Abstract
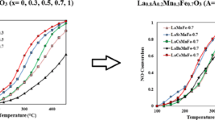
Perovskite formulations of LaB0.5Co0.5O3 (B=Cr, Mn and Cu) were examined and evaluated as catalyst for reduction of NOx by CO. Among the different and various formulations, LaMn0.5Co0.5O3 showed the highest activityselectivity with 89.84% conversion of NO and 9.62% yield of N2O at 400 °C. Therefore, more precise evaluations were implemented on LaMn x Co1-x O3 to find the optimum formulation. The LaMn0.25Co0.75O3 included the maximum performance (68.79% conversion of NO at 350 °C and 92.85% at 400 °C) and N2O yield (9.01); therefore, it was selected as optimum catalyst. Characterization methods were utilized to correlate activity and physical-chemical properties. The presented activity and reducibility of catalysts were improved due to partial substitution of Co3+ by B cation. Finally, no direct relationship was found between surface area and catalyst activity. The optimum catalyst showed complete activity-selectivity higher than 450 °C.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
F. Ahmed, H. J. Cho, J. K. Kim, N. U. Seong and Y. K. Yeo, Korean J. Chem. Eng., 32, 1 (2015).
A. Fritz and V. Pitchon, Appl. Catal. B: Environ., 13, 1 (1997).
M. D. Amiridis, T. Zhang and R. J. Farrauto, Appl. Catal. B: Environ., 10, 203 (1996).
R. Burch, J. Breen and F. Meunier, Appl. Catal. B: Environ., 39, 283 (2002).
Y. Jia, D. Du, X. Zhang, X. Ding and O. Zhong, Korean J. Chem. Eng., 30, 1735 (2013).
H. Dong, Y. Zhang and Z. Gu, Korean J. Chem. Eng., 31, 1002 (2014).
D. C. Chambers, D. E. Angove and N. W. Cant, J. Catal., 204, 11 (2001).
P. Granger, P. Malfoy, P. Esteves, L. Leclercq and G. Leclercq, J. Catal., 187, 321 (1999).
P. Granger, L. Delannoy, J. Lecomte, C. Dathy, H. Praliaud, L. Leclercq and G. Leclercq, J. Catal., 207, 202 (2002).
P. Bera, K. Patil, V. Jayaram, M. Hegde and G. Subbanna, J. Mater. Chem., 9, 1801 (1999).
N. A. Merino, B. P. Barbero, P. Grange and L. E. Cadús, J. Catal., 231, 232 (2005).
P. Femina and P. Sanjay, Research Journal of Recent Sciences, ISSN, 2277, 2502 (2012).
M. Alifanti, N. Blangenois, M. Florea and B. Delmon, Appl. Catal. A: Gen., 280, 255 (2005).
T. Screen, Platinum Metals Review, 51, 87 (2007).
Y. Nishihata, Nature, 418, 138 (2002).
P. Gallagher, D. Johnson and E. Vogel, J. American Ceramic Soc., 60, 28 (1977).
B. Levasseur and S. Kaliaguine, Appl. Catal. B: Environ., 88, 305 (2009).
S. A. Hosseini, M. T. Sadeghi, A. Alemi, A. Niaei, D. Salari and L. Kafi-Ahmadi, Chinese J. Catal., 31, 747 (2010).
J. Kim, T. Kim, J. W. Yoo, K. B. Lee and S.-I. Hong, Korean J. Chem. Eng., 29, 1329 (2012).
A. Giannakas, A. Ladavos and P. Pomonis, Appl. Catal. B: Environ., 49, 147 (2004).
A. Giannakas, A. Leontiou, A. Ladavos and P. Pomonis, Appl. Catal. A: Gen., 309, 254 (2006).
V. Belessi, C. Costa, T. Bakas, T. Anastasiadou, P. Pomonis and A. Efstathiou, Catal. Today, 59, 347 (2000).
T. Esaka, H. Sakaguchi and S. Kobayashi, Solid State Ionics, 166, 351 (2004).
M. Iwamoto, H. Yahiro, T. Kutsuno, S. Bunyu and S. Kagawa, Bulletin of the Chem. Soc. Japan, 62, 583 (1989).
R. Zhang, A. Villanueva, H. Alamdari and S. Kaliaguine, Appl. Catal. B: Environ., 64, 220 (2006).
R. Zhang, A. Villanueva, H. Alamdari and S. Kaliaguine, Appl. Catal. A: Gen., 307, 85 (2006).
H. He, M. Liu, H. Dai, W. Qiu and X. Zi, Catal. Today, 126, 290 (2007).
P. Ciambelli, S. Cimino, S. De Rossi, L. Lisi, G. Minelli, P. Porta and G. Russo, Appl. Catal. B: Environ., 29, 239 (2001).
R. Zhang, A. Villanueva, H. Alamdari and S. Kaliaguine, J. Mol. Catal. A: Chem., 258, 22 (2006).
S. A. Hosseini, D. Salari, A. Niaei and S. A. Oskoui, J. Ind. Eng. Chem., 19, 1903 (2013).
J. Zhou, L. Zhao, Q. Huang, R. Zhou and X. Li, Catal. Lett., 127, 277 (2009).
S. Maity, S. Ray and D. Bhattacharya, J. Phys. Chem. Solids, 74, 315 (2013).
X. Zhu, Y. Shi, A. Li and Q. Zhong, Ionics, 20, 1011 (2014).
J. Dacquin, C. Lancelot, C. Dujardin, P. Da Costa, G. Djega-Mariadassou, P. Beaunier, S. Kaliaguine, S. Vaudreuil, S. Royer and P. Granger, Appl. Catal. B: Environ., 91, 596 (2009).
F. Deganello, G. Marcì and G. Deganello, J. European Ceramic Soc., 29, 439 (2009).
P. Granger, C. Dujardin, J. F. Paul and G. Leclercq, J. Mol. Catal. A: Chem., 228, 241 (2005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Izadkhah, B., Niaei, A., Salari, D. et al. Catalytic removal of CO and NOx using sol-gel synthesized LaB0.5Co0.5O3 (B=Cr, Mn and Cu) and LaMn x Co1-x O3 nano-perovskites. Korean J. Chem. Eng. 33, 1192–1199 (2016). https://doi.org/10.1007/s11814-015-0254-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-015-0254-0