Abstract
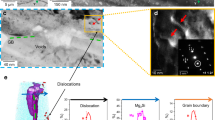
There is growing evidence for hydrogen uptake in aluminum alloys and its contribution to the crack growth of high-strength aluminum alloys, but less evidence for low-strength alloys. This paper summarizes the evidence for hydrogen uptake in a low-strength alloy, AA5083, and its contribution to the stress-corrosion cracking of this alloy. A key factor is the anodic dissolution of grain boundary β phase (Al3Mg2) and the associated hydrogen reduction that accompanies this dissolution.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
N.J.H. Holroyd, A.K. Vasudevan, and L. Christodolou, Aluminum Alloys, ed. A.K. Vasudevan and R.D. Doherty, (London: Academic Press, 1989), p. 463.
V.M. Polyanski, Sov. Mater. Sci., 21 (1985), p. 301.
H. Vogtand M.O. Speidel, “Stress Corrosion Cracking of Two Aluminum Alloys: A Comparison Between Experimental Observations and Data Based on Modeling,” Corrosion Science, 40 (1998) p. 251.
R.H. Jones et al, “Role of Magnesium in the Stress Corrosion Cracking of an Al-Mg Alloy,” Metall. and Mater. Trans A, 32A (2001). p. 1699.
M.O. Speidel and M.V. Hyatt, Advances in Corrosion Science and Technology, Vol. 2, ed. M.G. Fontana and R.W. Staehle (New York, NY: Plenum Press, 1972), p. 115.
A.F. Beck and P.R. Sperry, Fundamental Aspects of Stress Corrosion Cracking (Houston, TX: NACE, 1969), p. 513.
D.O. Sprowls and R.H. Brown, Fundamental Aspects of Stress Corrosion Cracking (Houston, TX: NACE, 1969), p. 466.
J.R. Pickens, J.R. Gordon, and J.A.S. Green, “The Effect of Loading Mode on the Stress-Corrosion Cracking of Al Alloy 5083,” Metall. and Mater. Trans. A, 14A (1983), p. 925.
Z.F. Wang et al., “The Effect of Microstructure on the Polarization and Stress Corrosion Cracking in Al-5Mg Alloys,” (Paper presented at Corrosion/2000, Orlando, FL 26–31 March 2000).
J.C. Chang and T.H. Chuang, “Stress-Corrosion Cracking Susceptibility of the Superplastically Formed 5083 Aluminum Alloy in 3.5 Pct. NaCl Solution,” Metall. and Mater. Trans. A, 30A (1999), p. 3191.
T. Enjo, T. Kuroda, and H. Shinonaga. “Effects of Relatively Insoluble Compounds and β Phase on Stress Corrosion Cracking of 5083 Aluminum Alloy,” Trans. JWRI, 8 (1) (1979), pp. 67.
M.C. Carroll et al., “Effects of Zn Additions on the Grain Boundary Precipitation and Corrosion of Al-5083,” Scripta Mater., 42 (2000), pp. 335.
P. Doig and J.W. Edington, “The Influence of Solute-Depleted Zones on the Stress-Corrosion Susceptibility of Aged Al-7.2 Mass % Mg and Al-4.4 Mass % Cu Alloys,” Proc. R. Soc. Lond. A. 339 (London: Royal Society, 1974), p. 37.
H. Yukawa et al., “Heterogeneous Distributions of Magnesium Atoms Near the Precipitate in Al-Mg Based Alloys,” Acta Metal. Mater. 43 (1995), p. 681.
L.I. Kaigorodova, “The Effect of Grain-Boundary Structure Formation on b-Precipitation in Aged Al-Mg Alloys,” Materials Science Forum 294–296, (1999), p. 477.
J.R. Scully, G.A. Young, Jr., and S.W. Smith, “Hydrogen Solubility, Diffusion and Trapping in High Purity Aluminum and Selected Al-Base Alloy,” Materials Science Forum, 331–337 (2000), p. 1583.
M.J. Danielson, “Use of the Devanathan-Stachurski Cell to Measure Hydrogen Permeation in Aluminum Alloys,” Corrosion Science, 44 (2002), p. 829.
