Abstract
Calcium score (CS) is a useful tool in evaluating the risk of cardiovascular events in asymptomatic patients. The absence of detectable calcification determines excellent cardiovascular prognosis, with event rates lower than that of negative stress studies, probably due to the latter’s inability to detect nonobstructive coronary artery disease (CAD). There are few primary prevention medications that would be cost-effective in such a low-risk patient population. The interval for retesting patients with zero CS is still open for debate but it should not be in less than 4 to 5 years. CS should not be used to rule out obstructive CAD in symptomatic patients, as its correlation with coronary stenosis is poor and obstructive CAD is commonly found among symptomatic zero CS patients. Most studies have found very low specificity values for CS to detect obstructive CAD in symptomatic patients, meaning it has limited ability to detect the true negative cases (ie, zero CS without obstructive CAD).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
For 20 years the measurement of coronary calcification by noncontrasted cardiac CT has been used for cardiovascular risk estimation [1]. The rationale behind the ability for the calcium score (CS) to predict cardiac events is straightforward: CT can accurately quantify coronary calcification [1, 2]; calcification is very specific (although not very sensitive) for atherosclerosis [3]; the amount of coronary calcium correlates linearly with total coronary plaque burden [2, 4, 5]; and the higher the atherosclerotic burden the higher the probability of a plaque rupture resulting in a cardiovascular event [5–10]. The CS ability to predict cardiovascular events in asymptomatic individuals has been confirmed in many large clinical trials and has been shown to be greater than that of traditional risk stratification tools, such as clinical risk factor assessment and carotid intima-media thickness [8, 11–15].
CS measurements are low risk; the radiation exposure is limited to 1 to 2 mSv, and the test does not require the use of iodinated contrast agents. Conversely, the main intrinsic limitation of coronary calcification as a predictor of cardiovascular events relates to the fact that there are other variables associated with myocardial infarction besides the anatomical existence of the plaque, such as inflammation, rheological forces, and blood coagulability [16, 17]. In addition, coronary calcification represents only one of the components of atherosclerotic plaques and may develop relatively late in the natural history of atherosclerosis [3, 4, 18, 19]. Although the potential contributions of inflammatory, hormonal, metabolic, and physical factors believed to underlie coronary calcification are still incompletely understood, the process is believed to represent a natural biologic response to arterial wall injury, activated for the purposes of increasing arterial wall stiffness in response to inflammatory injury and undue deformation [4, 18, 20–22]. Therefore, the amount of calcium accumulated in any given coronary arterial segment reflects not only the magnitude of plaque burden, but also the period of time during which plaques were exposed to the factors that underlie calcification, which renders it largely dependent on age. As a result, younger subjects have proportionally higher volumes of noncalcified plaque.
The value of undetectable coronary calcification by CT (ie, zero CS) is discussed below. Most published data consider calcified plaque any cluster of at least three pixels with attenuation of more than 130 Hounsfield units (HU) at coronary topography. However, it should be noted that some experts advocate decreasing this threshold to 90 HU, especially when using multidetector CT technology instead of electron beam CT, although, this would increase an already very high negative predictive value of zero CS for cardiac events at the expense of an already very low positive predictive value.
The Excellent Prognosis of Asymptomatic Patients with Zero CS
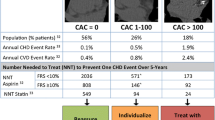
CS is best used for screening asymptomatic intermediate-risk patients. Very low risk patients (ie, <5% Framingham 10-year event rate) and high-risk patients (ie, >20% Framingham 10-year event rate) would rarely have their management changed based on the CS, rendering the test not cost-effective [11]. Conversely, intermediate-risk patients frequently have their risk reclassified after a CS [11, 14, 23, 24], and risk estimation is a fundamental part of good patient management.
