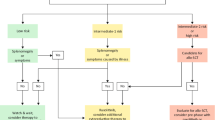
Abstract
Several international working groups cooperated to propose new diagnostic guidelines for polycythemia vera (PV), essential thrombocythemia (ET), and primary myelofibrosis (PMF) to the steering committee of the World Health Organization. Because JAK2 mutation status presents a decisive diagnostic test in PV, this feature was introduced as a major criterion. Minor criteria, such as characteristic bone marrow morphology, low erythropoietin level, and erythroid colony formation, were kept as supporting parameters. In PMF, major diagnostic criteria were established by histologic features independent of the presence of relevant fibrosis or myelofibrosis with myeloid metaplasia. JAK2 mutation status was restricted to positive findings to exclude reactive myelofibrosis. A decrease in the platelet level was proposed for ET, because vascular complications may occur at lower platelet counts. As with PMF, morphology plays a distinctive role in diagnosis, particularly for its differentiation from early-stage PV or prodromal PMF associated with thrombocytosis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References and Recommended Reading
Tefferi A, Vardiman JW: Classification and diagnosis of myeloproliferative neoplasms: the 2008 World Health Organization criteria and point-of-care diagnostic algorithms. Leukemia 2008, 22:14–22.
Tefferi A, Thiele J, Orazi A, et al.: Proposals and rationale for revision of the World Health Organization diagnostic criteria for polycythemia vera, essential thrombocythemia, and primary myelofibrosis: recommendations from an ad hoc international expert panel. Blood 2007, 110:1092–1097.
Levine RL, Pardanani A, Tefferi A, et al.: Role of JAK2 in the pathogenesis and therapy of myeloproliferative disorders. Nat Rev Cancer 2007, 7:673–683.
Nelson ME, Steensma DP: JAK2 V617F in myeloid disorders: What do we know now, and where are we headed? Leuk Lymphoma 2006, 47:177–194.
Wadleigh M, Gilliland DG: The JAK2V617F tyrosine kinase mutation in myeloproliferative disorders: summary of published literature and a perspective. Curr Hematol Malig Rep 2006, 1:75–80.
Scott LM, Tong W, Levine RL, et al.: JAK2 exon 12 mutations in polycythemia vera and idiopathic erythrocytosis. N Engl J Med 2007, 356:459–468.
Levine RL, Wadleigh M, Cools J, et al.: Activating mutation in the tyrosine kinase JAK2 in polycythemia vera, essential thrombocythemia, and myeloid metaplasia with myelofibrosis. Cancer Cell 2005, 7:387–397.
Tefferi A, Vardiman JW: The diagnostic interface between histology and molecular tests in myeloproliferative disorders. Curr Opin Hematol 2007, 14:115–122.
Nussenzveig RH, Swierczek SI, Jelinek J, et al.: Polycythemia vera is not initiated by JAK2V617F mutation. Exp Hematol 2007, 35:32–38.
Spivak JL: Polycythemia vera: myths, mechanisms, and management. Blood 2002, 100:4272–4290.
Tefferi A: The diagnosis of polycythemia vera: new tests and old dictums. Best Pract Res Clin Haematol 2006, 19:455–469.
Pearson TC: Evaluation of diagnostic criteria in polycythemia vera. Semin Hematol 2001, 38:21–24.
Murphy S: Diagnostic criteria and prognosis in polycythemia vera and essential thrombocythemia. Semin Hematol 1999, 36:9–13.
James C, Ugo V, Le Couedic JP, et al.: A unique clonal JAK2 mutation leading to constitutive signalling causes polycythaemia vera. Nature 2005, 434:1144–1148.
Jones AV, Kreil S, Zoi K, et al.: Widespread occurrence of the JAK2 V617F mutation in chronic myeloproliferative disorders. Blood 2005, 106:2162–2168.
Birgegard G, Wide L: Serum erythropoietin in the diagnosis of polycythaemia and after phlebotomy treatment. Br J Haematol 1992, 81:603–606.
Mossuz P, Girodon F, Donnard M, et al.: Diagnostic value of serum erythropoietin level in patients with absolute erythrocytosis. Haematologica 2004, 89:1194–1198.
Dobo I, Donnard M, Girodon F, et al.: Standardization and comparison of endogenous erythroid colony assays performed with bone marrow or blood progenitors for the diagnosis of polycythemia vera. Hematol J 2004, 5:161–167.
Gruppo Italiano Studio Policitemia: Polycythemia vera: the natural history of 1213 patients followed for 20 years. Ann Intern Med 1995, 123:656–664.
Ruggeri M, Tosetto A, Frezzato M, et al.: The rate of progression to polycythemia vera or essential thrombocythemia in patients with erythrocytosis or thrombocytosis. Ann Intern Med 2003, 139:470–475.
Thiele J, Kvasnicka HM, Diehl V: Initial (latent) polycythemia vera with thrombocytosis mimicking essential thrombocythemia. Acta Haematol 2005, 113:213–219.
Jantunen R, Juvonen E, Ikkala E, et al.: Development of erythrocytosis in the course of essential thrombocythemia. Ann Hematol 1999, 78:219–222.
Shih LY, Lee CT: Identification of masked polycythemia vera from patients with idiopathic marked thrombocytosis by endogenous erythroid colony assay. Blood 1994, 83:744–748.
Georgii A, Buesche G, Kreft A: The histopathology of chronic myeloproliferative diseases. Baillieres Clin Haematol 1998, 11:721–749.
Thiele J, Kvasnicka HM: Diagnostic impact of bone marrow histopathology in polycythemia vera (PV). Histol Histopathol 2005, 20:317–328.
