Abstract
Purpose of Review
The purpose of this review is to summarize current approaches to management of functional vision disorder (FVD).
Recent Findings
Several retrospective studies of FVD in both adults and children have shed light on the range of outcomes and the prevalence of psychosocial stressors among FVD patients. While the first line of treatment for FVD is reassurance and education, recent case reports highlight the use of additional treatment modalities including psychotherapy, hypnosis, and transcranial magnetic stimulation in specific cases.
Summary
Although the epidemiology and diagnosis of functional vision disorder are well described, there is limited evidence supporting treatment modalities. Nevertheless, the majority of patients improve with conservative management including reassurance, education, and appropriate follow-up. Additional approaches such as mental health care referral can be considered in refractory cases.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Functional vision disorder (FVD) is a condition in which patients experience a loss or disturbance to vision for which clinical examination and testing are inconsistent with an ophthalmic or neurological disease. Vision loss is the most common symptom impacting central and or peripheral vision in one or both eyes. Illusions such as polyopia or distortion can also occur. Efferent presentations are rarer (Table 1). It is contrasted with “organic” disease, which is caused by physical, measurable disease of the eye or nervous system. FVD falls under the umbrella of functional neurological disorders (FND) that encompass a wide array of motor and sensory disturbances. The occurrence of FVD in ophthalmology practices has been estimated to be between 0.5 and 5% [1]. It is even more common in children, in whom FVD can be a transient and self-resolving issue.
Far from being a diagnosis of exclusion, the Diagnostic and Statistical Manual of the Mental Disorders, fifth edition (DSM V) criteria for FND, also known as “conversion disorder,” require that the patient experiences distress or impairment related to visual symptoms, not consistent with known neurological disease process, that are not known to be voluntary in nature (i.e., either involuntary or unknown) [2]. In addition, although not a DSM criteria, any demonstration of reversibility of a deficit is characteristic of FND. There are several clinical signs and examination maneuvers used to specifically diagnose FVD, and they have been reviewed widely elsewhere [1, 3, 4••, 5, 6, 7, 8, 9, 10, 11]. The DSM V distinguishes FND from somatic symptom disorder, factitious disorder and malingering. Somatic symptom disorder may involve symptoms that are due to a known or unknown process. The central feature is the patient’s preoccupation with and excessive thoughts and feelings about the symptoms. Factitious disorder or malingering is diagnosed when there is evidence that the patient is voluntary producing the symptoms in order to adopt a sick role or for secondary gain (e.g. attention or money), respectively.
Comprehensive reviews of FVD treatment are lacking, likely because the evidence for therapeutic approaches is limited to retrospective studies and case series (Table 2). Even across FNDs, Class 1 evidence supporting treatment recommendations is limited with the strongest evidence existing for the treatment of functional motor disorder (FMD) and functional seizures, also known as psychogenic nonepileptic seizures (PNES) [12]. Nevertheless, there are numerous reports of successful outcomes in FVD. In this review, we summarize current approaches to FVD management, considering them in the broader context of FNDs. We highlight opportunities for FVD treatment research.
Education and Therapeutic Alliance
It is critically important to take patients who have FND (including FVD) seriously and offer them a diagnosis promptly, as there is some evidence that the time to recover is related to the time to diagnosis. In one study of patients with FMD, patients who completely recovered had an average of 0.7 years to diagnosis compared to 15.2 years for patients who did not recover. In this study, no other factor, including treatment modality, differed between patients who did or did not recover.[27] In another study of functional tremor, there was an average difference of 1.4 years in the time to diagnosis between patients who improved and those who did not [8]. For FVD specifically, it has been observed that the majority of patients will experience a full recovery with an explanation of the disease and reassurance alone [13]. In a study by Lim et al., reassurance alone, without additional referrals or therapy, was sufficient to result in symptom resolution in 89% of patients [19, 28].
Education and a trusting relationship are the most powerful tools a provider can employ when taking care of any FND patient including one with FVD. An accusatory, confrontational, or unsympathetic approach will lead to mistrust of the provider. Patients should not be told that their symptoms are not real or only due to psychological issues. Instead, the provider should explain that the patient’s symptoms are understood and believed, that it is not “in their heads” [6, 29, 30, 31]. It can be helpful to engage patients in a discussion of how they conceptualize the functional symptoms and validate their response. In addition, patients should be provided the opportunity to express all of their symptoms and complaints, including about prior interactions with the healthcare system [2, 29, 31••]. When documenting visits, avoid labeling patients as “malingering” or “faking” unless there is strong evidence that symptoms are occurring only in settings where secondary gain can be obtained (and thus a diagnosis of FVD is incorrect [32]). The current diagnostic criteria for FND (and FVD) do not require a judgement as to the voluntary nature of the deficits.