The absence of detectable calcification determines an excellent prognosis among intermediate-risk asymptomatic patient populations, as has been shown in multiple large-scale clinical trials with long follow-up [13, 15, 25]. A recent meta-analysis of more than 29,000 patients with zero CS with a mean follow-up of 50 months showed that the absence of coronary calcification is associated with excellent cardiovascular prognosis, with an annualized event rate of 0.12% [26••]. This event rate, which could theoretically be extrapolated to 1.2% over 10 years, represents a lower rate than those typically observed after negative nuclear myocardial perfusion studies or good exercise capacity, as demonstrated in Table 1. A substudy of the MESA (Multi-Ethnic Study of Atherosclerosis) cohort with more than 6700 participants has demonstrated that zero CS is also more protective for cardiovascular events than a negative carotid IMT, and even patients with greater than 75 percentile of IMT but zero CS had less than 1% per year risk for events [27].
If Asymptomatic, Having a Zero CS Is Better Than Having a Normal Stress Perfusion Test
The better performance of a zero CS to rule out cardiovascular events in the long run compared with functional tests most likely derives from the fact that the latter depend on ischemia for coronary artery disease (CAD) detection, missing a large segment of the at-risk population with nonobstructive CAD, who would reliably be detected by anatomical tests such as the CS. The importance of subclinical atherosclerosis is well known and highlighted by the finding that almost half of acute coronary events occur in previously asymptomatic patients [28] and that up to two thirds of ruptured plaques were previously nonobstructive [7]. Stress perfusion tests excel in assessing cardiovascular risk in the short term and are useful as a decision tool regarding coronary revascularization strategies, but screening patients for atherosclerosis instead of perfusion defects provide a longer-term assessment of risk, which is desirable both for better and earlier treatment planning and intervention as well as to minimize costs, as the interval between tests can be longer.
Additionally, because the prevalence of obstructive CAD is very low among asymptomatic individuals, ischemia-dependent test performances are generally poor in this patient population. As an example, we will discuss a 55-year-old asymptomatic woman referred for an exercise electrocardiogram test, which has sensitivity and specificity for obstructive CAD of 68% and 77%, respectively [29]. She has estimated probability of obstructive CAD of 4% [29], resulting in a positive predictive value of 12%. These results would not be much better if this patient underwent an exercise nuclear test, with sensitivity and specificity of 88% and 72%, respectively [29], rendering the positive predictive value 13%. These very low positive predictive values would require unnecessary follow-up studies, burdening the health care system and potentially harming the patient.
Zero CS in Diabetic and Other High-Risk Patient Populations
As noted above, evidence for the usefulness of routinely performing CS scans in high-risk patient populations is still lacking but, if performed, a CS of zero also places these patients previously thought to be at high risk at very low risk for cardiovascular events. One study of 10,377 asymptomatic individuals with no known previous CAD demonstrated that diabetic patients (903 in total) with zero CS had the same risk for events of nondiabetic patients after a mean follow-up of 5 years, although roughly 50% of the nondiabetic patients and 25% of the diabetic patients had zero CS [14]. In another study with a mean follow-up of 20 months, 300 diabetic patients with zero CS had the same survival rate of nondiabetic patients with zero CS (98.8% and 99.4%, respectively; P = not significant) [30]. A Third recent study found that diabetic patients with a CS of zero had no cardiovascular events during the 20 months of follow-up, similar to the nondiabetic patients [31].
Advancing age is an important risk factor for cardiovascular events and because coronary calcification prevalence increases dramatically with age (only 15% of 70-year-old males had zero CS in the MESA cohort), some resist using CS in this patient population. Nevertheless, CS may have important advantages in selected elderly people, because adverse effects related to multiple drug interactions are not uncommon and having zero CS may allow for reduction in the number of drugs prescribed. Vliegenthart et al. [32] demonstrated that after a mean follow-up of 3.3 years, elderly asymptomatic subjects (mean age, 71 years) with zero CS had 0.2% per year risk for hard cardiovascular events. Almost half the total patient population in their cohort had no coronary calcium. In another study, more than 40% of patients older than 70 years were correctly reclassified in their cardiovascular risk by a CS scan, approximately 40% of those to a lower risk than previously thought [33]. Zero CS is also associated with better future systolic and diastolic left ventricular function [34] and less incidence of clinical depression [35], also important aspects in the elderly.
