Abstract
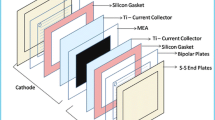
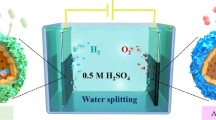
This work presents the electrocatalytic evaluation of Ni/TiO2 hollow sphere materials in PEM water electrolysis cell. All the electrocatalysts have shown remarkably enhanced electrocatalytic properties in comparison with their performance in aqueous electrolysis cell. According to cyclic voltammetric results, 0.36 A cm−2 peak current density has been exhibited in hydrogen evolution reaction (HER) from 30 wt% Ni/TiO2 electrocatalyst. 15 wt% Ni-doped titania sample has shown the best result in oxygen evolution reaction (OER) with the anodic peak current density of 0.3 A cm−2. In the anodic polarization curves, the performance of 15 wt% Ni/TiO2 hollow sphere electrocatalyst was evaluated up to 140 mA cm−2 at comparatively lower over-potential value. 20 wt% Ni/TiO2 hollow sphere electrocatalyst has also shown electrochemical stability in PEM water electrolyzer for 48 h long analysis. The comparative electrocatalytic behavior of hollow spherical materials with non-sphericals is also presented, which clearly shows the influence of hollow spherical structure in greater electrocatalytic activity of the materials. The physical characterization of all the hollow spherical materials is presented in this work, which has confirmed their better electrochemical behavior in PEM water electrolyzer.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Peavey, Fuel from water energy independence with hydrogen, Merit Products, USA (2003).
J. L. Corona-Guinto, L. Carderño-García, D.C. Martínez-Casillas, J. M. Sandoval-Pineda, P. Tamayo-Meza, R. Silva-casarin and R.G. González-Huerta, Int. J. Hydrog. Energy, DOI:10.1016/j.ijhydene. 2012.12.071 (2012).
Y.G. Yoon, G.G. Park, T.H. Yang, J. N. Han, W.Y. Lee and C. S. Kim, Int. J. Hydrog. Energy, 28, 657 (2003).
I. Radev, E. Slavcheva and E. Budevski, Int. J. Hydrogen Energy, 32, 872 (2007).
H. Beer, Improvements in or relating to electrodes for electrolytes, British Patent, 1,147,442 (1969).
S. Siracusano, V. Baglio, A. Di Blasi, N. Briguglio, A. Stassi and R. Ornelas, Int. J. Hydrog. Energy, 35, 5558 (2010).
Yang Zhang, Lixia Yue, Ke Teng, Shiyong Yuan and M. Hongchao, J. New Mat. Electrochem. Syst., 15, 271 (2012).
S. Trasatti, Electrochim. Acta, 29, 1503 (1984).
Y.Z. Zhu, H. B. Chen, Y. P. Wang, Z.H. Li, Y.L. Cao and Y. B. Chi, Chem. Lett., 35, 756 (2006).
M. Fujiwara, M.K. Shiokawa, K. Hayashi, K. Morigaki and Y. Nakahara, J. Biomed. Mater. Res. A, 8, 103 (2007).
G. R. Bamwenda, T. Ueisigi, Y. Abe, K. Sayama and H. Arakwa, Appl. Catal. A, 205, 117 (2001).
J. Chattopadhyay, H. R. Kim, S. B. Moon and D. Pak, Int. J. Hydrog. Energy, 33, 3270 (2008).
J. E. Son, J. Chattopadhyay and D. Pak, Int. J. Hydrog. Energy, 35, 420 (2010).
J. Chattopadhyay, R. Srivastava and P. K. Srivastava, J. Appl. Electrochem., 43, 279 (2013).
H. Shiho and N. Kawahashi, J. Colloid Interface Sci., 226, 91 (2000).
N. Kawahashi and H. Shiho, J. Matter. Chem., 10, 2294 (2000).
S. B. Yoon, J.Y. Kim, J. H. Kim, S.G. Park, J.Y. Kim, C.W. Lee and J. S. Yu, Curr. Appl. Phys., 6, 1059 (2000).
E. A. Ticianelli, C.R. Derouin, A. Redondo and S. Srinivasan, J. Electrochem. Soc., 135, 2209 (1988).
G. A. Niklasson and C.G. Granqvist, J. Mater. Sci., 17, 127 (2007).
D. R. Lide, CRC handbook of chemistry and physics, 73rd Ed., CRC Press, Boca Raton, USA (2000).
P. Lunkenheimer, A. Loidl, C. R. Ottermann and K. Bange, Phys. Rev. B, 44, 5927 (1991).
A. Marshall, B. Børrensen, G. Hagen, M. Tsypkin and R. Tunold, Electrochim. Acta, 51, 3161 (2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chattopadhyay, J., Srivastava, R. & Srivastava, P.K. Electrochemical performance of Ni/TiO2 hollow sphere in proton exchange membrane water electrolyzers system. Korean J. Chem. Eng. 30, 1571–1577 (2013). https://doi.org/10.1007/s11814-013-0085-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-013-0085-9