Patients should be told that they have an established diagnosis, how that diagnosis was made, and, importantly, that it is reversible. Stone and Edwards suggest using the metaphor that “this is like a software problem rather than a hardware problem” [33]. Baker et al. have also provided examples of how to describe FND to patients [34]. It can help to demonstrate to patients the physical signs and tests that confirm the nervous system is intact. For example, Hoover’s sign can be described to patients with functional leg weakness [33]. It is beneficial to include family members in the discussion, as they may contribute additional observations regarding symptoms and behaviors.
A modified SPIKES approach has been proposed for discussing the diagnosis of FVD with a patient [4••]. Briefly, the steps are:
-
1.
Setting up the interview, including preparation by the provider, selection of setting, and preparing the patient and family
-
2.
assessing the patient’s Perception, insight, and expectations about their illness
-
3.
Inviting the patient to establish what they would like to know and not know
-
4.
providing Knowledge tailored to the patient’s level of comprehension including emphasizing that the symptoms are real and have a good prognosis
-
5.
responding to expressed Emotions empathically
-
6.
Summarizing including a plan for future care.
Similar to other FNDs, in FVD it is valuable to discuss and explain the observations and examination findings that led to the diagnosis, and why these observations indicate normal neurological function [5]. For example, Yeo et al. utilized a YouTube video simulating an optokinetic drum to demonstrate to a patient that her eyes were functioning normally [5]. They also engaged family members in supporting patients through the diagnosis, including discussing with patients when they noticed behaviors that would indicate intact vision (such as navigating around objects while walking) [8]. Another example is provided by Moss and Jabbehdari, in which a patient’s symptoms resolved after pattern visual evoked potential (VEP) testing provided electrophysiological evidence that her visual pathways was intact [10]. It is essential to highlight the positive, reassuring aspects of the examination [10, 13] provide reassurance that there is no physical disease process, no structural damage to the eye or nervous system [19], and that there is no serious or life threatening illness [19, 29, 29]. The provider should explain that symptoms are caused by a problem with processing of visual information in the brain [32] and emphasize that complete recovery and symptom resolution is possible and in fact expected [35, 35, 36]. Patients can be directed to online resources such as https://www.neurosymptoms.org and https://www.fndhope.org, to supplement provider reassurance and opportunity for follow-up [37••]. Many providers are hesitant to prescribe placebo treatments such as eyedrops or eyeglasses unless they are specifically indicated, so as to not undermine the message that there is no physical disease [37, 38, 38, 39]. Kathol et al. found increased rates of recovery with reassurance alone than with reassurance combined with placebo treatments [40].
Management of Comorbidities
It is common for people with FVD to also have organic ophthalmic or neurological disease [17]. It is important to explain the co-existence and differences to the patient and to ensure appropriate management of the organic disease [5]. In fact, treatment of the organic disease may improve FVD symptoms [6, 30] because FVD may be related to experiencing organic disease symptoms or diagnosis [25].
A diagnosis of FVD does not preclude development of organic disease, and it is important to continue regular eye examinations and appropriate work-up of any new symptoms. The pitfalls of failing to do this are highlighted in a report of a patient whose organic disease diagnosis was delayed due to misattribution of new symptoms to a prior diagnosis of FVD [41].
Mental Health Care
Mental health care can be an important component of FND management for primary treatment, treatment of concurrent mental health disease or symptoms, and management of stressors. Kathol et al. reported 22 out of 42 FVD patients had a prior psychiatric diagnosis, including personality disorder [13]. Lim et al. reported 39% of FVD patients had prior psychiatric diagnoses. Diagnosed psychiatric diseases should be treated per current guidelines. Barriers to effective mental health therapy include time and cost, as well as patients resisting therapy due to belief that they do not have FND or that therapy will not help them [42]. Patients may also think they have an organic illness that doctors are not able to diagnose, increasing anxiety and leading to higher healthcare utilization through repeated work-ups and specialists.
A history of stressful life events is common in people with FND. Ludwig et al. found in their meta-analysis of FND case–control studies that patients with FND were more likely to have experienced stressful life events than controls. However, they did note that there was a substantial portion of patients across studies without such events [43]. Kozlowska et al. suggested that in children who have experienced adverse life events, FND can stem from specific patterns of emotional processing—for example, inhibiting negative emotions—and that specifically addressing these cognitive patterns is important for best outcomes [44]. Lim et al. found 36% of adults with FVD reported significant psychosocial stressors [19]. While these studies do not establish causation between the stressor and the symptoms, they do support trying stressor management as a potential therapeutic approach [31].