CS is Not Useful to Rule Out Obstructive CAD in Symptomatic Patients
Coronary calcification is only marginally related to the degree of coronary stenosis [36••, 37, 38] and it is well known that both obstructive and nonobstructive CAD as well as vascular thrombosis can occur in the absence of calcification [3, 39, 40]. Significant coronary stenoses are frequently noncalcified, and highly calcified plaques are frequently nonobstructive. It is widely assumed that coronary calcification predicts events based on the overall atherosclerosis burden rather than the detection of vulnerable or obstructive plaques.
Taking into consideration that coronary stenosis and calcification are not closely related but both have been shown to predict cardiovascular events, how can one then solve the apparent contradiction of the excellent prognosis of zero CS patients regardless of the presence of coronary obstruction among asymptomatic people? The answer for this apparent paradox lies with disease prevalence. Obstructive CAD among asymptomatic adults is estimated to range from 1% in a 30 year-old woman to 12% in a 70-year-old man [29]. By contrast, in patients presenting with atypical chest pain, prevalence of obstructive CAD is estimated to range from 12% in a 30-year-old woman to 67% in a 70-year-old man [29]. Any prognostic value obstruction has in asymptomatic patients is diluted by its low prevalence.
This is not the case with symptomatic patients, whose prevalence of obstructive CAD may tip this balance to the other side, and even more acutely so among emergency room (ER) chest pain patients, whose symptoms may be due to unstable obstructive coronary plaques. There is wide recognition of the importance of correctly diagnosing ER patients with chest pain and the risks of missed diagnoses [41].
A substudy of the multicenter CORE64 (Coronary Evaluation Using Multi-Detector Spiral Computed Tomography Angiography Using 64 Detectors) trial with patients referred for invasive coronary angiography demonstrated that 19% of the patients with no coronary calcification had at least one ≥50% coronary obstruction as seen by invasive coronary angiography, 15% had at least one ≥70% lesion, and 13% of these patients were revascularized, attesting for the clinical significance of those lesions [36••]. Interestingly, 20% of the totally occluded coronary vessels were not calcified, showing in vivo that calcification is dispensable not only for obstruction, but also for plaque rupture and coronary thrombosis. This is not an isolated finding. Many studies have demonstrated that the absence of coronary calcification is not able to exclude obstructive CAD in symptomatic patients (Table 2) [36••, 38, 42–50].
Some controversy has arisen lately concerning the CS test’s ability to rule out obstructive CAD in symptomatic patients. Most of the confusion originates on terminology. The role of CS in the ER, if any, is for low CSs (eg, zero) to rule out obstructive CAD, to discharge patients safely home; thus, a CS of zero might be considered positive for identifying no obstructive CAD patients. Therefore, the usual understandings of sensitivity and specificity for CAD detection have to be revisited; in this special case, the high sensitivity CS has for determining obstructive CAD seen on the studies in Table 2 are not helpful because CS has no use as a rule-in test because no one would send patients to invasive angiography exclusively based on a ≥1 CS. Specificity is the test determinant that would express the ability of negative (zero) CS to identify negative (ie, no obstructive lesions) cases, and one can clearly see that specificity is low across the studies, highlighting the inability of a zero CS to rule out obstructive CAD. An exception was made for the CORE64 substudy, which took zero CS as a positive test to rule out obstructive CAD (in the spirit of ruling out CAD), thus simply inverting the sensitivity and specificity values.
Another reason CS is not a good test for identifying obstructive CAD is the low prevalence of zero CS among symptomatic patients. Table 2 again shows that most of the studies had less than 20% prevalence of zero CS. Because we are only interested in the negative results as discussed above, performing a test that only yields useful data in 20% of the cases is clearly not ideal.
Therefore, searching for surrogate evidence of stenosis, as is the case with CS, makes the performance of the test rely heavily on the prevalence of obstructive CAD and other biological factors, rendering CS unsuitable for ruling out obstructive CAD in general practice. We believe this cautious approach on CS reflects the application of what we know in pathophysiology to clinical medicine and supports the results of previous studies indicating that symptomatic patients with suspected CAD should not be discharged from the emergency department based solely on the results of coronary calcium scores assessed by unenhanced CT [51].
