Abstract
The application of portable echocardiography to the screening of asymptomatic children and young adults for rheumatic heart disease (RHD) in developing countries indicates that the disease may affect 62 million to 78 million individuals worldwide, which could potentially result in 1.4 million deaths per year from RHD and its complications. The World Heart Federation has developed a guideline for the echocardiographic diagnosis of RHD in asymptomatic individuals without a history of acute rheumatic fever (ARF) in order to ensure the reliability, comparability, and reproducibility of findings of the echocardiographic screening studies. Early studies suggest that a third of individuals with asymptomatic subclinical RHD revert to normal echocardiographic findings on repeat testing after 6–12 months, suggesting that repeat echocardiography may be necessary to confirm the findings prior to consideration of interventions such as antibiotic prophylaxis. It is not known, however, whether echocardiographic screening for asymptomatic subclinical RHD or the introduction of antibiotic prophylaxis for affected individuals improves the prognosis of RHD. Furthermore, the cost-effectiveness of this screening method has not been established in the vast majority of affected countries. Therefore, echocardiographic screening for asymptomatic subclinical RHD remains a research tool until studies of impact on prognosis and cost-effectiveness are conducted.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The world is divided into countries and communities where the incidence of acute rheumatic fever (ARF)/rheumatic heart disease (RHD) is lower than 1 per 100, 000 per year (ie, developed countries) and those where the disease is endemic at a rate of greater than 10 per 100,000 per year (ie, the majority of developing countries and some indigenous communities in industrialized countries) [1]. The use of portable echocardiography to screen asymptomatic children and young adults in these endemic countries for RHD has shown that the disease may affect 62 million to 78 million individuals worldwide which could potentially result in 1.4 million deaths per year from RHD and its complications [2••]. The majority of these people are living in countries with limited access to cardiac surgery [3••, 4]. Thus, the prospect that detecting the earliest changes of RHD in high prevalent populations followed by institution of secondary prophylaxis to retard progress to overt disease has been met with great enthusiasm, resulting in a “rising tide” of echocardiographic screening studies for RHD over the past 15 years in Africa, South Asia, Middle East, South America, and Australasia [2••, 5•, 6, 7••, 8–16]. These studies have not only revealed a large burden of subclinical disease but also demonstrated the superiority of echocardiography over auscultation in detecting early rheumatic structural and functional changes of the mitral and aortic valves [17].
For a disease to be suitable for screening as a tool for prevention, it must satisfy the following conditions: (1) there must be evidence of a significant burden of disease, (2) the disease must have an initial latent stage, (3) which can be detected by simple, accessible, and sensitive tests, (4) the early stages must be treatable by adequate therapy, and (5) there must be evidence that early intervention improves prognosis in a cost-effective manner. Whilst the echocardiographic studies confirm the heavy burden of RHD in endemic countries, the additional conditions for screening of RHD require further discussion which is the subject of this commentary.
Natural History of Latent Stages of RHD
Subclinical carditis (echocardiographic lesions in the absence of a murmur) in the context of ARF is associated with either persistence or regression of disease in follow-up studies. Proven carditis with Aschoff nodules in the absence of an ante-mortem murmur was first described in autopsy specimens before the advent of echocardiography [18]. The Doppler echocardiographic features of subclinical rheumatic carditis [19] were characterized by Minich, while Figueroa and others were the first to report the natural history of subclinical carditis in patients with ARF [20]. Long-term follow-up of subclinical carditis associated with ARF determined that valve lesions persist in 30% of patients after 5 years [21–23]. A systematic review of studies of subclinical carditis also showed that approximately half of the patients diagnosed with subclinical carditis at time of ARF showed persistence or deterioration of their carditis in the following 2 years [24]. As a result stricter echocardiographic criteria were proposed using both qualitative and quantitative elements in an effort to identify those patients who are likely to have significant irreversible damage of the heart valves [25–27]. The Australian and New Zealand Guidelines for Rheumatic Fever Diagnosis, Management and Secondary Prevention include subclinical carditis as a major criterion for the diagnosis of ARF in endemic communities, [28–30] and recommend that all patients with suspected or confirmed ARF undergo echocardiography.
