Abstract
We investigated the influence of the Fusarium mycotoxin deoxynivalenol on the English grain aphid Sitobion avenae (Hemiptera: Aphididae) and its parasitic wasp Aphidius ervi (Hymenoptera: Braconidae) using in vitro laboratory experiments. In this tritrophic interaction, deoxynivalenol caused lethal (declined survival) and sublethal (prolonged nymphal development and reduced reproduction) effects on S. avenae aphids and consequentially led to a decreased production of parasitoid offspring resulting from parasitized deoxynivalenol-contaminated aphids. This paper highlights that the presence of mycotoxins should be considered in environmental risk assessment tests because they may alter the efficiency of biological control agents such as parasitoids through food chain contamination.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The trichothecene deoxynivalenol (DON) is a mycotoxin produced by fungal species of the genus Fusarium that causes Fusarium head blight disease on cereals (Bottalico and Perrone 2002; Goswami and Kistler 2004). DON can be translocated inside the plant through xylem vessels and phloem sieve tubes (Kang and Buchenauer 1999). Concentrations of DON found in wheat samples from fields all over Flanders (Belgium) fluctuate around 0.1–10 mg kg−1 (Audenaert et al. 2009; Isebaert et al. 2009; Landschoot et al. 2013). DON has been notorious because it provokes acute and chronic disease symptoms in humans and animals that consume contaminated grains (Bennett and Klich 2003). Its toxic effects range from diarrhea, vomiting, gastro-intestinal inflammation, necrosis of the intestinal tract, the bone marrow and the lymphoid tissues. It causes inhibition of mitochondrial function and has effects on cell division and membrane integrity and induces apoptosis (Pestka 2010). Finally, it also inhibits protein, DNA and RNA synthesis (Rocha et al. 2005).
To date, little research has been done regarding the influence of trichothecenes on insects, even though it is an interesting topic to explore because trichothecenes can pose a threat for insects feeding on contaminated plant tissue and alter insect–plant interactions. It was found that trichodermin and other 12,13-epoxytrichothecenes have larvicidal activity against the mosquito Aedes aegypti (Grove and Hosken 1975). Deoxynivalenol and especially nivalenol have toxic effects on lepidopteran Spodoptera frugiperda cells (Fornelli et al. 2004). Diacetoxyscirpenol and neosolaniol are potent antifeedants against larvae of Galleria mellonella (Mule et al. 1992) and a novel isoquinoline type pigment from F. moniliforme showed larvicidal activity against A. aegypti and Anopheles stephensi (Pradeep et al. 2015).
Also the impact of mycotoxins on the survival of insect natural enemies, developing in herbivorous insects that feed on mycotoxin-contaminated plants, is not well explored. On a higher trophic level, natural enemies such as parasitic wasps developing in contaminated herbivores, can be directly exposed to these secondary metabolites or indirectly be affected by the reduced growth of the host (Bukovinszky et al. 2012; Gols 2014). Endoparasitoids can be especially vulnerable to mycotoxins because they develop for a period of time inside the mycotoxin-contaminated host. It is already known that plant secondary metabolites can pose a threat to natural enemies (Barbosa et al. 1991; Campbell and Duffey 1981; Harvey et al. 2007; Ode et al. 2004; Roth et al. 1997), but also mycotoxins are important hazards for natural enemies that should not be neglected nor underestimated. The effect of fungus-infected plants on adult female parasitoid behavior and development has been investigated (Bultman et al. 2009; Cardoza et al. 2003; Harri et al. 2008; van Nouhuys and Laine 2008). Harri et al. (2009) investigated life-history traits of the parasitoid Aphidius ervi when it was exposed to the endophyte-tolerant aphid Metopolophium festucae feeding from Lolium perenne infected with the mycotoxin-producing endophyte Neotyphodium lolii. The presence of endophytes significantly increased the development time of A. ervi. The authors concluded that extended parasitoid development should ultimately reduce the population growth of A. ervi and thus endophyte presence may represent an advantage for endophyte-tolerant aphid species. Still, to our knowledge, research remains scarce on the impact of mycotoxins against parasitoids that develop inside herbivorous insects feeding directly from those mycotoxins.