Furthermore, CS is only a weak surrogate marker of stenosis. To rule out obstructive CAD based on indirect evidence would depend too heavily on obstructive disease prevalence, thus making this strategy difficult to generalize. In fact, this reliance on indirect markers has been proven troublesome in the past, such as young women with atypical chest pain in the ER who were frequently discharged based solely on their low likelihood of obstructive CAD, which was proved to be a mistake and corrected in the late 1900s and early 2000s by awareness campaigns such as The Red Dress headed by the American Heart Association [52].
How Does a Zero CS Change the Asymptomatic Patient Management?
The excellent short- to mid-term prognosis of asymptomatic zero CS patients renders any primary prevention therapy very unlikely to be cost-effective [53]. Data are missing regarding whether zero CS patients should have their cardiovascular medications withheld, but this may be appropriate nonetheless.
Patients with hypertension or diabetes should be treated to target end points regardless of the CS result, for they have been proven to improve long-term outcomes in numerous primary prevention trials regardless of patient short- to mid-term risk [54, 55]. In selected zero CS patients with drug-related adverse effects, relaxing the therapeutic target may be reasonable.
Primary prevention trials with statins are not as definitive. Data suggest that low-risk patients do not benefit much from cholesterol-lowering medications, as the number of cardiovascular events prevented by these drugs is too small (if any) and the number needed to treat too high, rendering these drugs probably not indicated in zero CS primary prevention patients unless cholesterol levels are very high [56–58]. Aspirin is also not recommended in primary prevention of patients with zero CS due to its poor relationship of adverse effects to benefit, even if the patient has diabetes. The American Diabetes Association has published guidelines that recommend aspirin in primary prevention only if the patient has a 10-year risk for cardiovascular events greater than 10%, which is not the case for any subgroup of zero CS patients [59]. Of course, lifestyle healthy changes such as exercise, eating habits, and smoking cessation should be encouraged regardless of the CS.
When Should a Patient with Zero CS Be Retested?
If the patient develops symptoms suggestive of cardiovascular disease he or she should be re-evaluated regardless of a previous zero CS. Remaining asymptomatic, evidence suggests that the CS should not be repeated frequently, certainly not yearly [11, 60, 61••].
One recent study has demonstrated the cumulative rate of “conversion” from a zero CS to ≥1 CS to be 15% in the first 4 years and 25% in the fifth year [61••]. The authors conclude that 4 years is the ideal “warranty period” for a zero CS. Although reasonable, their conclusion may be overstated because there are important issues remaining before retesting shall be considered: 1) the overwhelming majority of the patients who converted to ≥1 CS still remained with less than 100 CS, and are still considered low risk; 2) converting to ≥1 CS is not the end point, cardiac events are; 3) it has not been demonstrated so far that patients who convert to ≥1 CS would benefit from any change in therapy; and 4) there is no evidence retesting is cost-effective. Although CS retesting is still a subject of debate, it should be highlighted that subclinical atherosclerosis tests such as CS decrease retesting necessity compared with functional studies, as “warranty” periods for the latter are frequently considered to be 2 years [62].
Conclusions
Having a zero CS places asymptomatic subjects at very low risk for cardiac events, even if the patient is considered to be at high risk by clinical assessment. There are few primary prevention medications that would be cost-effective in such a low-risk patient population. Zero CS determines better risk stratification and is preferred over stress perfusion techniques for risk stratification of asymptomatic patients. Whether and when zero CS patients need to be retested is still open for debate, but it should not be in less than 4 to 5 years. CS should not be used to rule out obstructive CAD in symptomatic patients, as its correlation with coronary stenosis is poor and obstructive CAD is frequently found among symptomatic zero CS patients.
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Agatston AS, Janowitz WR, Hildner FJ, et al.: Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol 1990, 15:827–832.
Rumberger JA, Simons DB, Fitzpatrick LA, et al.: Coronary artery calcium area by electron-beam computed tomography and coronary atherosclerotic plaque area. A histopathologic correlative study. Circulation 1995, 92:2157–2162.
