Abstract
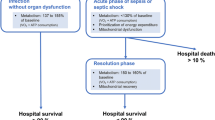
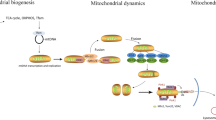
Sepsis is an increasingly common problem, particularly among critically ill patients. Mechanisms by which sepsis induces organ dysfunction have not been elucidated. The coexisting findings (unique to sepsis) of metabolic acidosis yet increased tissue oxygen tensions suggest cellular availability but decreased use of oxygen (tissue dysoxia). Because mitochondria use more than 90% of total body oxygen consumption for adenosine triphosphate (ATP) generation, a bioenergetic abnormality is implied. Cell and animal data have shown that nitric oxide (and its metabolites), produced in considerable excess in patients with sepsis, can affect oxidative phosphorylation by inhibiting several of its component respiratory enzymes. Human data are scarce. However, in skeletal muscle biopsies taken from patients with sepsis, we have recently demonstrated a relationship between increased nitric oxide production, antioxidant depletion, reduced respiratory chain complex I activity, and low ATP levels. These findings correlated with severity of disease and outcome and support the notion that mitochondrial dysfunction resulting in bioenergetic failure may be an important factor in the pathophysiology of sepsis-associated multiorgan failure. However, a reasonable argument can be made that the reduction in energy supply could represent a last-ditch adaptive response to ongoing inflammation, resulting in a cellular shutdown analogous to hibernation that allows eventual restoration of organ function and long-term survival in patients fit enough to survive the acute phase.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References and Recommended Reading
Hoyert DL, Arias E, Smith BL, et al.: Deaths: final data for 1999. Natl Vital Stat Rep 2001, 49:1–113.
Hotchkiss RS, Swanson PE, Freeman BD, et al.: Apoptotic cell death in patients with sepsis, shock, and multiple organ dysfunction. Crit Care Med 1999, 27:1230–1251. This study demonstrates that there is a distinct lack of apoptotic or necrotic cell death in most organs in postmortem tissue sampling of ICU patients dying of multiorgan failure.
Noble JS, MacKirdy FN, Donaldson SI, Howie JC: Renal and respiratory failure in Scottish ICUs. Anaesthesia 2001, 56:124–129.
Rosser DM, Stidwill RP, Jacobson D, Singer M: Oxygen tension in the bladder epithelium rises in both high and low cardiac output endotoxemic sepsis. J Appl Physiol 1995, 79:1878–1882.
VanderMeer TJ, Wang H, Fink MP: Endotoxemia causes ileal mucosal acidosis in the absence of mucosal hypoxia in a normodynamic porcine model of septic shock. Crit Care Med 1995, 23:1217–1226.
Boekstegers P, Weidenhofer S, Pilz G, Werdan K: Peripheral oxygen availability within skeletal muscle in sepsis and septic shock: comparison to limited infection and cardiogenic shock. Infection 1991, 19:317–323. This is the first demonstration of raised tissue oxygen tension in sepsis, as compared to other shock conditions or limited infection.
Rosser DM, Manji M, Cooksley H, Bellingan G: Endotoxin reduces maximal oxygen consumption in hepatocytes independent of any hypoxic insult. Intensive Care Med 1998, 24:725–729.
Kreymann G, Grosser S, Buggisch P, et al.: Oxygen consumption and resting metabolic rate in sepsis, sepsis syndrome, and septic shock. Crit Care Med 1993, 21:1012–1019. This study finds that the sicker the patient is with sepsis, the lower the patient’s oxygen consumption.
Heales SJ, Bolanos JP, Stewart VC, et al.: Nitric oxide, mitochondria and neurological disease. Biochim Biophys Acta 1999, 1410:215–228.
Fukuyama N, Takebayashi Y, Hida M, et al.: Clinical evidence of peroxynitrite formation in chronic renal failure patients with septic shock. Free Radic Biol Med 1997, 22:771–774.
Lanone S, Mebazaa A, Heymes C, et al.: Muscular contractile failure in septic patients: role of the inducible nitric oxide synthase pathway. Am J Respir Crit Care Med 2000, 162:2308–2315. This study shows a relationship in patients with sepsis between increasing muscle weakness (one can speculate this is caused by a mitochondrial pathology) and NO production.
Brealey D, Brand M, Hargreaves I, et al.: Association between mitochondrial dysfunction and severity and outcome of septic shock. Lancet 2002, 360:219–223. This is the first association of a relationship in septic patients between sepsis severity, outcome, ATP levels, mitochondrial inhibition, NO production, and glutathione depletion.