Mental health care for FND can start with the diagnosing provider. In fact, some experts have suggested that the majority of FVD patients do not require formal mental health care [1] [8]. A thorough psychiatric and social history including past trauma and current stressors should be conducted [6]. The patient should be told that stress, anxiety, and depression can affect their experience in many ways, including through vision loss, and asked if they can think of major sources of stress, anxiety and depression in their lives [19]. Some patients may object to this explanation or feel that it is dismissive, so it is important to reinforce that identifying and addressing a stressor that may be contributing to their symptoms could be helpful and that this stressor is not necessary psychological (for instance, it could be a reaction to burdensome existing organic disease). This concept can also be framed as “internal conflict” contributing to symptoms, which may resonate with patients who are unable to acknowledge stress, anxiety or mood issues. It can also be helpful to use the strategy of normalizing illness, for example, pointing out that the eyes can malfunction in response to the pressures of life similar to a gastric ulcer forming. If present, include family members in this discussion as well [19]. Screening metrics for anxiety and depression may be used to identify these symptoms and guide treatment [32••].
Referral to a mental health provider should be obtained for patients with a known or suspected history of psychiatric disease, sexual or physical abuse, or other known trauma [3, 7]. It may also be helpful for patients with long-term, refractory symptoms [3, 30]. Psychotherapy may also be needed to manage depressive symptoms resulting from the functional vision loss—there is a higher prevalence of reported depression and depression symptoms in adults with a loss of visual function from any cause [45]. There is a single conference report describing association between improvement in symptoms in four FVL patients and treatment with the serotonin–norepinephrine reuptake inhibitor duloxetine [46].
While older literature suggests psychotherapy may not be efficacious for FVD [3], modern studies suggest otherwise. Cognitive behavioral therapy (CBT) is often the approach of choice for FND. A randomized controlled trial of CBT found a greater reduction in severity scores for patients who underwent baseline psychiatric consultation in addition to CBT than patients who underwent the baseline consultation only [13]. A randomized control trial of CBT for functional seizures, a common FND, compared to standard medical care found that significantly more patients who underwent CBT had reduced functional seizure burden and increased functional seizure freedom at 3 months [29]. However, while a larger trial conducted in 2020 by the same group did not show an improvement in functional seizure burden with CBT, it did demonstrate a modest improvement in the proportion of patients functional seizure free at 12 months (20% vs 12%) as well as improvement in secondary measures including bothersomeness of seizures and health-related quality of life [47]. A randomized control trial of either sertraline or placebo in functional seizure patients showed that the sertraline-treated arm had significantly more functional seizure reduction than the placebo arm (45% versus 8%) [48]. A follow-up study randomizing functional seizure patients to no treatment, sertraline, CBT, or CBT plus sertraline, found improvement in both the CBT arms, but not the sertraline only arm [49]. A randomized control trial of a CBT-based guided self-help intervention did not show long-term improvement in overall health, but did reduce symptom burden and patient confidence in the possibility of improvement [50]. For FVD, similar to other forms of FND, cognitive behavioral therapy has been suggested as a second line treatment [51, 52].
There are additional modalities of mental health care that may be effective in FND treatment. Some improvement has been seen in functional motor disorder with psychodynamic psychotherapy (PDT) [53]. One case study also reported success with psychodynamic psychotherapy in FVD [54].
Hypnosis was studied in 44 FND patients, four of whom had either functional blepharospasm or ptosis, who were randomized to either an immediate hypnosis treatment or a waiting list awaiting future hypnosis therapy. The patients who underwent the hypnosis treatment were more likely to experience symptom improvement, which was maintained at 6-month follow-up [55]. However, when the same group conducted a randomized controlled trial comparing inpatient psychiatric care with inpatient psychiatric care in addition to hypnosis, hypnosis did not confer any additional benefit [56]. Greenleaf in 1971 presented a detailed description of successful treatment of a patient with “hysterical blindness” with hypnotherapy and discussed other contemporary reports of hypnotherapy for this condition [57]. Yeo et al. describe the use of hypnotherapy as one of several treatments for two FVD patients. Both eventually improved, and although it is possible that hypnotherapy did not contribute to their recovery, both patients had positive reactions to the treatment.[35].