Stary HC, Chandler AB, Dinsmore RE, et al.: A definition of advanced types of atherosclerotic lesions and a histological classification of atherosclerosis. A report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Arterioscler Thromb Vasc Biol 1995, 15:1512–1531.
Wexler L, Brundage B, Crouse J, et al.: Coronary artery calcification: pathophysiology, epidemiology, imaging methods, and clinical implications. A statement for health professionals from the American Heart Association. Writing Group. Circulation 1996, 94:1175–1192.
Ellis S, Alderman E, Cain K, et al.: Prediction of risk of anterior myocardial infarction by lesion severity and measurement method of stenoses in the left anterior descending coronary distribution: a CASS Registry Study. J Am Coll Cardiol 1988, 11:908–916.
Naghavi M, Libby P, Falk E, et al.: From vulnerable plaque to vulnerable patient: a call for new definitions and risk assessment strategies: Part II. Circulation 2003, 108:1772–1778.
Alderman EL, Corley SD, Fisher LD, et al.: Five-year angiographic follow-up of factors associated with progression of coronary artery disease in the Coronary Artery Surgery Study (CASS). CASS Participating Investigators and Staff. J Am Coll Cardiol 1993, 22:1141–1154.
Detrano R, Guerci AD, Carr JJ, et al.: Coronary calcium as a predictor of coronary events in four racial or ethnic groups. N Engl J Med 2008, 358:1336–1345.
Davies MJ, Thomas A: Thrombosis and acute coronary-artery lesions in sudden cardiac ischemic death. N Engl J Med 1984, 310:1137–1140.
Libby P: Current concepts of the pathogenesis of the acute coronary syndromes. Circulation 2001, 104:365–372.
Greenland P, Bonow RO, Brundage BH, et al.: ACCF/AHA 2007 clinical expert consensus document on coronary artery calcium scoring by computed tomography in global cardiovascular risk assessment and in evaluation of patients with chest pain: a report of the American College of Cardiology Foundation Clinical Expert Consensus Task Force (ACCF/AHA Writing Committee to Update the 2000 Expert Consensus Document on Electron Beam Computed Tomography) developed in collaboration with the Society of Atherosclerosis Imaging and Prevention and the Society of Cardiovascular Computed Tomography. J Am Coll Cardiol 2007, 49:378–402.
Greenland P, LaBree L, Azen SP, et al.: Coronary artery calcium score combined with Framingham score for risk prediction in asymptomatic individuals. JAMA 2004, 291:210–215.
Arad Y, Goodman KJ, Roth M, et al.: Coronary calcification, coronary disease risk factors, C-reactive protein, and atherosclerotic cardiovascular disease events: the St. Francis Heart Study. J Am Coll Cardiol 2005, 46:158–165.
Raggi P, Shaw LJ, Berman DS, Callister TQ: Prognostic value of coronary artery calcium screening in subjects with and without diabetes. J Am Coll Cardiol 2004, 43:1663–1669.
Budoff MJ, Shaw LJ, Liu ST, et al.: Long-term prognosis associated with coronary calcification: observations from a registry of 25,253 patients. J Am Coll Cardiol 2007, 49:1860–1870.
Falk E, Shah PK, Fuster V: Coronary plaque disruption. Circulation 1995, 92:657–671.
Libby P: Vascular biology of atherosclerosis: overview and state of the art. Am J Cardiol 2003, 91:3A–6A.
O’Rourke RA, Brundage BH, Froelicher VF, et al.: American College of Cardiology/American Heart Association Expert Consensus document on electron-beam computed tomography for the diagnosis and prognosis of coronary artery disease. Circulation 2000, 102:126–140.
Hoff JA, Chomka EV, Krainik AJ, et al.: Age and gender distributions of coronary artery calcium detected by electron beam tomography in 35,246 adults. Am J Cardiol 2001, 87:1335–1339.
Bostrom K, Watson KE, Horn S, et al.: Bone morphogenetic protein expression in human atherosclerotic lesions. J Clin Invest 1993, 91:1800–1809.
Ikeda T, Shirasawa T, Esaki Y, et al.: Osteopontin mRNA is expressed by smooth muscle-derived foam cells in human atherosclerotic lesions of the aorta. J Clin Invest 1993, 92:2814–2820.