The natural history of subclinical carditis in asymptomatic populations without a previous history of ARF, however, is less well characterized. Subclinical RHD may be defined as features of RHD on echocardiography in the absence of symptoms and signs of heart valve disease. A study of 12,050 asymptomatic school children screened by cardiac auscultation revealed 168 participants with a pathological murmur of RHD who were re-examined 4 years later [31, 32]. On review only 3% had progressive disease, whilst 57% had a persistent murmur and the remaining 40% had a normal cardiovascular examination. This study raised the possibility that the natural history of asymptomatic RHD detected on screening may be associated with an apparent reversion to normal in a substantial proportion of individuals.
There are at least 3 studies that have reported on short-term progress of subclinical RHD detected by echocardiography [2••, 7••, 10, 33]. Bhaya et al. showed that within 2 years of follow-up, isolated mitral regurgitation was likely to resolve in the absence of valve deformities. In comparison, mitral regurgitation coexisting with valve deformities was more likely to persist [33]. This implies that disease classified as definite by echocardiography (ie, with morphological and functional changes) is more likely to persist than borderline or possible categories (ie, with either minor morphological or functional changes). On follow up of 100 children over 3 to 27 months in the Rheumatic Heart Echocardiograms Utilisation and Monitoring Actuarial Trends in Indian Children (RHEUMATIC) study, the severity of subclinical RHD was non-progressive in 68% of children, while the changes worsened in 4% and regressed in 28% of children [7••]. A similar pattern was reflected in the follow-up of participants in the probable and possible categories in Nicaragua [2••]. In these categories, 69% persisted, 9% worsened whilst total regression was seen in 32%. These studies indicate that early forms of suspected RHD that are detected by either auscultation or echocardiography in asymptomatic populations may be associated with resolution of changes in 30%–40% of subjects. Follow-up examination may be needed to identify the ‘persistors and progressors’ who may be eligible for secondary prophylaxis.
Echocardiography as a Screening Test for RHD
Early detection of RHD in school children is traditionally done by listening for murmurs by stethoscope, followed by echocardiographic confirmation only in suspected cases. Recently, systematic echocardiographic screening in children and adults in many endemic countries and communities, have achieved a case detection rate by echocardiography approximately 10-fold that achieved by clinical examination only. Portable echocardiography is considered to be a more sensitive test for detecting the early stage of RHD than cardiac auscultation.
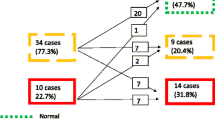
A key element to screening programs has been the need for a unified set of echocardiographic criteria for the diagnosis of subclinical RHD. In 2005, a joint National Institutes of Health-World Health Organization (NIH-WHO) working group developed standardized surveillance protocols for Group A streptococcal diseases, which included a diagnostic definition of RHD on echocardiography [34]. This was a consensus definition which needed validation in field studies [35••]. The NIH-WHO criteria were subsequently shown to be less sensitive than a combined set of criteria [36]. The importance of a unified set of criteria is demonstrated in Figure 1 which expresses the variability of the prevalence of RHD in 2170 Mozambican children using auscultation and 4 different echocardiographic criteria [14, 36, 37]. A standardized evidence-based guideline for the echocardiographic diagnosis of RHD has been developed by the World Heart Federation to improve the reliability, comparability and reproducibility of echocardiographic screening studies [38••]. It is envisaged that the new guideline may reduce the potential for the over-diagnosis of RHD in screening studies [39, 40]. A recent study highlighted this point by demonstrating the importance of excluding physiological valve regurgitation and congenital valve anomalies which may be present in at least 1% of normal children [6]. These guidelines provide a reproducible method which can be utilized to follow people with subclinical lesions detected in screening studies to determine progression of RHD and recurrence rates of ARF [41, 42, 43••].
Echocardiographic Screening Protocols
The first study to use Doppler echocardiography as the primary modality for the evaluation of the prevalence of RHD was performed in 1996 in rural schools of Kenya (Table 1) [9, 15]. The subsequent echocardiographic studies have used different screening protocols. A recent study conducted in Uganda reported a 2 minute screening echocardiogram of the left-sided valves by a single operator followed by detailed echocardiography of selected participants at a tertiary center, which allows the screening of over 200 individuals per day [5•]. Other investigators have reported an average examination time of 3 minutes, 31 seconds in a targeted echocardiography examination of the mitral and aortic valve [8].