Lyons J, Rauh-Pfeiffer A, Ming-Yu Y, et al.: Cysteine metabolism and whole blood glutathione synthesis in septic pediatric patients. Crit Care Med 2001, 29:870–877.
Borutaite V, Budriunaite A, Brown GC: Reversal of nitric oxide, peroxynitrite- and S-nitrosothiol-induced inhibition of mitochondrial respiration or complex I activity by light and thiols. Biochim Biophys Acta 2000, 1459:405–412.
Brookes PS, Bolanos JP, Heales SJ: The assumption that nitric oxide inhibits mitochondrial ATP synthesis is correct. FEBS Lett 1999, 446:261–263. The authors report that NO inhibits mitochondrial ATP synthesis.
Kantrow SP, Taylor DE, Carraway MS, Piantadosi CA: Oxidative metabolism in rat hepatocytes and mitochondria during sepsis. Arch Biochem Biophys 1997, 345:278–288.
Singer M, Brealey D: Mitochondrial dysfunction in sepsis. In Mitochondria and Cell Death. Edited by Brown GC, Nicholls DG, Cooper CE. London: Portland Press; 1999:149–166. This is a review of all published animal septic models examining mitochondrial function and ultrastructural changes.
Dahn MS, Lange MP, McCurdy B, Mahaffey S: Metabolic function of the isolated perfused rat liver in chronic sepsis. J Surg Res 1995, 59:287–291.
Jin MB, Shimahara Y, Yamaguchi T, et al.: The effect of a bolus injection of TNF-alpha and IL-1 beta on hepatic energy metabolism in rats. J Surg Res 1995, 58:509–515.
Fisch C, Robin MA, Letteron P, et al.: Cell-generated nitric oxide inactivates rat hepatocyte mitochondria in vitro but reacts with hemoglobin in vivo. Gastroenterology 1996, 110:210–220.
Dawson KL, Geller ER, Kirkpatrick JR: Enhancement of mitochondrial function in sepsis. Arch Surg 1988, 123:241–244.
Kurose I, Kato S, Ishii H, et al.: Nitric oxide mediates lipopolysaccharide-induced alteration of mitochondrial function in cultured hepatocytes and isolated perfused liver. Hepatology 1993, 18:380–388.
Tanaka J, Sato T, Kamiyama Y, et al.: Bacteremic shock: aspects of high-energy metabolism of rat liver following living Escherichia coli injection. J Surg Res 1982, 33:49–57.
Callahan LA, Stofan DA, Szweda LI, et al.: Free radicals alter maximal diaphragmatic mitochondrial oxygen consumption in endotoxin-induced sepsis. Free Radic Biol Med 2001, 30:129–138.
Markley MA, Pierro A, Eaton S: Hepatocyte mitochondrial metabolism is inhibited in neonatal rat endotoxaemia: effects of glutamine. Clin Sci (Lond) 2002, 102:337–344.
Crouser ED, Julian MW, Dorinsky PM: Ileal VO(2)-O(2) alterations induced by endotoxin correlate with severity of mitochondrial injury. Am J Respir Crit Care Med 1999, 160:1347–1353.
Crouser ED, Julian MW, Weinstein DM, et al.: Endotoxininduced ileal mucosal injury and nitric oxide dysregulation are temporally dissociated. Am J Respir Crit Care Med 2000, 161:1705–1712.
Crouser ED, Julian MW, Blaho DV, Pfeiffer DR: Endotoxininduced mitochondrial damage correlates with impaired respiratory activity. Crit Care Med 2002, 30:276–284.
Schaefer CF, Lerner MR, Biber B: Dose-related reduction of intestinal cytochrome a,a 3 induced by endotoxin in rats. Circ Shock 1991, 33:17–25.
Schaefer CF, Biber B, Lerner MR, et al.: Rapid reduction of intestinal cytochrome a,a 3 during lethal endotoxemia. J Surg Res 1991, 51:382–391.
Simonson SG, Welty-Wolf K, Huang YT, et al.: Altered mitochondrial redox responses in gram negative septic shock in primates. Circ Shock 1994, 43:34–43.
Gellerich FN, Trumbeckaite S, Opalka JR, et al.: Mitochondrial dysfunction in sepsis: evidence from bacteraemic baboons and endotoxaemic rabbits. Biosci Rep 2002, 22:99–113.