Thiele J, Kvasnicka HM, Muehlhausen K, et al.: Polycythemia rubra vera versus secondary polycythemias. A clinicopathological evaluation of distinctive features in 199 patients. Pathol Res Pract 2001, 197:77–84.
Ellis JT, Peterson P, Geller SA, et al.: Studies of the bone marrow in polycythemia vera and the evolution of myelofibrosis and second hematologic malignancies. Semin Hematol 1986, 23:144–155.
Thiele J, Kvasnicka HM, Vardiman J: Bone marrow histopathology in the diagnosis of chronic myeloproliferative disorders: a forgotten pearl. Best Pract Res Clin Haematol 2006, 19:413–437.
Barosi G, Mesa RA, Thiele J, et al.: Proposed criteria for the diagnosis of post-polycythemia vera and post-essential thrombocythemia myelofibrosis: a consensus statement from the International Working Group for Myelofibrosis Research and Treatment. Leukemia 2008, 22:437–438.
Murphy S, Peterson P, Iland H, et al.: Experience of the Polycythemia Vera Study Group with essential thrombocythemia: a final report on diagnostic criteria, survival, and leukemic transition by treatment. Semin Hematol 1997, 34:29–39.
Lengfelder E, Hochhaus A, Kronawitter U, et al.: Should a platelet limit of 600 × 10(9)/l be used as a diagnostic criterion in essential thrombocythaemia? An analysis of the natural course including early stages. Br J Haematol 1998, 100:15–23.
Sacchi S, Vinci G, Gugliotta L, et al.: Diagnosis of essential thrombocythemia at platelet counts between 400 and 600×10(9)/L. Gruppo Italiano Malattie Mieloproliferative Croniche (GIMMC). Haematologica 2000, 85:492–495.
Pardanani AD, Levine RL, Lasho T, et al.: MPL515 mutations in myeloproliferative and other myeloid disorders: a study of 1182 patients. Blood 2006, 108:3472–3476.
Thiele J, Kvasnicka HM: Clinicopathological criteria for differential diagnosis of thrombocythemias in various myeloproliferative disorders. Semin Thromb Hemost 2006, 32:219–230.
Florena AM, Tripodo C, Iannitto E, et al.: Value of bone marrow biopsy in the diagnosis of essential thrombocythemia. Haematologica 2004, 89:911–919.
Gianelli U, Vener C, Raviele PR, et al.: Essential thrombocythemia or chronic idiopathic myelofibrosis? A single-center study based on hematopoietic bone marrow histology. Leuk Lymphoma 2006, 47:1774–1781.
Kreft A, Buche G, Ghalibafian M, et al.: The incidence of myelofibrosis in essential thrombocythaemia, polycythaemia vera and chronic idiopathic myelofibrosis: a retrospective evaluation of sequential bone marrow biopsies. Acta Haematol 2005, 113:137–143.
Barosi G: Myelofibrosis with myeloid metaplasia: diagnostic definition and prognostic classification for clinical studies and treatment guidelines. J Clin Oncol 1999, 17:2954–2970.
Tefferi A: Myelofibrosis with myeloid metaplasia. N Engl J Med 2000, 342:1255–1265.
Dingli D, Mesa RA, Tefferi A: Myelofibrosis with myeloid metaplasia: new developments in pathogenesis and treatment. Intern Med 2004, 43:540–547.
Pikman Y, Lee BH, Mercher T, et al.: MPLW515L is a novel somatic activating mutation in myelofibrosis with myeloid metaplasia. PLoS Med 2006, 3:e270.
Thiele J, Kvasnicka HM: Hematopathologic findings in chronic idiopathic myelofibrosis. Semin Oncol 2005, 32:380–394.
Dickstein JI, Vardiman JW: Issues in the pathology and diagnosis of the chronic myeloproliferative disorders and the myelodysplastic syndromes. Am J Clin Pathol 1993, 99:513–525.
Buhr T, Busche G, Choritz H, et al.: Evolution of myelofibrosis in chronic idiopathic myelofibrosis as evidenced in sequential bone marrow biopsy specimens. Am J Clin Pathol 2003, 119:152–158.
Thiele J, Kvasnicka HM, Schmitt-Graeff A, et al.: Dynamics of fibrosis in chronic idiopathic (primary) myelofibrosis during therapy: a follow-up study on 309 patients. Leuk Lymphoma 2003, 44:949–953.
Thiele J, Kvasnicka HM, Facchetti F, et al.: European consensus on grading bone marrow fibrosis and assessment of cellularity. Haematologica 2005, 90:1128–1132.
Thiele J, Kvasnicka HM, Diehl V: Standardization of bone marrow features-Does it work in hematopathology for histological discrimination of different disease patterns? Histol Histopathol 2005, 20:633–644.
Wilkins BS, Erber WN, Bareford D, et al.: Bone marrow pathology in essential thrombocythemia: interobserver reliability and utility for identifying disease subtypes. Blood 2008, 111:60–70.
Manoharan A, Horsley R, Pitney WR: The reticulin content of bone marrow in acute leukaemia in adults. Br J Haematol 1979, 43:185–190.
Spivak JL, Silver RT: The revised World Health Organization diagnostic criteria for polycythemia vera, essential thrombocytosis and primary myelofibrosis: an alternative proposal. Blood 2008, 112:231–239.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thiele, J., Kvasnicka, H.M. The 2008 WHO diagnostic criteria for polycythemia vera, essential thrombocythemia, and primary myelofibrosis. Curr Hematol Malig Rep 4, 33–40 (2009). https://doi.org/10.1007/s11899-009-0005-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11899-009-0005-6