An abbreviated echocardiographic protocol was employed in the re-assessment of the original echocardiograms from the Mozambique study (Fig. 1) [37]. The study evaluated the value of a reference set of criteria (defined as a combination of Doppler and morphological rheumatic features of the aortic and/or mitral valves) [36] compared with an easy-to-use single mitral regurgitation jet-length criterion (simplified set of criteria). This study was able to demonstrate that in comparison with the previously published reference criteria, the simplified approach yielded a sensitivity of 73% and a positive predictive value of 92% for case detection. It thus appears feasible that a simple echocardiography-based screening protocol for RHD could be developed to allow for rapid and appropriate detection of a significant number of RHD cases on site.
Protocols including clinical auscultation have clearly demonstrated the improved yield of subclinical valve lesions by echocardiography compared with auscultation [2••, 6, 7••, 10, 14]. An auscultation protocol involving 2 auscultators (novice and experienced) in Tonga found that auscultatory screening alone missed more than half of the pathological lesions compared with echocardiography [13]. This may be reflected in the prevalence rates, which are substantially lower in auscultation studies compared with those that have employed echocardiography as a first-line screening technique [9, 11, 12, 44–48]. Notable exceptions are the studies from Yemen and Pakistan [9, 12] which may reflect a heavy burden of severe RHD due to the high intensity of social conditions of poverty [49].
Cost-Effectiveness and Impact on Prognosis of Screening for RHD
A preliminary study from Fiji suggests that the direct costs of echocardiographic screening per patient detected using a combined auscultation and echocardiographic protocol was US $59 [8]. These costs, which appear modest in the case of Fiji, need to be viewed in the context of a national program for the prevention and control of RHD. Furthermore, the additional costs for screening would be justifiable if the prognosis of RHD was improved. It is not yet proven that penicillin prophylaxis for all children with subclinical rheumatic valve lesions is the best option in clinical practice. Therefore, the case for the uptake of RHD screening in public health programs will only be answered once the impact of detecting subclinical lesions and early effective secondary prophylaxis has been determined.
Adults may be the appropriate target group for screening, rather than children.
It has been suggested that, as children represent only 15%–20% of the RHD load [50, 51], screening be extended to older age groups [52]. The first studies to do so have demonstrated important findings [2••, 53•, 54]. Paar et al. found a prevalence of 22:1000(8–37:1000 95% CI) in adults aged 20–35 years despite far more stringent criteria than those used in children. A similar prevalence of 18.6:1000 (10.3–30.6:1000 95% CI) was demonstrated in urban and suburban Chinese adults aged 18–74. Extrapolation using census data suggests that up to 2 million Chinese adults may have RHD [54]. Finally the high prevalence of RHD in adults living in endemic countries has been confirmed in a study examining women aged 15–42 years in antenatal facilities in Eritrea where a prevalence of 23:1000 (95% CI 0.7–3.9) was found [53•]. These data show a higher than predicted subclinical load in asymptomatic and pregnant adults who may need to be prioritized in future studies of the burden of RHD in endemic countries.
New Developments
Although the cost of portable machines is appreciably less than conventional echocardiograms machines, they are still prohibitively expensive for the health systems of endemic countries. The advent of hand-held echocardiogram machines ushers in a new possibility for their use in screening programs, amongst other point of care services. The WHF criteria require an accurate assessment of Doppler gradients which currently these machines are not able to provide. Thus, the incorporation of Doppler capability in hand-held devices is required to make them useful in screening for RHD.
Although the echocardiograms in the many of screening programs were performed by cardiologists, several employed either medical students or technologists to perform echocardiograms [2••, 8]. A training module for nurses to perform RHD-screening echocardiograms in resource-poor settings was piloted in Fiji [55] and found that nurses performed well in assessing mitral regurgitation by portable echocardiography. The WHF criteria will provide a reference for further training for nurses or community health workers, of particular relevance in resource-limited settings.
Much of the recent work has focused on population-based prevalence studies of RHD. A recent study provided much needed incident data [56••]. The Heart of Soweto study reported on the incidence and clinical characteristics of newly diagnosed RHD in adults aged 30–55 years from an urban African community and found an estimated incidence of 23.5 cases/100,000 per annum. These patients were moderate to severely affected with 17% having systolic dysfunction (EF< 45%), a further 18% with pulmonary hypertension as evidenced by elevated right-sided pressure, and 22% of this cohort requiring valve surgery within 1 year. A further 26% were admitted for initial diagnosis of suspected bacterial endocarditis within 30 months.