To fill this knowledge gap, we performed a case study investigating the influence of the mycotoxin DON on the phloem-feeding cereal aphids Sitobion avenae Fab. (Hemiptera: Aphididae). Both the mycotoxin-producing Fusarium spp. and the aphid S. avenae reside on wheat ears during anthesis, the critical fungal infection period. This can increase the chances that the cereal aphids come into contact with DON when feeding from the DON-contaminated phloem. Moreover, we examined the effect of DON-contaminated S. avenae on aphid parasitism by the parasitic wasp A. ervi Hal. (Hymenoptera: Braconidae), one of the most important parasitic wasps of S. avenae aphids in European cereal fields (Al Dobai et al. 1999; Barczak et al. 2014; Tomanovic et al. 2008).
Materials and methods
Insects and deoxynivalenol
A laboratory stock culture of S. avenae aphids was maintained on wheat seedlings at constant temperature of 22 °C, 60 % relative humidity and a photoperiod of 16 h light, stimulating parthenogenesis (De Zutter et al. 2012).
Sitobion avenae aphid mummies containing A. ervi were acquired from Biobest (Westerlo, Belgium). Newly emerged parasitoids were sexed and pairs consisting of one female and one male were put individually in Petri dishes to allow mating overnight in order to obtain naive females (i.e., females that have never laid an egg before) (Joseph et al. 2011; Pan and Liu 2014). All experiments were conducted in a climate chamber at 22 ± 2 °C, 60 % relative humidity and a photoperiod of 16 h light.
The mycotoxin deoxynivalenol (DON) (purity >99 %) was kindly provided by M. Lemmens (BOKU, Vienna, Austria). A stock solution (1000 mg L−1) was prepared in sterile water and stored at −20 °C.
Experimental setup to examine the lethal effects of deoxynivalenol on S. avenae and A. ervi
Aphids were dietary exposed to DON by using an aphid feeding apparatus as described in Sadeghi et al. (2009) that contained 200 µL of artificial diet based on formulation A of Prosser and Douglas (1992) amended with DON to a final concentration of 0.25, 1, 3 and 5 mg L−1 DON or sterile water (0 mg L−1 DON). During the experiment, the diet was changed every other day and amended with the different concentrations of the persistent molecule DON. Adult S. avenae aphids were randomly selected from the stock culture and put on wheat seedlings to produce neonates (nymphal stage 1). The age of these neonates was between 0 and 24 h (day 0). Five neonates were transported from the seedlings to each aphid feeding apparatus. At day 4 the aphids were exposed to one naive A. ervi female per aphid feeding apparatus. At this time the aphids were in the second or third nymphal stage. Subsequently, aphids remained in the feeding apparatus until mummification. The number of mummies was counted daily. Each mummy was then put in a small Petri dish in order to evaluate the time until emergence of the parasitoid progeny. In total, 60, 30, 45, 45 and 30 aphids divided into groups of five individuals per aphid feeding apparatus were used as starting population in this experiment and exposed to 0, 0.25, 1, 3 and 5 mg L−1 DON, respectively. During the experiment, the percentage of aphid populations was calculated at different stages: % surviving aphids in the DON treatment at the moment of exposure to the parasitoid female and % surviving aphids at the beginning of mummification. Moreover, the percentages of aphids that turned into mummies (% mummification) and newly emerged parasitoids (% successful parasitism) were calculated.