Blankenhorn DH, Stern D: Calcification of the coronary arteries. Am J Roentgenol Radium Ther Nucl Med 1959, 81:772–777.
Raggi P, Cooil B, Callister TQ: Use of electron beam tomography data to develop models for prediction of hard coronary events. Am Heart J 2001, 141:375–382.
LaMonte MJ, FitzGerald SJ, Church TS, et al.: Coronary artery calcium score and coronary heart disease events in a large cohort of asymptomatic men and women. Am J Epidemiol 2005, 162:421–429.
Shaw LJ, Raggi P, Schisterman E, et al.: Prognostic value of cardiac risk factors and coronary artery calcium screening for all-cause mortality. Radiology 2003, 228:826–833.
•• Sarwar A, Shaw LJ, Shapiro MD, et al.: Diagnostic and prognostic value of absence of coronary artery calcification. JACC Cardiovasc Imaging 2009, 2:675–688. This meta-analysis demonstrated the excellent prognosis of more than 29,000 zero CS patients
Folsom AR, Kronmal RA, Detrano RC, et al.: Coronary artery calcification compared with carotid intima-media thickness in the prediction of cardiovascular disease incidence: the Multi-Ethnic Study of Atherosclerosis (MESA). Arch Intern Med 2008, 168:1333–1339.
Murabito JM, Evans JC, Larson MG, Levy D: Prognosis after the onset of coronary heart disease. An investigation of differences in outcome between the sexes according to initial coronary disease presentation. Circulation 1993, 88:2548–2555.
Fraker Jr TD, Fihn SD: 2007 chronic angina focused update of the ACC/AHA 2002 guidelines for the management of patients with chronic stable angina: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines Writing Group to develop the focused update of the 2002 guidelines for the management of patients with chronic stable angina. J Am Coll Cardiol 2007, 50:2264–2274.
Elkeles RS, Godsland IF, Feher MD, et al.: Coronary calcium measurement improves prediction of cardiovascular events in asymptomatic patients with type 2 diabetes: the PREDICT study. Eur Heart J 2008, 29:2244–2251.
Van Werkhoven JM, Cademartiri F, Seitun S, et al.: Diabetes: prognostic value of CT coronary angiography—comparison with a nondiabetic population. Radiology 2010, 256:83–92.
Vliegenthart R, Oudkerk M, Hofman A, et al.: Coronary calcification improves cardiovascular risk prediction in the elderly. Circulation 2005, 112:572–577.
Raggi P, Gongora MC, Gopal A, et al.: Coronary artery calcium to predict all-cause mortality in elderly men and women. J Am Coll Cardiol 2008, 52:17–23.
Colletti PM, Dustin LD, Wong ND, et al.: Does coronary calcium score predict future cardiac function? Association of subclinical atherosclerosis with left ventricular systolic and diastolic dysfunction at MR imaging in an elderly cohort. Radiology 2010, 257:64–70.
Hamer M, Kivimaki M, Lahiri A, et al.: Persistent cognitive depressive symptoms are associated with coronary artery calcification. Atherosclerosis 2010, 210:209–213.
•• Gottlieb I, Miller JM, Arbab-Zadeh A, et al.: The absence of coronary calcification does not exclude obstructive coronary artery disease or the need for revascularization in patients referred for conventional coronary angiography. J Am Coll Cardiol 2010, 55:627–634. This multicenter study has important findings, such as a high prevalence of obstructive CAD among symptomatic patients with zero CS, the weak relationship between CS and degree of coronary stenosis, and the in vivo demonstration of totally occluded arteries with ruptured plaques without evidence of calcification.
Chang SM, Nabi F, Xu J, et al.: The coronary artery calcium score and stress myocardial perfusion imaging provide independent and complementary prediction of cardiac risk. J Am Coll Cardiol 2009, 54:1872–1882.
Lau GT, Ridley LJ, Schieb MC, et al.: Coronary artery stenoses: detection with calcium scoring, CT angiography, and both methods combined. Radiology 2005, 235:415–422.