Physical Therapy
Physical therapy (PT) is a major component of the treatment for FNDs, especially those that are motor-related. The goal of PT in FMD is for patients to be able to perform specific activities that will improve their day-to-day function. Shifting the patient’s focus of attention from their symptoms to functional goals, such as getting up from bed or walking, employs the concept of distraction leading to symptom suppression in FND [58]. A commonly used tool is the “stop and reset” in which maladaptive behaviors are stopped and the patient goes back to square one, attempting again to carry out a healthy movement. This helps patients embody the role of a “healthy” rather than a “sick” individual, and resume their usual activities [59]. It also helps to re-train any abnormal behaviors that patients may have developed [32••]. Rehabilitation should follow a similar course of treatment as in organic disease, as the lives of FND patients are impacted by their symptoms to the same degree as other patients [60]. In one case–control study of PT for FMD, significantly more PT-treated patients experienced improvement compared to controls not treated with PT (60.4% vs 21.9%). Unfortunately, 87.5% of all patients, whether treated with PT or not, did not experience complete resolution of symptoms [61].
There are very limited descriptions of the use of occupational and vision therapy for FVD. The goals of occupational therapy for vision loss due to FVD may include the ability to perform daily activities independently (for example, through modifications for light sensitivity), and reducing compensatory habits that patients may have which hinder their recovery [62••]. One patient had success (improvement in vision from 20/150 to 20/70) after five sessions of optometric vision therapy, including monocular fixation, pursuits, accommodative work, and binocular accommodation and vergence [63••]. In addition, although this approach is impractical in most contexts, the military previously had success with “retinal rest,” which involved patching and covering both eyes and physically isolating patients until vision improved [1]. There are no studies comparing the efficacy of vision therapy to other lines of treatment, and its use in treating FVD is based only on anecdotal evidence [6, 7].
Transcranial Magnetic Stimulation (TMS)
There has been evidence for symptom improvement in FMD with TMS of the motor cortex to elicit movement. It has been proposed that the mechanism of benefit of this procedure is the experience of movement by the patient in response to the stimulation, thus demonstrating that symptom reversal is possible [64]. This is supported by a study in which patients received equivalent benefits from both TMS and root magnetic stimulation, which induces movement but does not stimulate the cortex, and showed similar benefit [65].
In a study by Parain and Chastan, ten FVL patients (pediatric and adult) were treated with large-field, low-frequency, repetitive transcranial magnetic stimulation centered on the occipito-parietal area. Nine patients experienced improvement, six of whom completely recovered [66]. Yeo et al. describe the use of occipital TMS in two patients, both of whom eventually recovered though not immediately after the TMS treatment. Similar to what has been reported for other FNDs, TMS may have had a placebo-like effect on these patients, as they both reported that the temporary visual experiences that were triggered during the TMS improved their confidence that they could regain their vision.[35].
Management of FVD in Children
Although much of the above discussion applies when treating both adults and children with FVD, there are some special considerations in the pediatric population. FVD in children is considered to be shorter-lived and requires less intervention than in adults.[67] As with adults, most children do not have associated psychiatric conditions [68].
When approaching the child’s family, it is important to tell the parents that the child is not purposely faking or fabricating their symptoms [1]. Providers should instruct parents to be supportive and encouraging toward the child, especially in regard to their vision, rather than punitive [1, 6, 9]. Similar instructions should be given to the child’s school. An example of a letter to school explaining the cause of the child’s visual symptoms is given by Moy et al. [10]. It is possible that some children may be feigning visual symptoms in order to obtain glasses [10, 16, 19]. In general, telling the child that this type of visual complaint is common and that return of normal vision is expected in a number of weeks, allows children to recover without embarrassment [23]. It is especially important in children to take a detailed social history and assess for possible sources of trauma or stress, including within the family [25, 25]. Porteous et al. present three cases of medically unexplained vision loss in children associated with either physical abuse, social stress factors, or history of anxiety [40]. In children, FVD has been associated with sexual abuse, so it is important to appropriately screen for this [69, 70].
In a study by Catalono et al., only one out of 23 patients were referred for psychologic therapy, the rest being treated with reassurance alone, and 74% of these children had complete resolution of symptoms after two months [14]. School- or family-related psychological factors are often triggers for FVD in children and should be managed with mental health care if necessary [30]. An eight-year-old boy whose bilateral vision loss was attributed to bullying at school, fully recovered after discussion of the bullying with parents and teachers [71].