Experimental design to examine sublethal effects of deoxynivalenol on S. avenae aphids
S. avenae neonates were exposed to DON in a final concentration of 0.25 or 1 mg L−1 DON or sterile water (0 mg L−1 DON) using aphid feeding apparatus as described above. Aphids were examined daily to examine the nymphal development and mortality. The presence of exuvia was used for the determination of a molt. To examine reproduction, the progeny of surviving females was counted daily and carefully removed using a fine brush. Progeny of females who died during nymphal development was considered zero. The intrinsic rate of increase (r m ) (Wyatt and White 1977) was calculated by following formula: r m = [0.738 ln(Md)]/D, with Md = numbers of nymphs produced by one female during its whole adult life (D). In total, ten aphids divided into ten aphid feeding apparatus (one per apparatus) were monitored daily for each concentration of DON (0, 0.25 and 1 mg L−1 DON).
Dual-choice assay to test preference of A. ervi for control and DON-contaminated aphids
In the preference test, naive female A. ervi were given the choice between aphids fed with DON and control aphids. A repeat consisted of two aphids in a small Petri dish (3.5 cm diameter), one fed with 3 mg L−1 DON and one control aphid (fed with 0 mg L−1 DON). Both aphids were synchronized to the second or third nymphal stage (Pan and Liu 2014). In the Petri dish, one naive female was introduced and allowed to choose between the DON-contaminated aphid and the control aphid. The female was monitored for a maximum of 10 min. The aphid that was first stabbed by the female and the time until this first stab (i.e., parasitoid touching the aphid with its ovipositor) occurred was recorded. Females that required more than 10 min to stab an aphid were labeled as ‘no choice’ (Daza-Bustamante et al. 2003). In total, the choice of 60 females was tested.
Statistical analyses
Data were analyzed using SPSS Statistics 22. Statistical differences of (sub)lethal effects by different DON concentrations against S. avenae and A. ervi (Fig. 1; Table 2) and the duration of A. ervi development (Table 4) were analyzed using a nonparametric Kruskal–Wallis test. In case of significant differences between the treatments, nonparametric Mann–Whitney tests were used to compare two treatments. Data in Fig. 1 were analyzed using one-sided Mann–Whitney tests corrected with the Benjamini and Hochberg False Discovery Rate (B&H FDR) at a level of 0.05 for controlling the type I error rate (Benjamini and Hochberg 2000). All tests were conducted with a significance level of α = 0.05.
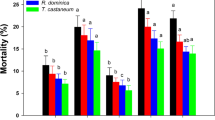
The percentage of aphid populations (mean ± SE) at different stages: % surviving deoxynivalenol (DON) at the moment of exposure to the parasitoid female, % surviving at the beginning of mummification, % aphids becoming mummies and % aphids that produced newly formed parasitoid progeny. Different letters represent significant differences (P < 0.05) between treatments with different concentrations of DON using one-sided Mann–Whitney tests corrected with B&H FDR. Starting populations (100 %) consisted of 60, 30, 45, 45 and 30 aphids divided into groups of five individuals per aphid feeding apparatus exposed to 0, 0.25, 1, 3 and 5 mg L−1 DON, respectively
Results
(Sub)lethal effects of deoxynivalenol on S. avenae and A. ervi
In this experiment S. avenae aphids were dietary exposed to five different concentrations of DON (0, 0.25, 1, 3 and 5 mg L−1 DON). The lethal effects of DON were measured by calculating the percentage of surviving S. avenae, and for A. ervi by calculating the percentage of aphids that turned into mummies and resulted into newly emerged parasitoid progeny. When the aphids that were feeding on the different concentrations of DON were exposed to the female parasitoid, a negative effect of 5 mg L−1 DON on the survival of S. avenae was found (Fig. 1; Table 1). The surviving aphid population feeding on 5 mg L−1 DON was significantly lower than the other treatments. Treatment of the aphids with 3 mg L−1 DON also caused a decrease in the aphid population but at a later time (when mummification started).