It has previously been reported that RHD follows a malignant course in Africa and India [57]. Oli et al. report a mean age of death in his patients of 25.89 years (SD 11.05) with the median age for males being only 19 years (IQ 16–32). A particularly disturbing late diagnosis of RHD is that made perinatally. Almost 50% mortality was noted in group of women with mitral stenosis in Senegal, with an overall maternal mortality of 34% in pregnant patients with RHD [58].
It is these reports and others [59–62] detailing the severe outcomes and patterns of valvulopathies associated with RHD that have stimulated renewed efforts to delineate the contemporary clinical epidemiology and outcome of the RHD. This is the aim of the Global Rheumatic Heart Disease Registry (REMEDY), which is a project arising from the Awareness, Surveillance, Advocacy and Prevention (A.S.A.P) program instituted by the Pan-African Society of Cardiology (PASCAR) [63, 64]. The REMEDY study aims to provide comprehensive and contemporary clinical features and outcome of patients with RHD and will help in the development of strategies to prevent and manage RHD and its complications.
Conclusion
There is clear evidence that the burden of RHD is high in developing countries and indigenous communities in some industrialized countries. RHD has an early latent course which is characterized by normalization in 30%–40% of individuals, which makes it mandatory to confirm the presence of subclinical disease after a period 6–12 months before considering the use of interventions such as antibiotic prophylaxis. Portable echocardiography is a promising screening test with a high sensitivity for the detection of early forms of the disease. However, it is not known whether the treatment of asymptomatic subclinical RHD with antibiotic prophylaxis improves the prognosis of the disease. Studies are needed to assess the impact of screening of RHD on prognosis before consideration for incorporation in public health programs for the prevention and control of the disease.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Tibazarwa KB, Volmink JA, Mayosi BM. Incidence of acute rheumatic fever in the world: a systematic review of population-based studies. Heart. 2008;94:1534–40.
•• Paar JA, Berrios NM, Rose JD, Caceres M, Pena R, Perez W, et al. Prevalence of rheumatic heart disease in children and young adults in Nicaragua. Am J Cardiol. 2010;105:1809–14. First screening study to include young adults as well as early follow-up data.
•• Jackson SJ, Steer AC, Campbell H. Systematic Review: estimation of global burden of non-suppurative sequelae of upper respiratory tract infection: rheumatic fever and post-streptococcal glomerulonephritis. Trop Med Int Health. 2011;16:2–11. Important estimates on burden of disease of rheumatic heart disease.
Mocumbi AO. The challenges of cardiac surgery for African children. Cardiovasc J Afr. 2012;23:165–7.
• Beaton A, Okello E, Lwabi P, Mondo C, McCarter R, Sable C. Echocardiography screening for rheumatic heart disease in Ugandan schoolchildren. Circulation. 2012;125:3127–32. Recent article employing a rapid echocardiographic technique.
Webb RH, Wilson NJ, Lennon DR, Wilson EM, Nicholson RW, Gentles TL, et al. Optimizing echocardiographic screening for rheumatic heart disease in New Zealand: not all valve disease is rheumatic. Cardiol Young. 2011;21:436–43.
•• Saxena A, Ramakrishnan S, Roy A, Seth S, Krishnan A, Misra P, et al. Prevalence and outcome of subclinical rheumatic heart disease in India: the RHEUMATIC (Rheumatic Heart Echo Utilisation and Monitoring Actuarial Trends in Indian Children) study. Heart. 2011;97:2018–22. One of the recent articles looking at short term follow-up as well as risk factors assoicated with positive screen results in India.
Reeves BM, Kado J, Brook M. High prevalence of rheumatic heart disease in Fiji detected by echocardiography screening. J Paediatr Child Health. 2011;47:473–8.
Ba-Saddik IA, Munibari AA, Al-Naqeeb MS, Parry CM, Hart CA, Cuevas LE, et al. Prevalence of rheumatic heart disease among school-children in Aden. Yemen Ann Trop Paediatr. 2011;31:37–46.
Bhaya M, Panwar S, Beniwal R, Panwar RB. High prevalence of rheumatic heart disease detected by echocardiography in school children. Echocardiography. 2010;27:448–53.