In one case series of 85 children and adolescents with FVD, all but one resolved with reassurance only [67], consistent with previous reports that FVD symptoms are usually milder and shorter-lived in children than in adults, unless associated with trauma or abuse [9]. Another retrospective study of children with FVD found 85% of patients had full recovery within one year of diagnosis, and the time to recovery was not related to psychosocial factors, age of onset or time to diagnosis [21]. In another study, 9 out of 12 pediatric patients experienced full recovery [22].
Conclusions
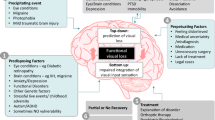
Although there is limited evidence for the use of any particular treatment for FVD, the current literature supports a combination of trust-building and careful reassurance for initial management of most FVD patients, with mental health therapy being useful in specific cases. Additional approaches such as hypnosis or TMS might be considered in long-standing refractory cases (Fig. 1).
Although education and reassurance are the first line of treatment for FVD, it can be difficult to achieve in the constraints of a busy clinic. In a survey of UK ophthalmologists, a noted barrier was that they did not have enough time with their FVD patients to address all their concerns and that it was difficult for their patients to access psychological care and other resources [72••]. It might be valuable for future prospective studies to focus on approaches to overcoming these barriers, including specific aspects of how and by whom education and reassurance is performed, including time spent face-to-face with the patient, number of visits, and the manner in which the diagnosis was explained. Future studies should stratify patients based on whether they have a concurrent mental health diagnosis to help identify those likely to benefit from mental health care [8]. Finally, a major challenge in FVD research is the small numbers of patients included in most studies. Multicenter trials or those focused on neuro-ophthalmology practices which may have higher FVD patient volumes can help improve the current state of our knowledge on how to best treat FVD.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Liu GT, Volpe NJ, Galetta SL. 11 - Functional (Nonorganic) Visual Loss. In: Liu GT, Volpe NJ, Galetta SL, editors. Liu, Volpe, and Galetta’s Neuro-Ophthalmology (Third Edition) [Internet]. Elsevier; 2019 [cited 2021 Dec 21]. p. 379–93. Available from: https://www.sciencedirect.com/science/article/pii/B9780323340441000110.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5. 5th ed. Arlington: American Psychiatric Publishing; 2013.
Dattilo M, Biousse V, Bruce BB, Newman NJ. Chapter 29 - Functional and simulated visual loss. In: Hallett M, Stone J, Carson A, editors. Handbook of Clinical Neurology [Internet]. Elsevier; 2016 [cited 2021 Dec 2]. p. 329–41. (Functional Neurologic Disorders; vol. 139). Available from: https://www.sciencedirect.com/science/article/pii/B9780128017722000291.
•• Raviskanthan S, Wendt S, Ugoh PM, Mortensen PW, Moss HE, Lee AG. Functional vision disorders in adults: a paradigm and nomenclature shift for ophthalmology. SurvOphthalmol [Internet]. 2021 Mar 15 [cited 2021 Dec 2]; Available from: https://www.sciencedirect.com/science/article/pii/S0039625721000758. (A review of the diagnostic approach to FVD, which also proposes a framework for discussing a diagnosis of FVD with patients.)
Vaphiades MS, Kline LB. Functional Neuro-ophthalmic Conditions. Contin Lifelong Learn Neurol. 2009;15(4):106.
Egan RA, LaFrance WC. Functional Vision Disorder. Semin Neurol. 2015;35(5):557–63.
Pula J. Functional vision loss. Curr Opin Ophthalmol. 2012;23(6):460–5.
Bruce BB, Newman NJ. Functional Visual Loss. Neurol Clin. 2010;28(3):789–802.
Bose S, Kupersmith MJ. Neuro-Ophthalmologic Presentations of Functional Visual Disorders. Neurol Clin. 1995;13(2):321–39.
Beatty S. Non-organic visual loss. Postgrad Med J. 1999;75(882):201–7.
Pasol J. Nonorganic Visual Disorders. Int Ophthalmol Clin. 2019;59(3):141–52.
Rosebush PI, Mazurek MF. Treatment of conversion disorder in the 21st century: have we moved beyond the couch? Curr Treat Options Neurol. 2011;13(3):255–66.
Kathol RG, Cox TA, Corbett JJ, Thompson HS. Functional visual loss. Follow-up of 42 cases. Arch Ophthalmol Chic Ill 1960. 1983;101(5):729–35.
Catalono RA, Simon JW, Krohel GB, Rosenberg PN. Functional visual loss in children. Ophthalmology. 1986;93(3):385–90.
Drinnan MJ, Marmor MF. Functional visual loss in Cambodian refugees: a study of cultural factors in ophthalmology. Eur J Ophthalmol. 1991;1(3):115–8.