Sublethal effects of DON were measured by investigating the aphids nymphal development and reproduction while feeding on 0, 0.25 and 1 mg L−1 DON (Tables 2, 3). The duration of the last three nymphal stages (days) of aphids dietary exposed to 1 mg L−1 DON was significantly longer (P < 0.05) compared to 0 or 0.25 mg L−1 DON. When aphids were fed with 0, 0.25 and 1 mg L−1 DON, Md (i.e., the number of nymphs produced per female during its whole adult life), D (i.e., the whole adult life of the aphid in days) and r m (i.e., the aphid population intrinsic rate of increase) were calculated. Md, D and r m showed no significant differences between 0 and 0.25 mg L−1, but both treatments were significantly higher (P < 0.05) than 1 mg L−1 DON, except for r m where the effect by 0.25 and 1 mg L−1 DON was not significantly different (P = 0.057).
Table 4 presents the duration of A. ervi development in days inside S. avenae aphids contaminated with different concentrations of DON (0, 0.25, 1 and 3 mg L−1 DON). No significant differences were found between treatments 0, 0.25, 1 and 3 mg L−1 DON (P = 0.349 and 0.340 for oviposition until mummification and from mummification until emergence, respectively, according to Kruskal–Wallis tests). No mummies were found for the 5 mg L−1 DON treatment. Aphids feeding on 3 mg L−1 DON produced significantly fewer mummies compared to aphids feeding on 0.25 mg L−1 DON (Fig. 1; Table 1). Significantly less parasitoid progeny emerged from aphids fed on 3 mg L−1 DON compared to the lower concentrations tested.
Preference of parasitic wasp to attack control and DON-contaminated S. avenae aphids
In our binary-choice experiment, female A. ervi did not discriminate between DON-contaminated and control aphids. Out of a total of 60 wasps, 21 stabbed the control aphid first, 21 stabbed the DON-contaminated aphid first, and 18 were recorded as ‘no choice’. The mean time (±SE) needed by females to make a choice was not significantly different between both treatments (2.9 ± 0.5 min for control aphids and 3.3 ± 0.6 min for DON-contaminated aphids).
Discussion
Survival, nymphal development and reproduction of S. avenae aphids were negatively influenced by DON. In our experiment, female A. ervi did not discriminate between control and DON-contaminated aphids, but successful parasitism of the aphids by A. ervi (i.e., emergence of parasitoid progeny) decreased. This could mean that the parasitoid eggs or larvae were directly or indirectly susceptible to DON or DON derivatives inside the aphid body. Directly, DON causes inhibition of mitochondrial function, has effects on cell division and membrane integrity, induces apoptosis and inhibits protein, DNA and RNA synthesis (Pestka 2010; Rocha et al. 2005). Possibly these toxic effects can lead to mortality of the parasitic wasp larvae inside the aphid body. Indirectly, it is possible that DON-contaminated aphids were feeding less efficiently than control ones (indeed, their nymphal development is slower), leaving the parasitoid larvae with fewer nutritional resources for its development. Nevertheless, there was a negative effect on the parasitoid developing inside DON-contaminated aphids. This could suggest that DON causes a decreased efficiency of biological control agents such as parasitoids through food chain contamination.