Steer AC, Kado J, Wilson N, Tuiketei T, Batzloff M, Waqatakirewa L, et al. High prevalence of rheumatic heart disease by clinical and echocardiographic screening among children in Fiji. J Heart Valve Dis. 2009;18:327–35; discussion 36.
Sadiq M, Islam K, Abid R, Latif F, Rehman AU, Waheed A, et al. Prevalence of rheumatic heart disease in school children of urban Lahore. Heart. 2009;95:353–7.
Carapetis JR, Hardy M, Fakakovikaetau T, Taib R, Wilkinson L, Penny DJ, et al. Evaluation of a screening protocol using auscultation and portable echocardiography to detect asymptomatic rheumatic heart disease in Tongan schoolchildren. Nat Clin Pract Cardiovasc Med. 2008;5:411–7.
Marijon E, Ou P, Celermajer DS, Ferreira B, Mocumbi AO, Jani D, et al. Prevalence of rheumatic heart disease detected by echocardiographic screening. N Engl J Med. 2007;357:470–6.
Anabwani GM, Bonhoeffer P. Prevalence of heart disease in school children in rural Kenya using color-flow echocardiography. East Afr Med J. 1996;73:215–7.
Zühlke LJ, Mayosi BM. The challenge of screening for asymptomatic rheumatic heart disease in South Africa. SA Heart. 2009;6:100–3.
Marijon E, Tafflet M, Jouven X. Time to use ultrasound and not stethoscopes for rheumatic heart disease screening. Nat Clin Pract Cardiovasc Med. 2008;5:E1–3.
Tedeschi CG, Wagner BM. The problem of subclinical rheumatic carditis. Am J Med Sci. 1956;231:382–8.
Minich LL, Tani LY, Pagotto LT, Shaddy RE, Veasy LG. Doppler echocardiography distinguishes between physiologic and pathologic "silent" mitral regurgitation in patients with rheumatic fever. Clin Cardiol. 1997;20:924–6.
Figueroa FE, Fernandez MS, Valdes P, Wilson C, Lanas F, Carrion F, et al. Prospective comparison of clinical and echocardiographic diagnosis of rheumatic carditis: long-term follow-up of patients with subclinical disease. Heart. 2001;85:407–10.
Lanna CC, Tonelli E, Barros MV, Goulart EM, Mota CC. Subclinical rheumatic valvitis: a long-term follow-up. Cardiol Young. 2003;13:431–8.
Karaaslan S, Demiroren S, Oran B, Baysal T, Baspinar O, Ucar C. Criteria for judging the improvement in subclinical rheumatic valvitis. Cardiol Young. 2003;13:500–5.
Ozkutlu S, Hallioglu O, Ayabakan C. Evaluation of subclinical valvar disease in patients with rheumatic fever. Cardiol Young. 2003;13:495–9.
Tubridy-Clark M, Carapetis JR. Subclinical carditis in rheumatic fever: a systematic review. Int J Cardiol. 2007;119:54–8.
Caldas AM, Terreri MT, Moises VA, Silva CM, Carvalho AC, Hilario MO. The case for utilizing more strict quantitative Doppler echocardiographic criterions for diagnosis of subclinical rheumatic carditis. Cardiol Young. 2007;17:42–7.
Wilson N. Echocardiography and subclinical carditis: guidelines that increase sensitivity for acute rheumatic fever. Cardiol Young. 2008;18:565–8.
Vijayalakshmi IB, Vishnuprabhu RO, Chitra N, Rajasri R, Anuradha TV. The efficacy of echocardiographic criterions for the diagnosis of carditis in acute rheumatic fever. Cardiol Young. 2008;18:586–92.
The Australian guideline for prevention, diagnosis, and management of acute rheumatic fever and rheumatic heart disease. 2012. Available At: http://www.rhdaustralia.org.au/sites/default/files/guideline_0.pdf. Accessed accessed on November 13, 2012.
Walsh W, Brown A, Carapetis J. The diagnosis and management of chronic rheumatic heart disease–an Australian guideline. Heart Lung Circ. 2008;17:271–89.
Atatoa-Carr P, Lennon D, Wilson N. Rheumatic fever diagnosis, management, and secondary prevention: a New Zealand guideline. N Z Med J. 2008;121:59–69.
