Abstract
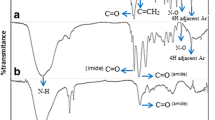
New porous chelating resin beads CPN-IDA were prepared by the reaction of poly (acrylonitrile-co-N,N'- methylenebisacrylamide) with etheylenediamine and sodium chloroacetate, respectively. The composition and morphology of the chelating resin was characterized by Fourier transform infrared spectroscopy (FTIR), thermal analysis (TGA and DrTGA), scanning electron microscopy (SEM),surface area measurements by N2adsorption (S BET ) and water regain (w%). Batch adsorption experiments were employed to study the impact of some factors that control the adsorption of metal ions including solution pH, concentration of metal ions, contact time and the temperature of the solution. Batch sorption results showed that the chelating resin had high affinity towards Cu(II), Cd(II) and Pb(II). The saturated adsorption capacity at 25 oC was 2.43, 1.93 and 1.45mmolg−1 resin for Cu(II), Cd(II) and Pb(II), respectively. Among the empirical isotherm models, the equilibrium adsorption results were obviously fitted with Langmuir model. The kinetics was analyzed using pseudo-first-order, pseudo-second-order, and intra-particle diffusion equations. The adsorption kinetic data were well elucidated with pseudo-second-order kinetic model. Thermodynamic parameters were calculated for the uptake of the metal ions under study and it was found to be a spontaneous process. Moreover, the adsorption of metal ions has been studied using column technique. The regeneration of CPN-IDA was experimentally performed by using nitric acid. The chelating resin was used repeatedly for five times with a little decrease in sorption of metal ions.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
M. A. Barakat, Arab. J. Chem., 4, 361 (2011).
R. S. Dobson and J. E. Burgess, Miner. Eng., 20, 519 (2007).
F. Fu and Q. Wang, J. Environ. Manage., 92, 407 (2011).
World Health Organisation, Guidelines for Drinking Water Quality, Recommendations, WHO, Geneva (1984).
F. Zhao, W. Z. Tang, D. Zhao, Y. Meng, D. Yin and M. Sillanpää, J. Water Process Eng., 4, 47 (2014).
J. Gao, F. Liu, P. Ling, J. Lei, L. Li, C. Li and A. Li, Chem. Eng. J., 222, 240 (2013).
A. A. Atia, A. M. Donia and K. Z. Elwakeel, Sep. Purif. Technol., 43, 43 (2005).
X. Liu, H. Chen, C. Wang, R. Qu, C. Ji, C. Sun and Y. Zhang, J. Hazard. Mater., 175, 1014 (2010).
J. Chen, R. Qu, Y. Zhang, C. Sun, C. Wang, C. Ji, P. Yin, H. Chen and Y. Niu, Chem. Eng. J., 209, 235 (2012).
A. F. Shaaban, D. A. Fadel, A. A. Mahmoud, M. A. Elkomy and S. M. Elbahy, J. Environ. Chem. Eng., 2, 632 (2014).
A. F. Shaaban, D. A. Fadel, A. A. Mahmoud, M. A. Elkomy and S. M. Elbahy, J. Environ. Chem. Eng., 1, 208 (2013).
A. F. Shaaban, D. A. Fadel, A. A. Mahmoud, M. A. Elkomy and S. M. Elbahy, Desalin. Water Treat., 51, 5526 (2013).
M. Cegłowski and G. Schroeder, Chem. Eng. J., 263, 402 (2015).
C. Y. Chen, C. L. Chiang and C. R. Chen, Sep. Purif. Technol., 54, 396 (2007).
M. V. Dinu and E. S. Dragan, React. Funct. Polym., 68, 1346 (2008).
A. A. Atia, A. M. Donia and A. M. Yousif, Sep. Purif. Technol., 61, 348 (2008).
F. Liu, L. Li P. Ling, X. Jing, C. Li, A. Li and X. You, Chem. Eng. J., 173, 106 (2011).
P. Ling, F. Liu, L. Li, X. Jing, B. Yina, K. Chena and A. Li, Talanta, 81, 424 (2010).
M. V. Dinu, E. S. Dragan and A. W. Trochimczuk, Desalination, 249, 374 (2009).
K. Z. Elwakeel and M. Rekaby, J. Hazard. Mater., 188, 10 (2011).
A. W. Trochimczuk and B. N. Kolarz, Eur. Polym. J., 36, 2359 (2000).
E. S. Dragan, E. Avram and M. V. Dinu, Polym. Adv. Technol., 17, 571 (2006).
A. Gurses, M. Yalcin, M. Sozbilir and C. Dogar, Fuel Process. Technol., 81, 57 (2003).
M. E. Malla, M. B. Alvarez and D. A. Batistoni, Talanta, 57, 277 (2002).
J. E. Huheey, Inorganic chemistry principles of structure and reactivity, 2nd Ed. New York, Harper International Edition, Harper & Row (1978).
I. Langmuir, J. Am. Chem. Soc., 40, 1361 (1918).
T. W. Weber and R. K. Chakravot, AIChE J., 20, 228 (1974).
A. Sari, M. Tuzen, D. Citak and M. Soylak, J. Hazard. Mater., 149, 283 (2007).
X. S. Wang, J. Huang, H. Q. Hua, J. Wang and Y. Qin, J. Hazard. Mater., 142, 468 (2007).
H. Freundlich, Phys. Chem. Soc., 40, 1361 (1906).
M. J. Temkin and V. Phyzev, Acta Physiochim, USSR, 12, 217 (1940).
H. Kasgoz, S. Ozgumus and M. Orbay, Polymer, 44, 1785 (2003).
S. Lagergren and B. K. Svenska, Veternskapsakad Handlingar, 24, 1 (1898).
Y. S. Ho, Water Res., 40, 119 (2006).
W. J. Weber and J. C. Morris, J. Sanitary Eng. Div., 90, 79 (1964).
B. Guo, L. Hong and H. X. Jiang, Ind. Eng. Chem. Res., 42, 5559 (2003).
A. Nilchi, R. Saberi, M. Moradi, H. Azizpour and R. Zarghami, Chem. Eng. J., 172, 572 (2011).
S. M. El-Bahy and Z. M. El-Bahy, J. Environ. Chem. Eng., 4, 276 (2016).
Y. Liu, Sep. Purif. Technol., 61, 229 (2008).
F. Gode, Removal of chromium ions from aqueous solutions by the adsorption method, in: A. A. Lewinsky (Ed.), Hazardous Materials and Wastewater: Treatment, Removal and Analysis, Nova Science Publishers, Inc., New York, 275 (2007).
C. H. Xiong, Y. Li, G. Wang, L. Fang, S. Zhou, C. Yao, Q. Chen, X. Zheng, D. Qi, Y. Fu and Y. Zhu, Chem. Eng. J., 259, 257 (2015).
S. S. Guptaa and K. G. Bhattacharyya, J. Environ. Manage., 87, 46 (2008).
A. M. Donia, A. A. Atia, H. El-Boraey and D. H. Mabrouk, Sep. Purif. Technol., 49, 64 (2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El-Bahy, S.M., El-Bahy, Z.M. Synthesis and characterization of a new iminodiacetate chelating resin for removal of toxic heavy metal ions from aqueous solution by batch and fixed bed column methods. Korean J. Chem. Eng. 33, 2492–2501 (2016). https://doi.org/10.1007/s11814-016-0070-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-016-0070-1