Abstract
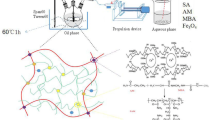
In this study, the water-based ferromagnetic fluid and magnetic resin made from chitosan and cerium complex (MRCCC) were successfully prepared by using the chemical co-precipitation technique and by the reversed-phase suspension cross-linking polymerization. MRCCC presented uniform and narrow particle size distribution as determined by the Laser Particles Sizer. The Inductively Coupled Plasma-Atomic Emission Spectrometry (ICP-AES), Fourier transform infrared spectroscopy (FT-IR), differential scanning calorimetry (DSC) and X-ray powder diffraction (XRD) study demonstrated that there were iron and cerium existing in MRCCC. The movement of MRCCC under magnetic field proved its magnetic property. The swelling kinetics in water or solutions with different pH indicated that MRCCC could be applied in solutions with pH greater than 1.0. The ferromagnetic fluid particles were stable in MRCCC soaked in solutions with pH >2.0. In view of these results, MRCCC can be used as material for separation, clarification, adsorption, sustained release and hydrolysis activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Denkbaş, E. B., Kiliçay, E., Birlikseven, C., and Öztürk, E., 2002. Magnetic chitosan microspheres: preparation and characterization. React. Funct. Polym., 50: 225–232.
Donia, A. M., Atia, A. A., and Elwakeel, K. Z., 2008. Selective separation of mercury (II) using magnetic chitosan resin modified with Schiff’s base derived from thiourea and glutaraldehyde. J. Hazard. Mater., 151: 372–379.
Franklin, S. J., 2001. Lanthanide-mediated DNA hydrolysis. Curr. Opin. Chem. Biol., 5: 201–208.
Frederick, S. R., 1980. Terrbrium (III) and Europ ium (III) ions as luminescent probes and staines forbiomolecular systems. Chem. Rev., 5: 541–552.
He, Q. X., Yang, H., Chen, Q. Q., Liu, S. Q., and Huang, K. L., 2003. Study on preparative condition of magnetic Fe3O4 nanoparticle synthesized from method of microemulsification. J. Magn. Mater. Devices., 34: 9–11 (in Chinese with English abstract).
Jakabský, Š., Lovás, M., Mockovčiaková, A., and Hredzák, S., 2000. Utilization of ferromagnetic fluids in mineral processing and water treatment. J. Radioanal. Nucl. Chem., 246: 543–547.
Kitamura, Y., Sumaoka, J., and Komiyama, M., 2003. Hydrolysis of DNA by cerium (IV)/EDTA complex. Tetrahedron, 59: 10403–10408.
Lin, H. K., Liu, Q., and Lin, H., 2006. Study on kinetics and mechanism of mononuclear rare earth metal complexes in promoting the hydrolysis of p-nitrophenyl phosphate (NPP). J. Mol. Catal. A: Chem., 259: 11–16.
Liu, Q., Chen, H. M., Lin, H., and Lin, H. K., 2007. Study on kinetics and mechanism of mononuclear rare earth metal complexes in promoting the hydrolysis of 2-hydroxy-propyl-p-nitrophenyl phosphate (HPNP). J. Mol. Catal. A: Chem., 269: 104–109.
Park, J. H., Im, K. H., Lee, S. H., Kim, D. H., Lee, D. Y., Lee, Y. K., et al., 2005. Preparation and characterization of magnetic chitosan particles for hyperthermia application. J. Magn. Magn. Mater., 293: 328–333.
Rinaudo, M., 2006. Chitin and chitosan: Properties and applications. Prog. Polym. Sci., 31: 603–632.
Santos, J. E. D., Dockal, E. R., and Cavalheiro, É. T. G., 2005. Synthesis and characterization of Schiff bases from chitosan and salicylaldehyde derivatives. Carbohyd. Polym., 60: 277–282.
Shangguan, G. Q., and Qu, X. G., 2006. Study on the stability of polyazamacrocycle-Eu (Tb) by potentiometry and catalytic hydrolysis on phosphate diester of BDBPH-Tb complexes. Chin. J. Anal. Chem., 34: 10–15.
Sumaoka, J., Kajimura, A., Ohno, M., and Komiyama, M., 1997. Homogeneous metal complexes for DNA hydrolysis and their application to artificial restriction enzymes. J. Inorg. Biochem., 67: 361–361.
Tang, W. J., Wang, C. X., and Chen, D. H., 2005. Kinetic studies on the pyrolysis of chitin and chitosan. Polym. Degrad. Stab., 87: 389–394.
Thünemann, A. F., Schütt, D., Kaufner, L., Pison, U., and Mohwald, H., 2006. Maghemite nanoparticles protectively coated with poly (ethylene imine) and poly (ethylene oxide)-block-poly (glutamic acid). Langmuir, 22: 2351–2357.
Tremaine, P. R., and Xiao, C. B., 1999. Enthalpies of formation and heat capacity functions for maricite, NaFePO4(cr), and sodium Na3Fe(PO4)2·(Na4/3H2/3O)(cr). J. Chem. Thermodyn., 31: 1307–1320.
Wang, D. F., Luo, Y., Sun, J. P., Du, D. H., Wang, C. H., Zhou, X. L., et al., 2005. The use of complexes of algae polysaccharides and Ce4+ to degrade compounds containing peptides or phosphate ester bonds. Carbohydr. Polym., 62: 1–5.
Wang, D. F., Sun, J. P., Du, D. H., Sun, L. P., Chen, Z. D., and Xue, C. H., 2007. Degradation of extraction from seaweed and its complex with rare earths for organophosphorous pesticides. J. Rare Earths, 25: 93–99 (in Chinese with English abstract).
Wang, D. F., Yu, L. N., Su, L., Zhang, Y. W., and Wang, C. H., 2006. Resins of chitosan-Ce4+ complexes application in apple juice production. Chin. Rare Earths, 27: 11–14 (in Chinese with English abstract).
Wu, H., Wang, D. F., Yu, L. N., Li, H. Y., and Shi, Y. L., 2006. Preparation of chitosan-Ce (IV) complex and its effects on preserving cucumbers and degrading parathion. J. Chin. Rare Earth Soc., 24 (suppl.): 176–181 (in Chinese with English abstract).
Yu, L. N., Wang, D. F., Li, H. Y., Su, L., and Wang, J. L., 2006. Hydrolysis activities of resins of complexes made from polysaccharides and Ce4+. J. Rare Earths, 24(special): 125–129 (in Chinese with English abstract).
Zhang, J., Zhang, S. T., Wang, Y. P., and Zeng, J. Y., 2007. Composite magnetic microspheres: Preparation and characterization. J. Magn. Magn. Mater., 309: 197–201.
Zhang, W. J., Zhang H. F., and Li D. Z., 2006. Preparation of Fe3O4 magnetic fluid by one-step method with a microemulsion reactor. Front. Chem. Chin., 3: 272–276.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, L., Wang, D., Li, H. et al. Preparation and characterization of magnetic resin made from chitosan and cerium. J. Ocean Univ. China 9, 185–192 (2010). https://doi.org/10.1007/s11802-010-0185-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11802-010-0185-7