Bain KE, Beatty S, Lloyd C. Non-organic visual loss in children. Eye Lond Engl. 2000;14(Pt 5):770–2.
Scott JA, Egan RA. Prevalence of organic Neuro-ophthalmologic disease in patients with functional visual loss. Am J Ophthalmol. 2003;135(5):670–5.
Taich A, Crowe S, Kosmorsky GS, Traboulsi EI. Prevalence of psychosocial disturbances in children with nonorganic visual loss. J AAPOS Off Publ Am Assoc Pediatr Ophthalmol Strabismus. 2004;8(5):457–61.
Lim SA, Siatkowski RM, Farris BK. Functional Visual Loss in Adults and Children: Patient Characteristics, Management, and Outcomes. Ophthalmology. 2005;112(10):1821–8.
Ney JJ, Volpe NJ, Liu GT, Balcer LJ, Moster ML, Galetta SL. Functional visual loss in idiopathic intracranial hypertension. Ophthalmology. 2009;116(9):1808-1813.e1.
Toldo I, Pinello L, Suppiej A, Ermani M, Cermakova I, Zanin E, et al. Nonorganic (Psychogenic) Visual Loss in Children: A Retrospective Series. J Neuroophthalmol. 2010;30(1):26–30.
Kinori M, Wygnanski-Jaffe T, Huna-Baron R. Functional Visual Loss in an Israeli Pediatric Population. Isr Med Assoc J. 2011;13(11):684–8.
Muñoz-Hernández AM, Santos-Bueso E, Sáenz-Francés F, Méndez-Hernández CD, García-Feijoó J, Gegúndez-Fernández JA, et al. Nonorganic Visual Loss and Associated Psychopathology in Children. Eur J Ophthalmol. 2012;22(2):269–73.
O’Leary ÉD, McNeillis B, Aybek S, Riordan-Eva P, David AS. Medically unexplained visual loss in a specialist clinic: a retrospective case–control comparison. J Neurol Sci. 2016;361:272–6.
Somers A, Casteels K, Van Roie E, Spileers W, Casteels I. Non-organic visual loss in children: prospective and retrospective analysis of associated psychosocial problems and stress factors. Acta Ophthalmol (Copenh). 2016;94(5):e312–6.
Daniel MC, Coughtrey A, Heyman I, Dahlmann-Noor AH. Medically unexplained visual loss in children and young people: an observational single site study of incidence and outcomes. Eye. 2017;31(7):1068–73.
Schwingenschuh P, Pont-Sunyer C, Surtees R, Edwards MJ, Bhatia KP. Psychogenic movement disorders in children: a report of 15 cases and a review of the literature. Mov Disord Off J Mov Disord Soc. 2008;23(13):1882–8.
McKeon A, Ahlskog JE, Bower JH, Josephs KA, Matsumoto JY. Psychogenic tremor: long-term prognosis in patients with electrophysiologically confirmed disease. Mov Disord Off J Mov Disord Soc. 2009;24(1):72–6.
Dhanji S, Lawlor M. Functional visual loss. Curr Opin Neurol. 2017;30(2):200–5.
Egan RA. Functional visual loss. Ophthalmol Clin N Am. 2004;17(3):321-8.vi.
Griffiths PG, Ali N. Medically unexplained visual loss in adult patients. Curr Opin Neurol. 2009;22(1):41–5.
•• Gilmour GS, Nielsen G, Teodoro T, Yogarajah M, Coebergh JA, Dilley MD, et al. Management of functional neurological disorder. J Neurol. 2020;267(7):2164–72. (A review of general management principles for all FNDs.)
Stone J, Edwards M. Trick or treat? Showing patients with functional (psychogenic) motor symptoms their physical signs. Neurology. 2012;79(3):282–4.
Baker J, Barnett C, Cavalli L, Dietrich M, Dixon L, Duffy JR, et al. Management of functional communication, swallowing, cough and related disorders: consensus recommendations for speech and language therapy. J Neurol Neurosurg Psychiatry. 2021;92(10):1112–25.
Yeo JM, Carson A, Stone J. Seeing again: treatment of functional visual loss. Pract Neurol. 2019;19(2):168–72.
Moss H, Jabbehdari S. Application of hemi-field visual electrophysiology to diagnose functional vision loss. J Neuro-Ophthalmol Off J North Am Neuro-Ophthalmol Soc. 2020;40(4):527–9.
Thompson HS. Functional visual loss. Am J Ophthalmol. 1985;100(1):209–13.