R.J. Gest and A.R. Troiano, “Stress Corrosion and H Embrittlement in an Al Alloy,” Corrosion, 30 (8) (1974), p. 274.
R.S. Stampella, R.P.M. Procter, and V. Ashworth, “Environmentally-Induced Cracking of Magnesium,” Corrosion Science, 24 (1984), pp. 325.
E. Charitidou et al, “Characterization of Trapped Hydrogen in Exfoliation Corroded Aluminum Alloy 2024,” Scripta Materiala, (1999), p. 1327.
G.N. Haidemenopoulos et al., “Hydrogen Absorption into Aluminum Alloy 2024-T3 During Exfoliation and Alternate Immersion Testing,” Corrosion, 54 (1998), p. 73.
G.M. Scamans and A.S. Rehal, “Electron Metallography of the Aluminum-Water Vapor Reaction and its Relevance to Stress Corrosion Susceptibility,” J. of Mater. Sci., 14 (1979), p. 2459.
G.M. Scamans, “Hydrogen Bubbles in Embrittled Al-Zn-Mg Alloys,” J. of Mater. Sci., 13 (1978), pp. 27.
R. Alani and P.R. Swann, “Water Vapour Embrittlement and Hydrogen Bubble Formation in Al-Zn-Mg Alloys,” Br. Corrs. J., 12 (1977), p. 80.
J.R. Scully and G.A. Young, Jr., “The Effects of Temper, Test Temperature, and Alloyed Copper on the Hydrogen-Controlled Crack Growth Rate of an Al-Zn-Mg(Cu) Alloy,” Corrosion/2000, Paper No. 368, (Paper presented at Corrosion/2000, Orlando, FL 26–31 March 2000)
D.E. Azofeifa, N. Clark, A. Amador, and A. Saenz, “Determination of Hydrogen Absorption in Pd Coated Al Thin Films,” Thin Solid Films, 300, (1997), p. 295.
M.J. Danielson, private communication, July 2000.
G.M. Scamans, M.J.H. Holroyd, and C.D.S. Tuck, “The Role of Magnesium Segregation in the Intergranular Stress Corrosion Cracking of Aluminum Alloys,” Corrosion Science, 27 (1987), p. 329.
R.G. Song et al, “Grain Boundary Segregation and Hydrogen-Induced Fracture in 7050 Aluminum Alloy,” Acta Mater., 44 (1996), p. 3241.
C.F. Windisch, “Corrosion of Mg and Cu Implanted Al in 3.5% NaCl Solution,” Proceedings of Symposium E1: Corrosion and Corrosion Prevention of Low Density Metals and Alloy, 198th Meeting of the Electrochemical Society, Phoenix, AZ, October 22–27, 2000.
R.K. Viswanadham, T.S. Sun, and J.A.S. Green, Metall. Trans. A, 11A (1980), p. 85.
L.J. Qiao, J.L. Luo, Scripta Mater., 35 (1996), p. 1475.
R.H. Jones et al., “Interactions of Crack-Tips with Electrochemically Active Particles,” R.H. Jones, ed., Chemistry and Electrochemistry of Corrosion and Stress Corrosion Cracking: A Symposium Honoring the Contributions of R.W. Staehle, (Warrendale, PA: TMS, 2002), p. 583.
D. Tanguy et al. Corrosion Sci., 44 (2002), p. 1163.
Author information
Authors and Affiliations
Additional information
For more information, contact R.H. Jones, Pacific Northwest National Laboratory, P.O. Box 999, MSIN p8–15, Richland, WA 99352-0999; (509) 376–4276; fax (509) 376-0418; e-mail rh.jones@pnl.gov.
Rights and permissions
About this article
Cite this article
Jones, R.H. The influence of hydrogen on the stress-corrosion cracking of low-strength Al-Mg alloys. JOM 55, 42–46 (2003). https://doi.org/10.1007/s11837-003-0225-5
Issue Date:
DOI: https://doi.org/10.1007/s11837-003-0225-5