Abstract
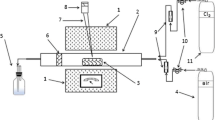
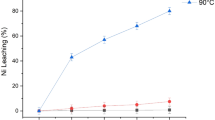
A leaching process was established based on the ability of platinum-group metals to form stable chloro-complexes in acidic chloride solutions. Industrial catalyst losses were examined for the recovery of platinum, palladium, and rhodium by leaching with a mixture of sulfuric acid and sodium chloride to avoid using aqua regia or autoclave conditions. Extraction of platinum and rhodium in 60% H2SO4 at 135°C steadily increased with increasing NaCl concentrations reaching 95% and 85%, respectively, at 0.1 M NaCl after two hours. By comparison, palladium was dissolved more quickly but also reached 85% under the same conditions. Extraction of each metal increased with temperatures up to 125°C but plateaued at higher temperatures. Similar behavior was observed with increasing H2SO4 concentrations up to 60%. More than 99% extraction of each metal was obtained after ten hours using 0.1 M NaCl and 60% H2SO4 at 125°C.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
G.B. Harris, “A Review of Precious Metals Refining,” Proceedings of the 17th International Precious Metals Institute Conference (Austin, TX: IPMI, 1993), p. 351.
T.N. Angelidis and E. Skouraki “Preliminary Studies of Platinum Dissolution from a Spent Industrial Catalyst,” Applied Catalysis A: General, 142 (1996), p. 387.
K.A. Wu, K.D. Wisecarver, and M.A. Abraham, “Rhodium, Platinum and Palladium Recovery From New and Spent Automotive Catalyst,” Proceedings of the 17th International Precious Metals Institute Conference (Austin, TX: IPMI, 1993), p. 343.
C.A. Fleming et al., “A Process for the Simultaneous Leaching and Recovery of Gold, Platinum Group Metals and Base Metals from Ores and Concentrates,” EPD Congress 2000, ed. P.R. Taylor (Warrendale, PA: TMS, 2000), p. 419.
B.W. Mountain and S.A. Wood, “Solubility and Transport of Platinum Group Elements in Hydrothermal Solutions: Thermodynamic and Physical Chemical Constraints,” Geo-Platinum 87 (London: Elsevier Applied Science, 1988), p. 57.
P.L. Sibrell and G.B. Atkinson, “Leaching of Petroleum Catalyst With Cyanide for Palladium Recovery,” Recycling of Metals and Engineered Materials, ed. P.B. Queneau and R.D. Peterson (Warrendale, PA: TMS, 1995), p. 121.
C.M. MacInnes, G.J. Sparrow, and J.T. Woodcock, “Extraction of Platinum, Palladium and Gold by Cyanidation of Coronation Hill Ore,” Hydrometallurgy, 35 (1994), p. 141.
R.J. Kuczynski, G.B. Atkinson, and W.J. Dolinar “Recovery of Platinum Group Metals from Automobile Catalyst—Pilot Plant Operation,” Recycling of Metals and Engineered Materials, ed. P.B. Queneau and R.D. Peterson (Warrendale, PA: TMS, 1995), p. 527.
K.N. Han and X. Meng “Recovery of Platinum Group Metals and Rhenium From Materials Using Halogen Reagents,” U.S. patent 5,542,957 (August 1996).
K.N. Han, “Leaching of Precious Metals From Secondary Sources Using Halogen Salts,” The XXI International Mineral Processing Congress (Amsterdam, The Netherlands: Elsevier Science, 2000), pp. A6–33.
F. Solomon, “Process for Extracting Noble Metals,” U.S. patent 4,997,532 (1989).
W.P.C. Duyvesteyn, H. Lue, and S. Duyvesteyn, “Recovery Platinum Group Metals From Oxides Ores,” U.S. patent 5,292,490 (1994).
CRC Handbook of Chemistry and Physics (London: CRC Press, 1994–1995).
R. Loewen, “Small Scale Refining of Jewelers Wastes,” (Crosby, TX: Jean Wilson Word Processing Services, 1995), p. 166.
Author information
Authors and Affiliations
Additional information
For more information contact Mohamed Hesham Hassan Mahmoud in the Extractive Metallurgy Department, Central Metallurgical R&D Institute, P.O. Box 87, Helwan, Cairo, Egypt; (202)5010642, Ext. 213; Fax (202)5010639; e-mail mheshamm@hotmail.com
Rights and permissions
About this article
Cite this article
Mahmoud, M.H.H. Leaching platinum-group metals in a sulfuric acid/chloride solution. JOM 55, 37–40 (2003). https://doi.org/10.1007/s11837-003-0086-y
Issue Date:
DOI: https://doi.org/10.1007/s11837-003-0086-y