Gittinger JW. Diagnosis and management of medically unexplained visual loss: One clinician’s perspective. Surv Ophthalmol [Internet]. 2020 Dec 15 [cited 2021 Dec 2]; Available from: https://www.sciencedirect.com/science/article/pii/S0039625720301776.
Kini A, Ajjeya MT, Sudhakar P. Nonorganic Vision Loss. 2020 Nov 16 [cited 2021 Dec 2]; Available from: https://www.touchophthalmology.com/neuro-ophthalmology/journal-articles/nonorganic-vision-loss/.
Brodsky MC. Transient, Unexplained, and Psychogenic Visual Loss in Children. In: Brodsky MC, editor. Pediatric Neuro-Ophthalmology [Internet]. New York, NY: Springer; 2010 [cited 2021 Dec 21]. p. 213–52. Available from: https://doi.org/10.1007/978-0-387-69069-8_5
Ansel T, Nguyen T, Shenouda-Awad N, Zerilli-Zavgorodni T. The Complexity of Co-existing Functional Vision Loss and Organic Diagnoses. Optom Vis Perform. 2018;6(2):17–116.
Ataoglu A, Ozcetin A, Icmeli C, Ozbulut O. Paradoxical therapy in conversion reaction. J Korean Med Sci. 2003;18(4):581–4.
Ludwig L, Pasman JA, Nicholson T, Aybek S, David AS, Tuck S, et al. Stressful life events and maltreatment in conversion (functional neurological) disorder: systematic review and meta-analysis of case-control studies. Lancet Psychiatry. 2018;5(4):307–20.
Kozlowska K, Scher S, Williams LM. Patterns of emotional-cognitive functioning in pediatric conversion patients: implications for the conceptualization of conversion disorders. Psychosom Med. 2011;73(9):775–88.
Zhang X, Bullard KM, Cotch MF, Wilson MR, Rovner BW, McGwin G, et al. Association Between Depression and Functional Vision Loss in Persons 20 Years of Age or Older in the United States, NHANES 2005–2008. JAMA Ophthalmol. 2013;131(5):573–81.
Egan, Robert. Poster 122: Treatment of functional vision loss with duloxetine. North American Neuro-Ophthalmology Society Annual Meeting. 2012. Available at https://www.collections.lib.utah.edu/ark:/87278/s6031z1z.
Allen LA, Woolfolk RL, Escobar JI, et al. Cognitive-behavioral therapy for somatization disorder: a randomized controlled trial. Arch Intern Med. 2006;166(14):1512–8.
Goldstein LH, Chalder T, Chigwedere C, Khondoker MR, Moriarty J, Toone BK, et al. Cognitive-behavioral therapy for psychogenic nonepileptic seizures: a pilot RCT. Neurology. 2010;74(24):1986–94.
Goldstein LH, Robinson EJ, Mellers JDC, Stone J, Carson A, Reuber M, et al. Cognitive behavioural therapy for adults with dissociative seizures (CODES): a pragmatic, multicentre, randomised controlled trial. Lancet Psychiatry. 2020;7(6):491–505.
LaFrance WC, Keitner GI, Papandonatos GD, Blum AS, Machan JT, Ryan CE, et al. Pilot pharmacologic randomized controlled trial for psychogenic nonepileptic seizures. Neurology. 2010;75(13):1166–73.
LaFrance WC, Baird GL, Barry JJ, Blum AS, Frank Webb A, Keitner GI, et al. Multicenter pilot treatment trial for psychogenic nonepileptic seizures: a randomized clinical trial. JAMA Psychiat. 2014;71(9):997–1005.
Sharpe M, Walker J, Williams C, Stone J, Cavanagh J, Murray G, et al. Guided self-help for functional (psychogenic) symptoms: a randomized controlled efficacy trial. Neurology. 2011;77(6):564–72.
Hinson VK, Weinstein S, Bernard B, Leurgans SE, Goetz CG. Single-blind clinical trial of psychotherapy for treatment of psychogenic movement disorders. Parkinsonism Relat Disord. 2006;12(3):177–80.
Schoenfeld MA, Hassa T, Hopf J-M, Eulitz C, Schmidt R. Neural Correlates of Hysterical Blindness. Cereb Cortex. 2011;21(10):2394–8.
Moene FC, Spinhoven P, Hoogduin KAL, van Dyck R. A randomized controlled clinical trial of a hypnosis-based treatment for patients with conversion disorder, motor type. Int J Clin Exp Hypn. 2003;51(1):29–50.
