Abstract
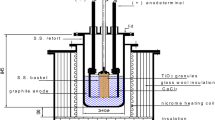
Calciothermic reduction of TiO2 provides a potentially low-cost route to titanium production. Presented in this article is a suitably designed diagram, useful for assessing the degree of reduction of TiO2 and residual oxygen contamination in metal as a function of reduction temperature and other process parameters. The oxygen chemical potential diagram à la Ellingham-Richardson-Jeffes is useful for visualization of the thermodynamics of reduction reactions at high temperatures. Although traditionally the diagram depicts oxygen potentials corresponding to the oxidation of different metals to their corresponding oxides or of lower oxides to higher oxides, oxygen potentials associated with solution phases at constant composition can be readily superimposed. The usefulness of the diagram for an insightful analysis of calciothermic reduction, either direct or through an electrochemical process, is discussed. Identified are possible process variations, modeling and optimization strategies.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
W. J. Kroll, Trans. Am. Electrochem. Soc., 78 (1940), pp. 35–47.
T. Oki and H. Inoue, Mem. Fac. Eng., Nagoya Univ., 19(1) (1967), pp. 164–166.
T. Deura et al., Proceedings of the 24th Symposium on Molten Salt Chemistry, ed. Y. Ito (Kyoto, Japan: Electrochemical Society of Japan, Kyoto, 1992), pp. 3–4.
G.Z. Chen, D.J. Fray, and T.W. Farthing, Nature, 407 (2000), pp. 361–363.
K. Ono and R.O. Suzuki, JOM, 54(2) (2002), pp. 59–61.
R.O. Suzuki, K. Teranuma, and K. Ono, Metall. Mater. Trans. B, 34 (2003), pp. 287–296.
T. Abiko, I. Park, and T.H. Okabe, Proceedings of 10th World Conference on Titanium (Weinheim, Germany: Wiley-VCH, 2003), pp. 253–260.
Y.Y. Xiao and D.J. Fray, Adv. Funct. Mater., 15 (2005), pp. 1757–1761.
K.T. Jacob and J.H.E. Jeffes, Trans. Inst. Min. Metall., Sec. C, 80 (1971), pp. C181–C189.
J.H.E. Jeffes and K.T. Jacob, Metallurgical Chemistry (London: H.M.S.O., 1972), pp. 513–525.
O. Kubaschewski, Titanium: Physico-Chemical Properties of Its Compounds and Alloys, ed. K.L. Komarek, Atomic Energy Review, Special Issue No. 9 (Vienna, Austria: International Atomic Energy Agency, 1983), pp. 22–30.
K.T. Jacob, S.M. Hoque, and Y. Waseda, Mater. Trans. JIM, 41 (2000), pp. 681–689.
M.W. Chase, Jr., NIST-JANAF Thermochemical Tables, Fourth Edition, Monograph No. 9, Journal of Physical and Chemical Reference Data (New York: American Institute of Physics, 1998), pp. 641–1807.
M. Cancarevic, M. Zinkevich, and F. Aldinger, Calphad, 31 (2007), pp. 330–342.
L.B. Pankratz, Thermodynamic Properties of Elements and Oxides, Bulletin 672 (Washington, D.C.: U.S. Department of the Interior, Bureau of Mines, 1982), pp. 93–475.
T.H. Okabe et al., Metall. Trans. B, 24B (1993), pp. 449–455.
T.H. Okabe, T. Oishi, and K. Ono, J. Alloys Compounds, 184 (1992), pp. 43–56.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jacob, K.T., Gupta, S. Calciothermic reduction of TiO2: A diagrammatic assessment of the thermodynamic limit of deoxidation. JOM 61, 56–59 (2009). https://doi.org/10.1007/s11837-009-0072-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11837-009-0072-0