Mizobata Y, Prechek D, Rounds JD, et al.: The duration of infection modifies mitochondrial oxidative capacity in rat skeletal muscle. J Surg Res 1995, 59:165–173.
Kaneda K, Yoshioka Y, Makita K, et al.: Effects of carboxy-PTIO on systemic hemodynamics, liver energetics, and concentration of liver metabolites during endotoxic shock in rabbits: a 31P and 1H magnetic resonance spectroscopic study. Crit Care Med 1997, 25:1019–1029.
Liaudet L, Fishman D, Markert M, et al.: L-canavanine improves organ function and tissue adenosine triphosphate levels in rodent endotoxemia. Am J Respir Crit Care Med 1997, 155:1643–1648.
Revelly JP, Liaudet L, Frascarolo P, et al.: Effects of norepinephrine on the distribution of intestinal blood flow and tissue adenosine triphosphate content in endotoxic shock. Crit Care Med 2000, 28:2500–2506.
Levy B, Mansart A, Bollaert PE, et al.: Effects of epinephrine and norepinephrine on hemodynamics, oxidative metabolism, and organ energetics in endotoxemic rats. Intensive Care Med 2003, 29:292–300.
Poderoso JJ, Boveris A, Jorge MA, et al.: Function mitocondrial en el shock septico. Medicina 1978, 38:371–377.
Bergstrom J, Bostrom H, Furst P, et al.: Preliminary studies of energy-rich phosphagens in muscle from severely ill patients. Crit Care Med 1976, 4:197–204.
Liaw KY, Askanazi J, Michelson CB, et al.: Effect of injury and sepsis on high-energy phosphates in muscle and red cells. J Trauma 1980, 20:755–759.
Tresadern JC, Threlfall CJ, Wilford K, Irving MH: Muscle adenosine 5’-triphosphate and creatine phosphate concentrations in relation to nutritional status and sepsis in man. Clin Sci (Lond) 1988, 75:233–242.
Yassen KA, Galley HF, Lee A, Webster NR: Mitochondrial redox state in the critically ill. Br J Anaesth 1999, 83:325–327.
Satoi S, Kamiyama Y, Kitade H, et al.: Nitric oxide production and hepatic dysfunction in patients with postoperative sepsis. Clin Exp Pharmacol Physiol 2000, 27:197–201.
Boulos M, Astiz ME, Barua RS, Osman M: Impaired mitochondrial function induced by serum from septic shock patients is attenuated by inhibition of nitric oxide synthase and poly(ADP-ribose) synthase. Crit Care Med 2003, 31:353–358. This study finds that factors present in septic serum depress mitochondrial function in cell lines.
Szabo C, Zingarelli B, Salzman AL: Role of poly-ADP ribosyltransferase activation in the vascular contractile and energetic failure elicited by exogenous and endogenous nitric oxide and peroxynitrite. Circ Res 1996, 78:1051–1063.
Khan AU, Delude RL, Han YY, et al.: Liposomal NAD(+) prevents diminished O2 consumption by immunostimulated Caco-2 cells. Am J Physiol Lung Cell Mol Physiol 2002, 282:L1082-L1091.
Zingarelli B, O’Connor M, Wong H, et al.: Peroxynitrite-mediated DNA strand breakage activates poly-adenosine diphosphate ribosyl synthetase and causes cellular energy depletion in macrophages stimulated with bacterial lipopolysaccharide. J Immunol 1996, 156:350–358.
Ozdulger A, Cinel I, Unlu A, et al.: Poly(Adp-ribose) synthetase inhibition prevents lipopolysaccharide-induced peroxynitrite mediated damage in diaphragm. Pharmacol Res 2002, 46:67–73.
Goldfarb RD, Marton A, Szabo E, et al.: Protective effect of a novel, potent inhibitor of poly(adenosine 5’-diphosphateribose) synthetase in a porcine model of severe bacterial sepsis. Crit Care Med 2002, 30:974–980.
Cinel I, Buyukafsar K, Cinel L, et al.: The role of poly(ADPribose) synthetase inhibition in preventing endotoxemiainduced intestinal epithelial apoptosis. Pharmacol Res 2002, 46:119–127.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brealey, D., Singer, M. Mitochondrial dysfunction in sepsis. Curr Infect Dis Rep 5, 365–371 (2003). https://doi.org/10.1007/s11908-003-0015-9
Issue Date:
DOI: https://doi.org/10.1007/s11908-003-0015-9