Virmani R, Kolodgie FD, Burke AP, et al.: Lessons from sudden coronary death: a comprehensive morphological classification scheme for atherosclerotic lesions. Arterioscler Thromb Vasc Biol 2000, 20:1262–1275.
Virmani R, Burke AP, Farb A, Kolodgie FD: Pathology of the vulnerable plaque. J Am Coll Cardiol 2006, 47:C13–C18.
Mehta RH, Eagle KA: Missed diagnoses of acute coronary syndromes in the emergency room—continuing challenges. N Engl J Med 2000, 342:1207–1210.
Becker A, Leber A, White C, et al.: Multislice computed tomography for determination of coronary artery disease in a symptomatic patient population. Int J Cardiovasc Imaging 2007, 23:361–367.
Haberl R, Tittus J, Bohme E, et al.: Multislice spiral computed tomographic angiography of coronary arteries in patients with suspected coronary artery disease: an effective filter before catheter angiography? Am Heart J 2005, 149:1112–1119.
Bielak LF, Rumberger JA, Sheedy PF II, et al.: Probabilistic model for prediction of angiographically defined obstructive coronary artery disease using electron beam computed tomography calcium score strata. Circulation 2000, 102:380–385.
Baumgart MD, Schmermund MD, Goerge MD, et al.: Comparison of electron beam computed tomography with intracoronary ultrasound and coronary angiography for detection of coronary atherosclerosis. J Am Coll Cardiol 1997, 30:57–64.
Budoff MJ, Georgiou D, Brody A, et al.: Ultrafast computed tomography as a diagnostic modality in the detection of coronary artery disease: a multicenter study. Circulation 1996, 93:898–904.
Fallavollita J, Brody A, Bunnell I, et al.: Fast computed tomography detection of coronary calcification in the diagnosis of coronary artery disease. Comparison with angiography in patients < 50 years old. Circulation 1994, 89:285–290.
Drosch T, Brodoefel H, Reimann A, et al.: Prevalence and clinical characteristics of symptomatic patients with obstructive coronary artery disease in the absence of coronary calcifications. Acad Radiol 2010, 17:1254–1258.
Knez A, Becker A, Leber A, et al.: Relation of coronary calcium scores by electron beam tomography to obstructive disease in 2,115 symptomatic patients. Am J Cardiol 2004, 93:1150–1152.
John K, Robert S, Shaojun W, et al.: Coronary calcium and standard risk factors in symptomatic patients referred for coronary angiography. Am Heart J 1998, 135:696–702.
Gottlieb I, Brinker J, Lima JAC, Rochitte CE: Journal of the American College of Cardiology 2010, 56(7):614-615.
Mosca L, Mochari H, Christian A, et al.: National study of women’s awareness, preventive action, and barriers to cardiovascular health. Circulation 2006, 113:525–534.
Furberg CD, Hennekens CH, Hulley SB, et al.: 27th Bethesda Conference: matching the intensity of risk factor management with the hazard for coronary disease events. Task Force 2. Clinical epidemiology: the conceptual basis for interpreting risk factors. J Am Coll Cardiol 1996, 27:976–978.
Redberg RF, Benjamin EJ, Bittner V, et al.: AHA/ACCF [corrected] 2009 performance measures for primary prevention of cardiovascular disease in adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on performance measures (writing committee to develop performance measures for primary prevention of cardiovascular disease): developed in collaboration with the American Academy of Family Physicians, American Association of Cardiovascular and Pulmonary Rehabilitation, and Preventive Cardiovascular Nurses Association. Circulation 2009, 120:1296–1336.
National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III): Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) Final Report. Circulation 2002, 106:3143–3421.
Mills EJ, Rachlis B, Wu P, et al.: Primary prevention of cardiovascular mortality and events with statin treatments: a network meta-analysis involving more than 65,000 patients. J Am Coll Cardiol 2008, 52:1769–1781.
Brugts JJ, Deckers JW: Statin prescription in men and women at cardiovascular risk: to whom and when? Curr Opin Cardiol 2010, 25:484–489.
Ray KK, Seshasai SR, Erqou S, et al.: Statins and all-cause mortality in high-risk primary prevention: a meta-analysis of 11 randomized controlled trials involving 65,229 participants. Arch Intern Med 2010, 170:1024–1031.
