–101.
Franke B, Faraone SV, Asherson P, Buitelaar J, Bau CHD, Ramos-Quiroga JA, et al. The genetics of attention deficit/hyperactivity disorder in adults, a review. Mol Psychiatry. 2012;17(10):960–87.
Bellgrove MA, Hawi Z, Kirley A, Gill M, Robertson IH. Dissecting the attention deficit hyperactivity disorder (ADHD) phenotype: sustained attention, response variability and spatial attentional asymmetries in relation to dopamine transporter (DAT1) genotype. Neuropsychologia. 2005;43(13):1847–57.
Bellgrove MA, Barry E, Johnson KA, Cox M, Daibhis A, Daly M, et al. Spatial attentional bias as a marker of genetic risk, symptom severity, and stimulant response in ADHD. Neuropsychopharmacology. 2008;33(10):2536–45.
Faraone SV, Spencer TJ, Madras BK, Zhang-James Y, Biederman J. Functional effects of dopamine transporter gene genotypes on in vivo dopamine transporter functioning: a meta-analysis. Mol Psychiatry. 2013;19:880–889.
Noaín D, Avale ME, Wedemeyer C, Calvo D, Peper M, Rubinstein M. Identification of brain neurons expressing the dopamine D4 receptor gene using BAC transgenic mice. Eur J Neurosci. 2006;24(9):2429–38.
Usiello A, Baik JH, Rouge-Pont F, Picetti R, Dierich A, LeMeur M, et al. Distinct functions of the two isoforms of dopamine D2 receptors. Nature. 2000;408(6809):199–203.
Beischlag TV, Marchese A, Meador-Woodruff JH, Damask SP, O’Dowd BF, Tyndale RF, et al. The human dopamine D5 receptor gene: cloning and characterization of the 5′-flanking and promoter region. Biochemistry. 1995;34(17):5960–70.
Li S, Cullen WK, Anwyl R, Rowan MJ. Dopamine-dependent facilitation of LTP induction in hippocampal CA1 by exposure to spatial novelty. Nat Neurosci. 2003;6(5):526–31.
Stahl SM. Neurotransmission of cognition, part 3. Mechanism of action of selective NRIs: both dopamine and norepinephrine increase in prefrontal cortex. J Clin Psychiatry. 2003;64(3):230–1.
Mick E, Neale B, Middleton FA, McGough JJ, Faraone SV. Genome-wide association study of response to methylphenidate in 187 children with attention-deficit/hyperactivity disorder. Am J Med Genet B. 2008;147B(8):1412–8.
Oades RD, Daniels R, Rascher W. Plasma neuropeptide-Y levels, monoamine metabolism, electrolyte excretion and drinking behavior in children with attention-deficit hyperactivity disorder. Psychiatry Res. 1998;80(2):177–86.
Waldman ID, Nigg JT, Gizer IR, Park L, Rappley MD, Friderici K. The adrenergic receptor alpha-2A gene (ADRA2A) and neuropsychological executive functions as putative endophenotypes for childhood ADHD. Cogn Affect Behav Neurosci. 2006;6(1):18–30.
Hong J, Shu-Leong H, Tao X, Lap-Ping Y. Distribution of catechol-O-methyltransferase expression in human central nervous system. Neuroreport. 1998;9(12):2861–4.
Brunner HG, Nelen M, Breakefield XO, Ropers HH, Vanoost BA. Abnormal-behavior associated with a point mutation in the structural gene for monoamine oxidase-A. Science. 1993;262(5133):578–80.
Cases O, Lebrand C, Giros B, Vitalis T, De Maeyer E, Caron MG, et al. Plasma membrane transporters of serotonin, dopamine, and norepinephrine mediate serotonin accumulation in atypical locations in the developing brain of monoamine oxidase A knock-outs. J Neurosci. 1998;18(17):6914–27.
Deckert J, Catalano M, Syagailo YV, Bosi M, Okladnova O, Di Bella D, et al. Excess of high activity monoamine oxidase A gene promoter alleles in female patients with panic disorder. Hum Mol Genet. 1999;8(4):621–4.
Gaspar P, Berger B, Febvret A, Vigny A, Henry JP. Catecholamine innervation of the human cerebral cortex as revealed by comparative immunohistochemistry of tyrosine hydroxylase and dopamine-beta-hydroxylase. J Comp Neurol. 1989;279(2):249–71.
Rogeness GA, Maas JW, Javors MA, Macedo CA, Fischer C, Harris WR. Attention deficit disorder symptoms and urine catecholamines. Psychiatry Res. 1989;27(3):241–51.
Caylak E. Biochemical and genetic analyses of childhood attention deficit/hyperactivity disorder. Am J Med Genet B. 2012;159B(6):613–27. This is an review on biochemistry and genetics of ADHD. The authors reported many published studies about biochemical abnormalities in ADHD as well as many published genetic studies implicating several genes (dopaminergic: DRD4, DAT1, DRD5, COMT; noradrenergic: DBH, ADRA2A; serotonergic: 5-HTT, HTR1B, HTR2A; cholinergic: CHRNA4, and central nervous system development pathway: SNAP25, BDNF) in the etiology of ADHD.
Mattson MP. Glutamate and neurotrophic factors in neuronal plasticity and disease. Ann N Y Acad Sci. 2008;1144:97–112.