Cohen M, Pocock WA, Lakier JB, McLaren MJ, Lachman AS, Barlow JB. Four year follow-up of black schoolchildren with non-ejection systolic clicks and mitral systolic murmurs. Am Heart J. 1978;95:697–701.
McLaren MJ, Hawkins DM, Koornhof HJ, Bloom KR, Bramwell-Jones DM, Cohen E, et al. Epidemiology of rheumatic heart disease in black shcoolchildren of Soweto. Johannesburg Br Med J. 1975;3:474–8.
Bhaya M, Beniwal R, Panwar S, Panwar RB. Two years of follow-up validates the echocardiographic criteria for the diagnosis and screening of rheumatic heart disease in asymptomatic populations. Echocardiography. 2011;28:929–33.
Carapetis J PJ, Cherian T. Standardisation of epidemiologic protocols for surveillance of post-streptococcal sequelae:acute rheumatic fever, rheumatic heart disease and acute post-streptococcal glomerulonephritis. In: Protocols for surveillance of Streptococcal pyogenes infections and their sequelae. Bathesda, USA. 2010. Available at: http://www.niaid.nih.gov/topics/streptococcal/pages/protocols.aspx. Accessed October 11, 2012.
•• Marijon E, Mirabel M, Celermajer DS, Jouven X. Rheumatic heart disease. Lancet. 2012;379:953–64. Excellent overview of acute rheumatic fever, rheumatic heart disease, and covers pathogenesis,diagnosis, propylaxis. and management.
Marijon E, Celermajer DS, Tafflet M, El-Haou S, Jani DN, Ferreira B, et al. Rheumatic heart disease screening by echocardiography: the inadequacy of World Health Organization criteria for optimizing the diagnosis of subclinical disease. Circulation. 2009;120:663–8.
Mirabel M, Celermajer DS, Ferreira B, Tafflet M, Perier MC, Karam N, et al. Screening for rheumatic heart disease: evaluation of a simplified echocardiography-based approach. Eur Heart J Cardiovasc Imaging. 2012;13:1024–29.
•• Remenyi B, Wilson N, Steer A, Ferreira B, Kado J, Kumar K, et al. World Heart Federation criteria for echocardiographic diagnosis of rheumatic heart disease an evidence-based guideline. Nat Rev Cardiol. 2012;9:297–309. Landmark paper bringing together experts and evidence to produce guidelines for echocardiographic screening of RHD.
Pandve HT, Bhawalkar J, Banerjee A, Bhuyar P. Can echocardiography be used for screening of rheumatic heart disease? Indian J Community Med. 2009;34:79.
Marijon E, Jouven X. Early detection of rheumatic heart disease and prevention of heart failure in sub-Saharan Africa. J Am Coll Cardiol. 2008;51:1125–6. author reply 6.
Sliwa K, Zilla P. Rheumatic heart disease: the tip of the iceberg. Circulation. 2012;125:3060–2.
King A. Public health: value of echocardiographic screening for RHD in Ugandan children. Nat Rev Cardiol. 2012;9:433.
•• Carapetis JR, Zuhlke LJ. Global research priorities in rheumatic fever and rheumatic heart disease. Ann Pediatr Cardiol. 2011;4:4–12. First paper to detail the research priorities needed in the next years to consolidate the work of the recent past and bring about meaningful change in RHD patterns and prevalence.
Longo-Mbenza B, Bayekula M, Ngiyulu R, Kintoki VE, Bikangi NF, Seghers KV, et al. Survey of rheumatic heart disease in school children of Kinshasa town. Int J Cardiol. 1998;63:287–94.
Thakur JS, Negi PC, Ahluwalia SK, Vaidya NK. Epidemiological survey of rheumatic heart disease among school children in the Shimla Hills of northern India: prevalence and risk factors. J Epidemiol Community Health. 1996;50:62–7.
Vashistha VM, Kalra A, Kalra K, Jain VK. Prevalence of rheumatic heart disease in school children. Indian Pediatr. 1993;30:53–6.
Kumar P, Garhwal S, Chaudhary V. Rheumatic heart disease: a school survey in a rural area of Rajasthan. Indian Heart J. 1992;44:245–6.
Shrestha UK, Bhattarai TN, Pandey MR. Prevalence of rheumatic fever and rheumatic heart disease in school children in a rural community of the hill region of Nepal. Indian Heart J. 1991;43:39–41.