Executive summary: Standards of medical care in diabetes—2010 [no authors listed]. Diabetes Care 2010, 33:S4–S10.
Gopal A, Nasir K, Liu ST, et al.: Coronary calcium progression rates with a zero initial score by electron beam tomography. Int J Cardiol 2007, 117:227–231.
•• Min JK, Lin FY, Gidseg DS, et al.: Determinants of coronary calcium conversion among patients with a normal coronary calcium scan: what is the “warranty period” for remaining normal? J Am Coll Cardiol 2010, 55:1110–1117. This study demonstrated that zero CS patients develop coronary calcification very rarely before 4 years, hinting that retesting should not be done in less time than that.
Hachamovitch R, Hayes S, Friedman JD, et al.: Determinants of risk and its temporal variation in patients with normal stress myocardial perfusion scans: what is the warranty period of a normal scan? J Am Coll Cardiol 2003, 41:1329–1340.
Arad Y, Spadaro LA, Goodman K, et al.: Prediction of coronary events with electron beam computed tomography. J Am Coll Cardiol 2000, 36:1253–1260.
Wong ND, Budoff MJ, Pio J, Detrano RC: Coronary calcium and cardiovascular event risk: evaluation by age- and sex-specific quartiles. Am Heart J 2002, 143:456–459.
Kondos GT, Hoff JA, Sevrukov A, et al.: Electron-beam tomography coronary artery calcium and cardiac events: a 37-month follow-up of 5635 initially asymptomatic low- to intermediate-risk adults. Circulation 2003, 107:2571–2576.
Taylor AJ, Bindeman J, Feuerstein I, et al.: Coronary calcium independently predicts incident premature coronary heart disease over measured cardiovascular risk factors: mean three-year outcomes in the Prospective Army Coronary Calcium (PACC) project. J Am Coll Cardiol 2005, 46:807–814.
Becker A, Leber A, Becker C, Knez A: Predictive value of coronary calcifications for future cardiac events in asymptomatic individuals. Am Heart J 2008, 155:154–160.
Hachamovitch R, Berman DS, Shaw LJ, et al.: Incremental prognostic value of myocardial perfusion single photon emission computed tomography for the prediction of cardiac death: differential stratification for risk of cardiac death and myocardial infarction. Circulation 1998, 97:535–543.
Vanzetto G, Ormezzano O, Fagret D, et al.: Long-term additive prognostic value of thallium-201 myocardial perfusion imaging over clinical and exercise stress test in low to intermediate risk patients: study in 1137 patients with 6-year follow-up. Circulation 1999, 100:1521–1527.
Gibbons RJ, Hodge DO, Berman DS, et al.: Long-term outcome of patients with intermediate-risk exercise electrocardiograms who do not have myocardial perfusion defects on radionuclide imaging. Circulation 1999, 100:2140–2145.
Sandvik L, Erikssen J, Thaulow E, et al.: Physical fitness as a predictor of mortality among healthy, middle-aged Norwegian men. NEngl J Med 1993, 328:533–537.
Kampert JB, Blair SN, Barlow CE, Kohl HW: Physical activity, physical fitness, and all-cause and cancer mortality: a prospective study of men and women. Ann Epidemiol 1996, 6:452–457.
Mora S, Redberg RF, Sharrett AR, Blumenthal RS: Enhanced risk assessment in asymptomatic individuals with exercise testing and Framingham risk scores. Circulation 2005, 112:1566–1572.
Disclosure
Conflicts of interest: I. Gottlieb: none; L. Sara: none; J.A. Brinker: none; J.A.C. Lima: receives grant support from the National Institutes of Health (Bethesda, MD), the D.W. Reynolds Foundation, and Toshiba Medical Systems (Otawara, Japan); C.E. Rochitte: none.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gottlieb, I., Sara, L., Brinker, J.A. et al. CT Coronary Calcification: What Does a Score of “0” Mean?. Curr Cardiol Rep 13, 49–56 (2011). https://doi.org/10.1007/s11886-010-0151-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11886-010-0151-4