In our experiment we used concentrations of DON ranging from 0 to 5 mg L−1. DON is a water soluble compound and is translocated in ears of cereal crops through phloem sieve tubes (Kang and Buchenauer 1999). Concentrations found in wheat samples from fields all over Flanders (Belgium) fluctuate around 0.1–10 mg kg−1 DON (Audenaert et al. 2009; Isebaert et al. 2009; Landschoot et al. 2013). These concentrations are present in grains at harvest and thus do not necessarily represent concentrations in the phloem during the time period that aphids are feeding from the phloem sieve tubes. Exact concentrations of DON and its derivatives in the phloem and dosages inside aphids that impair both the aphids and their parasitoids are unknown. Maybe DON can accumulate inside the aphids, but it could also be converted into other less toxic compounds such as DON-3-glucoside. These molecular mechanisms provide an interesting future research topic. To cope with xenobiotics, S. avenae has several enzyme families which show increased activities upon contamination. Cytochrome P450 monooxygenases (P450, phase I metabolic enzymes) and glutathione-S-transferases (GST, phase II metabolic enzymes) activities increased with the secondary plant compound hydroxamic acid levels in wheat (Castaneda et al. 2010) and GST activities also increased when S. avenae was dietary exposed to phenol catechol, alkaloid gramine and non-protein amino acid l-ornithine-HCI (Cai et al. 2009; Zhang et al. 2013). Lu and Gao (2009) suggested the involvement of P450s and GSTs in the susceptibility to the insecticide pirimicarb. More specifically for the role of these mechanisms in detoxifying mycotoxins, studies with aphids remain scarce. In contrast to the aphids, hymenopteran insects such as honeybees, bumblebees and the parasitoid Nasonia vitripennis, for which the whole genomes are sequenced, are known to have substantially fewer genes coding for detoxification enzymes (Claudianos et al. 2006; Oakeshott et al. 2010; Sadd et al. 2015; Werren et al. 2010) compared to other insects, leaving them more vulnerable for xenobiotics. Exposure of Hymenoptera parasitoids, such as A. ervi, to mycotoxins through food chain contamination is thus another important parameter when assessing the risk against natural enemies.
In conclusion, this study emphasized the importance of mycotoxin (DON) in food chain contamination from the plant to insects (insect–plant interactions; grain aphids S. avenae) and their natural enemies (higher trophic interactions; parasitic wasp A. ervi). Based on our data, we believe that DON can cause a decreased efficiency of biological control agents such as parasitoids through food chain contamination. Therefore, it is important also to consider the presence of mycotoxins in environmental risk assessments when insect pest control systems are based on biological control with natural enemies, such as the use of parasitic wasps against aphids.
References
Al Dobai S, Praslicka J, Mistina T (1999) Parasitoids and hyperparasitoids of cereal aphids (Homoptera, Aphididae) on winter wheat in Slovakia. Biologia 54:573–580
Audenaert K, Van Broeck R, Bekaert B, De Witte F, Heremans B, Messens K, Hofte M, Haesaert G (2009) Fusarium head blight (FHB) in Flanders: population diversity, inter-species associations and DON contamination in commercial winter wheat varieties. Eur J Plant Pathol 125:445–458
Barbosa P, Gross P, Kemper J (1991) Influence of plant allelochemicals on the tobacco hornworm and its parasitoid, Cotesia congregata. Ecology 72:1567–1575
Barczak T, Debek-Jankowska A, Bennewicz J (2014) Primary parasitoid and hyperparasitoid guilds (Hymenoptera) of grain aphid (Sitobion avenae F.) in northern Poland. Arch Biol Sci 66:1141–1148
Benjamini Y, Hochberg Y (2000) On the adaptive control of the false discovery fate in multiple testing with independent statistics. J Educ Behav Stat 25:60–83