Steer AC, Carapetis JR, Nolan TM, Shann F. Systematic review of rheumatic heart disease prevalence in children in developing countries: the role of environmental factors. J Paediatr Child Health. 2002;38:229–34.
Carapetis JR. Pediatric rheumatic heart disease in the developing world: echocardiographic vs clinical screening. Nat Clin Pract Cardiovasc Med. 2008;5:74–5.
Carapetis JR. Rheumatic heart disease in developing countries. N Engl J Med. 2007;357:439–41.
Veasy LG, Tani LY, Minich L. The logic for extending the use of echocardiography beyond childhood to detect subclinical rheumatic heart disease. Cardiol Young. 2009;19:30–3.
• Otto H, Saether SG, Banteyrga L, Haugen BO, Skjaerpe T. High prevalence of subclinical rheumatic heart disease in pregnant women in a developing country: an echocardiographic study. Echocardiography. 2011;28:1049–53. Recent report detailing echocardiographic screening in a particularly vulnerable population: pregnant women.
Zhimin W, Yubao Z, Lei S, Xianliang Z, Wei Z, Li S, et al. Prevalence of chronic rheumatic heart disease in Chinese adults. Int J Cardiol. 2006;107:356–9.
Colquhoun SM, Carapetis JR, Kado JH, Reeves BM, Remenyi B, May W, et al. Pilot study of nurse-led rheumatic heart disease echocardiography screening in Fiji - a novel approach in a resource-poor setting. Cardiology in the Young. 2012;1–7. doi:10.1017/S1047951112001321.
•• Sliwa K, Carrington M, Mayosi BM, Zigiriadis E, Mvungi R, Stewart S. Incidence and characteristics of newly diagnosed rheumatic heart disease in Urban African adults: insights from the Heart of Soweto Study. Eur Heart J. 2010;31:719–27. Only incidence study of rheumatic heart disease in an urban African population in the past decade.
Gunther G, Asmera J, Parry E. Death from rheumatic heart disease in rural Ethiopia. Lancet. 2006;367:391.
Diao M, Kane A, Ndiaye MB, Mbaye A, Bodian M, Dia MM, et al. Pregnancy in women with heart disease in sub-Saharan Africa. Arch Cardiovasc Dis. 2011;104:370–4.
Sani MU, Karaye KM, Borodo MM. Prevalence and pattern of rheumatic heart disease in the Nigerian savannah: an echocardiographic study. Cardiovasc J Afr. 2007;18:295–9.
Tantchou Tchoumi JC, Butera G. Rheumatic valvulopathies occurence, pattern and follow-up in rural area: the experience of the Shisong Hospital, Cameroon. Bull Soc Pathol Exot. 2009;102:155–8.
Periwal KL, Gupta BK, Panwar RB, Khatri PC, Raja S, Gupta R. Prevalence of rheumatic heart disease in school children in Bikaner: an echocardiographic study. J Assoc Physicians India. 2006;54:279–82.
Yuko-Jowi C, Bakari M. Echocardiographic patterns of juvenile rheumatic heart disease at the Kenyatta National Hospital. Nairobi East Afr Med J. 2005;82:514–9.
Karthikeyan G, Zuhlke L, Engel M, Rangarajan S, Yusuf S, Teo K, et al. Rationale and design of a Global Rheumatic Heart Disease Registry: the REMEDY study. Am Heart J. 2012;163:535–40 e1.
Watkins DA, Omokhodion SI, Mayosi BM. The history of the Pan-African Society of Cardiology (PASCAR): the first 30 years, 1981–2011. Cardiovasc J Afr. 2011;22:122–3.
Disclosure
Conflicts of interest: L. Zühlke: none; B.M. Mayosi: has received grant support from World Heart Federation Else Krone Award, Medical Research Council of South Africa, Lily and Ernst Hausmann Research Trust, Lifehealthcare Foundation, and Thrasher Foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Valvular Heart Disease
Rights and permissions
About this article
Cite this article
Zühlke, L., Mayosi, B.M. Echocardiographic Screening for Subclinical Rheumatic Heart Disease Remains a Research Tool Pending Studies of Impact on Prognosis. Curr Cardiol Rep 15, 343 (2013). https://doi.org/10.1007/s11886-012-0343-1
Published:
DOI: https://doi.org/10.1007/s11886-012-0343-1