Söllner T, Whiteheart SW, Brunner M, Erdjument-Bromage H, Geromanos S, Tempst P, et al. SNAP receptors implicated in vesicle targeting and fusion. Nature. 1993;362(6418):318–24.
Ghanizadeh A, Berk M. Zinc for treating of children and adolescents with attention-deficit hyperactivity disorder: a systematic review of randomized controlled clinical trials. Eur J Clin Nutr. 2013;67(1):122–4.
Bloch MH, Qawasmi A. Omega-3 fatty acid supplementation for the treatment of children with attention-deficit/hyperactivity disorder symptomatology: systematic review and meta-analysis. J Am Acad Child Adolesc Psychiatry. 2011;50(10):991–1000.
Chovanova Z, Muchova J, Sivonova M, Dvorakova M, Zitnanova I, Waczulikova I, et al. Effect of polyphenolic extract, Pycnogenol (R), on the level of 8-oxoguanine in children suffering from attention deficit/hyperactivity disorder. Free Radic Res. 2006;40(9):1003–10.
Dvorakova M, Jezova D, Blazicek P, Trebaticka J, Skodacek I, Suba J, et al. Urinary catecholamines in children with attention deficit hyperactivity disorder (ADHD): Modulation by a polyphenolic extract from pine bark (Pycnogenol((R))). Nutr Neurosci. 2007;10(3–4):151–7.
Garcia RJ, Francis L, Dawood M, Lai ZW, Faraone SV, Perl A. Attention deficit and hyperactivity disorder scores Are elevated and respond to N-acetylcysteine treatment in patients with systemic lupus erythematosus. Arthritis Rheum US. 2013;65(5):1313–8.
Snyder SM, Hall JR. A meta-analysis of quantitative EEG power associated with attention-deficit hyperactivity disorder. J Clin Neurophysiol. 2006;23(5):440–55.
Tye C, Rijsdijk F, McLoughlin G. Genetic overlap between ADHD symptoms and EEG theta power. Brain Cogn. 2014;87:168–72.
Kofler MJ, Rapport MD, Sarver DE, Raiker JS, Orban SA, Friedman LM, et al. Reaction time variability in ADHD: a meta-analytic review of 319 studies. Clin Psychol Rev. 2013;33(6):795–811. This is a meta-analysis of 319 studies of RT variability in children, adolescents, and adults with ADHD relative to typically developing (TD) groups, clinical control groups, and themselves. The results of the meta-analysis indicate that RT variability reflects a stable feature of ADHD and other clinical disorders that is robust to systematic differences across studies.
Lange KW, Hauser J, Lange KM, Makulska-Gertruda E, Takano T, Takeuchi Y et al. Utility of cognitive neuropsychological assessment in attention-deficit/hyperactivity disorder. Atten Defic Hyperact Disord. 2014 Mar 18. doi:10.1007/s12402-014-0132-3. This is an interesting review on the utility of neuropsychological tests in ADHD. The authors concluded that cognitive neuropsychological assessment appears to be at present of limited clinical use and confined to individual descriptions.
Kasparek T, Theiner P, Filova A. Neurobiology of ADHD from childhood to adulthood: findings of imaging methods. J Atten Disord. 2013;20(10):1–13. doi:10.1177/1087054713505322. The authors concluded that changes in neuronal plasticity may be behind persisting brain changes in ADHD. Current treatment approaches seem to improve these neuroplastic processes, and, therefore, may have a positive effect on the neuropathology of ADHD.
Bellgrove MA, Mattingley JB, Hawi Z, Mullins C, Kirley A, Gill M, et al. Impaired temporal resolution of visual attention and dopamine beta hydroxylase genotype in attention-deficit/hyperactivity disorder. Biol Psychiatry. 2006;60(10):1039–45.
Wasserman T, Wasserman LD. The sensitivity and specificity of neuropsychological tests in the diagnosis of attention deficit hyperactivity disorder. Appl Neuropsychol Child. 2012;1(2):90–9.
Acknowledgments
This research was supported by grants from the Fondazione Mariani (RF2006) and from the Italian Ministry of Health (Ricerca Corrente).
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
Cristian Bonvicini Ph.D., Catia Scassellati declare that they have no conflict of interest.
In the past year, Stephen V. Faraone received consulting income, travel expenses and/or research support from Ironshore, Shire, Akili Interactive Labs, Alcobra, VAYA Pharma, and SynapDx and research support from the National Institutes of Health (NIH). His institution is seeking a patent for the use of sodium-hydrogen exchange inhibitors in the treatment of ADHD. In previous years, he received consulting fees or was on Advisory Boards or participated in continuing medical education programs sponsored by: Shire, Alcobra, Otsuka, McNeil, Janssen, Novartis, Pfizer and Eli Lilly. Dr. Faraone receives royalties from books published by Guilford Press: Straight Talk about Your Child’s Mental Health and Oxford University Press: Schizophrenia: The Facts.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Attention-Deficit Disorder
Rights and permissions
About this article
Cite this article
Faraone, S.V., Bonvicini, C. & Scassellati, C. Biomarkers in the Diagnosis of ADHD – Promising Directions. Curr Psychiatry Rep 16, 497 (2014). https://doi.org/10.1007/s11920-014-0497-1
Published:
DOI: https://doi.org/10.1007/s11920-014-0497-1