Bennett JW, Klich M (2003) Mycotoxins. Clin Microbiol Rev 16:497–516
Bottalico A, Perrone G (2002) Toxigenic Fusarium species and mycotoxins associated with head blight in small-grain cereals in Europe. Eur J Plant Pathol 108:611–624
Bukovinszky T, Gols R, Smid HM, Kiss GB, Dicke M, Harvey JA (2012) Consequences of constitutive and induced variation in the host’s food plant quality for parasitoid larval development. J Insect Physiol 58:367–375
Bultman TL, Rodstrom JL, Radabaugh KR, VanDop JD, Librizzi JM, Longwell LL, Pulas C, Grant L, Sullivan TJ (2009) Influence of genetic variation in the fungal endophyte of a grass on an herbivore and its parasitoid. Entomol Exp Appl 130:173–180
Cai QN, Han Y, Cao YZ, Hu Y, Zhao X, Bi JL (2009) Detoxification of gramine by the cereal aphid Sitobion avenae. J Chem Ecol 35:320–325
Campbell BC, Duffey SS (1981) Alleviation of alpha-tomatine-induced toxicity to the parasitoid, Hyposoter exiguae, by phytosterols in the diet of the host, Heliothis zea. J Chem Ecol 7:927–946
Cardoza YJ, Teal PEA, Tumlinson JH (2003) Effect of peanut plant fungal infection on oviposition preference by Spodoptera exigua and on host-searching behavior by Cotesia marginiventris. Environ Entomol 32:970–976
Castaneda LE, Figueroa CC, Nespolo RF (2010) Do insect pests perform better on highly defended plants? Costs and benefits of induced detoxification defences in the aphid Sitobion avenae. J Evol Biol 23:2474–2483
Claudianos C, Ranson H, Johnson RM, Biswas S, Schuler MA, Berenbaum MR, Feyereisen R, Oakeshott JG (2006) A deficit of detoxification enzymes: pesticide sensitivity and environmental response in the honeybee. Insect Mol Biol 15:615–636
Daza-Bustamante P, Fuentes-Contreras E, Niemeyer HM (2003) Acceptance and suitability of Acyrthosiphon pisum and Sitobion avenae as hosts of the aphid parasitoid Aphidius ervi (Hymenoptera: Braconidae). Eur J Entomol 100:49–53
De Zutter N, Audenaert K, Haesaert G, Smagghe G (2012) Preference of cereal aphids for different varieties of winter wheat. Arthropod Plant Interact 6:345–350
Fornelli F, Minervini F, Mule G (2004) Cytotoxicity induced by nivalenol, deoxynivalenol, and fumonisin B, in the SF-9 insect cell line. In Vitro Cell Dev Biol Anim 40:166–171
Gols R (2014) Direct and indirect chemical defences against insects in a multitrophic framework. Plant, Cell Environ 37:1741–1752
Goswami RS, Kistler HC (2004) Heading for disaster: Fusarium graminearum on cereal crops. Mol Plant Pathol 5:515–525
Grove JF, Hosken M (1975) Larvicidal activity of some 12,13-epoxytrichothec-9-enes. Biochem Pharmacol 24:959–962
Harri SA, Krauss J, Muller CB (2008) Fungal endosymbionts of plants reduce lifespan of an aphid secondary parasitoid and influence host selection. Proc R Soc B Biol Sci 275:2627–2632
Harri SA, Krauss J, Muller CB (2009) Extended larval development time for aphid parasitoids in the presence of plant endosymbionts. Ecol Entomol 34:20–25
Harvey JA, Van Dam NM, Witjes LMA, Solerro R, Gols R (2007) Effects of dietary nicotine on the development of an insect herbivore, its parasitoid and secondary hyperparasitoid over four trophic levels. Ecol Entomol 32:15–23
Isebaert S, De Saeger S, Devreese R, Verhoeven R, Maene P, Heremans B, Haesaert G (2009) Mycotoxin-producing Fusarium species occurring in winter wheat in Belgium (Flanders) during 2002–2005. J Phytopathol 157:108–116
Joseph JR, Ameline A, Couty A (2011) Effects on the aphid parasitoid Aphidius ervi of an insecticide (Plenum (R), pymetrozine) specific to plant-sucking insects. Phytoparasitica 39:35–41
Kang Z, Buchenauer H (1999) Immunocytochemical localization of Fusarium toxins in infected wheat spikes by Fusarium culmorum. Physiol Mol Plant Pathol 55:275–288