Moene FC, Spinhoven P, Hoogduin KAL, van Dyck R. A randomised controlled clinical trial on the additional effect of hypnosis in a comprehensive treatment programme for in-patients with conversion disorder of the motor type. Psychother Psychosom. 2002;71(2):66–76.
Greenleaf E. The Red House: Hypnotherapy of Hysterical Blindness. Am J Clin Hypn. 1971;13(3):155–61.
Espay AJ, Aybek S, Carson A, Edwards MJ, Goldstein LH, Hallett M, et al. Current Concepts in Diagnosis and Treatment of Functional Neurological Disorders. JAMA Neurol. 2018;75(9):1132–41.
Ness D. Physical Therapy Management for Conversion Disorder: Case Series. J Neurol Phys Ther. 2007;31(1):30–9.
Heruti RJ, Levy A, Adunski A, Ohry A. Conversion motor paralysis disorder: overview and rehabilitation model. Spinal Cord. 2002;40(7):327–34.
Czarnecki K, Thompson JM, Seime R, Geda YE, Duffy JR, Ahlskog JE. Functional movement disorders: Successful treatment with a physical therapy rehabilitation protocol. Parkinsonism Relat Disord. 2012;18(3):247–51.
•• Nicholson C, Edwards MJ, Carson AJ, Gardiner P, Golder D, Hayward K, et al. Occupational therapy consensus recommendations for functional neurological disorder. J Neurol Neurosurg Psychiatry. 2020;91(10):1037–45. (Consensus recommendations for the use of occupational therapy in different manifestations of FND.)
•• Polacco AM, Mozlin R. A comprehensive review of functional vision loss. Optom Vis Perform. 2021;9(1):46–54. (A case report of the use of vision therapy for a patient with FVD.)
Carson AJ, Brown R, David AS, Duncan R, Edwards MJ, Goldstein LH, et al. Functional (conversion) neurological symptoms: research since the millennium. J Neurol Neurosurg Psychiatry. 2012;83(8):842–50.
Garcin B, Roze E, Mesrati F, Cognat E, Fournier E, Vidailhet M, et al. Transcranial magnetic stimulation as an efficient treatment for psychogenic movement disorders. J Neurol Neurosurg Psychiatry. 2013;84(9):1043–6.
Parain D, Chastan N. Large-field repetitive transcranial magnetic stimulation with circular coil in the treatment of functional neurological symptoms. Neurophysiol Clin Neurophysiol. 2014;44(4):425–31.
Coughtrey AE, Daniel MC, Dahlmann-Noor A, Heyman I. Functional visual symptoms in children and young people: mental health profiles and case review. Eye. 2021;30:1–2.
Clarke WN, Noël LP, Bariciak M. Functional visual loss in children: a common problem with an easy solution. Can J Ophthalmol J Can Ophtalmol. 1996;31(6):311–3.
Moy AR, Pietrantonio BJ, Perzan E. Functional Vision Loss in a Community Health Care Setting: A Teaching Case Report. Optom Educ. 2013;38(2):67–75.
Porteous AM, Clarke MP. Medically unexplained visual symptoms in children and adolescents: an indicator of abuse or adversity? Eye. 2009;23(9):1866–7.
Karagiannis D, Kontadakis G, Brouzas D, Moschos M, Damanakis A. Nonorganic visual loss in a child due to school bullying. Am J Ophthalmol Case Rep. 2017;5:90–1.
•• Laginaf M, Costello H, Price G. How do ophthalmologists manage functional visual symptoms? A UK survey of ophthalmologists’ experience.Graefes Arch Clin Exp Ophthalmol [Internet]. 2021 Oct 11 [cited 2021 Dec 19]; Available fromhttps://doi.org/10.1007/s00417-021-05433-4. (A survey of ophthalmologists' experiences treating FVD, finding that many ophthalmologists do not feel adequately trained to discuss a diagnosis of FVD with patients, or adequately supported in caring for FVD patients.)
Acknowledgements
The editors would like to thank Dr. John Brust for taking the time to review this manuscript.
Funding
NIH P30 026877, unrestricted grant from Research to Prevent Blindness to the Stanford Department of Ophthalmology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Ragini Phansalkar, A. Juliana Lockman, Surbhi Bansal, and Heather E. Moss each declare no potential conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Neuro-Ophthalmology
Rights and permissions
About this article
Cite this article
Phansalkar, R., Lockman, A.J., Bansal, S. et al. Management of Functional Vision Disorders. Curr Neurol Neurosci Rep 22, 265–273 (2022). https://doi.org/10.1007/s11910-022-01191-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11910-022-01191-w