Landschoot S, Audenaert K, Waegeman W, De Baets B, Haesaert G (2013) Influence of maize-wheat rotation systems on Fusarium head blight infection and deoxynivalenol content in wheat under low versus high disease pressure. Crop Prot 52:14–21
Lu Y, Gao X (2009) Multiple mechanisms responsible for differential susceptibilities of Sitobion avenae (Fabricius) and Rhopalosiphum padi (Linnaeus) to pirimicarb. Bull Entomol Res 99:611–617
Mule G, Dambrosio A, Logrieco A, Bottalico A (1992) Toxicity of mycotoxins of Fusarium sambucinum for feeding in Galleria mellonella. Entomol Exp Appl 62:17–22
Oakeshott JG, Johnson RM, Berenbaum MR, Ranson H, Cristino AS, Claudianos C (2010) Metabolic enzymes associated with xenobiotic and chemosensory responses in Nasonia vitripennis. Insect Mol Biol 19:147–163
Ode PJ, Berenbaum MR, Zangerl AR, Hardy ICW (2004) Host plant, host plant chemistry and the polyembryonic parasitoid Copidosoma sosares: indirect effects in a tritrophic interaction. Oikos 104:388–400
Pan MZ, Liu TX (2014) Suitability of three aphid species for Aphidius gifuensis (Hymenoptera: Braconidae): parasitoid performance varies with hosts of origin. Biol Control 69:90–96
Pestka JJ (2010) Toxicological mechanisms and potential health effects of deoxynivalenol and nivalenol. World Mycotoxin J 3:323–347
Pradeep FS, Palaniswamy M, Ravi S, Thangamani A, Pradeep BV (2015) Larvicidal activity of a novel isoquinoline type pigment from Fusarium moniliforme KUMBF1201 against Aedes aegypti and Anopheles stephensi. Process Biochem 50:1479–1486
Prosser WA, Douglas AE (1992) A test of the hypotheses that nitrogen is upgraded and recycled in an aphid (Acyrthosiphon pisum) symbiosis. J Insect Physiol 38:93–99
Rocha O, Ansari K, Doohan FM (2005) Effects of trichothecene mycotoxins on eukaryotic cells: a review. Food Addit Contam 22:369–378
Roth S, Knorr C, Lindroth RL (1997) Dietary phenolics affects performance of the gypsy moth (Lepidoptera: Lymantriidae) and its parasitoid Cotesia melanoscela (Hymenoptera: Braconidae). Environ Entomol 26:668–671
Sadd BM et al (2015) The genomes of two key bumblebee species with primitive eusocial organization. Genome Biol 16:76–107
Sadeghi A, Van Damme EJM, Smagghe G (2009) Evaluation of the susceptibility of the pea aphid, Acyrthosiphon pisum, to a selection of novel biorational insecticides using an artificial diet. J Insect Sci 9:1–8
Tomanovic Z, Kavallieratos NG, Stary P, Petrovic-Obradovic O, Athanassiou CG, Stanisavljevic LZ (2008) Cereal aphids (Hemiptera: Aphidoidea) in Serbia: seasonal dynamics and natural enemies. Eur J Entomol 105:495–501
van Nouhuys S, Laine AL (2008) Population dynamics and sex ratio of a parasitoid altered by fungal-infected diet of host butterfly. Proc R Soc B Biol Sci 275:787–795
Werren JH et al (2010) Functional and evolutionary insights from the genomes of three parasitoid nasonia species. Science 327:343–348
Wyatt IJ, White PF (1977) Simple estimation of intrinsic increase rates for aphids and tetranychid mites. J Appl Ecol 14:757–766
Zhang MX, Fang TT, Pu GL, Sun XQ, Zhou XG, Cai QN (2013) Xenobiotic metabolism of plant secondary compounds in the English grain aphid, Sitobion avenae (F.) (Hemiptera: Aphididae). Pest Biochem Physiol 107:44–49
Acknowledgments
Nathalie De Zutter is holder of a Ph.D. grant by the Institute for the Promotion of Innovation by Science and Technology in Flanders (IWT mandate No 111432).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Robert Glinwood.
Geert Haesaert and Guy Smagghe are equal last author.
Rights and permissions
About this article
Cite this article
De Zutter, N., Audenaert, K., Ameye, M. et al. Effect of the mycotoxin deoxynivalenol on grain aphid Sitobion avenae and its parasitic wasp Aphidius ervi through food chain contamination. Arthropod-Plant Interactions 10, 323–329 (2016). https://doi.org/10.1007/s11829-016-9432-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-016-9432-1