Abstract
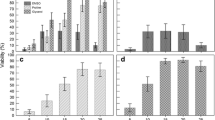
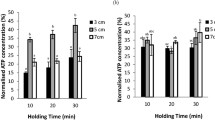
Gametophytes of Laminaria japonica were cryopreserved in liquid nitrogen using encapsulation-dehydration with two-step cooling method. Gametophytes cultured at 10°C and under continuous irradiance of 30 μmol m−2 s−1 for 3 weeks were encapsulated in calcium alginate beads. The beads were dehydrated in 0.4 molL−1 sucrose prepared with seawater for 6 h, desiccated in an incubator set at 10°C and 70% relative humidity for 4 h, pre-frozen at either −40°C or −60°C for 30 min, and stored in liquid nitrogen for >24 h. As high as 43% of survival rate was observed when gametophytes were thawed by placing the beads in 40°C seawater and re-hydrated in 0.05 molL−1 citrate sodium prepared using 30‰ NaCl 7 d later. More cells of male gametophytes survived the whole procedure in comparison with female gametophytes. The cells of gametophytes surviving the preservation were able to grow asexually and produce morphologically normal sporophytes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arbault, S., P. Renard, R. Perez, and R. Kass, 1990. Cryopreservation trials on the gametophytes of the food alga Undaria pinnatifida (Laminariale). Aquat. Living Resour., 3: 207–215.
Ben-Amotz, A., and A. Gilboa, 1980. Cryopreservation of marine unicellular algae. 2. Induction of freezing tolerance. Mar. Ecol. Progr. Ser., 2: 221–224.
Cañavate, J. P., and L. M., Lubian, 1997. Effects of culture age on cryopreservation of marine microalgae. Eur. J. Phycol., 32: 87–90.
Crutchfield, A. L. M., K. Diller, and J. J. Brand, 1999. Cryopreservation of Chlamydomonas reinhardtii (Chlorophyta). Eur. J. Phycol., 34: 43–52.
Day, J. G., and C, Fenwick, 1993. Cryopreservation of members of the genus Tetraselmis used in aquaculture. Aquaculture 118: 151–160.
Day, J. G., R. A. Fleck, and E. E. Benson, 2000. Cryopreservation-recalcitrance in microalgae: novel approaches to identify and avoid cryo-injury. J. Appl. Phycol., 12: 369–377.
Day, J. G., R. A. Fleck, and E. E. Benson, 1998. Cryopreservation of multicellular algae: problems and perspectives. Cryo-Letters, 19: 205–206.
Fang, Z. X., and J. X. Dai, 1980. The use of hapiloid phases in the genetic study of Laminaria japonica. Acta Genetica Sinica, 7: 19–25 (in Chinese).
Fenwick, C., and J. G. Day, 1992. Cryopreservation of Tetraselmis suecica cultured under different nutrients regimes. J. Appl. Phycol., 4: 105–109.
Ginsburger-Vogel, T., S. Arbeult, and R. Pérez, 1992. Ultrastructural study on the effect of freezing-thawing on the gametophytes of the brown alga Undaria pinnatifida. Aquaculture, 106: 171–181.
Grout, B. W. W. 1995. Introduction to the in vitro preservation of plant cells, tissues and organs. In: Genetic Preservation of Plant Cells in vitro. Grout, B. W. W., ed., Springer-Verlag, Berlin, 1–20.
Hatanaka, T., T. Yasuda, and T. Yamaguchi, 1994. Direct regrowth of encapsulated somatic embryos of coffee after freezing in liquid nitrogen. Cryo-Letters, 19: 7–52.
Hirata, K., M. Phunchindawan, J. Takamoto, S. Goda, and K. Miyamoto, 1996. Cryopreservation of microalgae using encapsulation-dehydration. Cryo-Letters, 17: 321–328.
Kuwano, K., S. Kono, Y. H. Jo, J. A. Shin, and N. Saga, 2004. Cryopreservation of the gametophytic cells of Laminaria (Phaeophyta) in liquid nitrogen. J. Phycol., 40: 606–610.
Kuwano, K., and N. Saga, 2000. Cryopreservation of marine algae: applications in biotechnology. In: Recent Advances in Marine Biotechnology, Vol. 4, Aquaculture., Part A, Seaweeds and invertebrates. Fingerman, M., and Nagabhushanam, R., eds., Science Publishers, USA, 23–40.
Li, Y., Q. H. Wang, H. Li, and T. T. Li, 2003. Cryopreservation of Chaetoceros muelleri by encapsulation-dehydration. Marine Sciences, 27(7): 48–51 (in Chinese).
Li, C., C. S. Loh, and W. Q. Sun, 1999. An improved dehydration protocol for cryopreservation of Brassica napus somatic embryos. Cryo-Letters, 20: 263–268.
Mortain-Bertrand, A., F. Etchart, and M. T. Deboucaud, 1996. A method for the cryoconservation of Dunaliella salina (Chlorophyceae) — Effect of glycerol and cold adaptation. J. Phycol., 32: 346–352.
Scottez, C., E. Chevreau, and N. Godard, 1992. Cryo-preservation of cold-acclimated shoot tips of pear in vitro cultures after Encapsulation-dehydration. Cryobiology, 29: 691–700.
Stillinger, F. A. 1995. Topographic view of supercooled liquids and glass formation. Science, 267: 1935–1939.
Taylor, R., and R. L. Fletcher, 1999. Cryopreservation of eukaryotic algae: a review of methodologies. J. Appl. Phycol., 10: 481–501.
Tseng, C. K. 2001, Algal biotechnology industries and research activities in China. J. Appl. Phycol., 13: 375–380.
Tseng, C. K. 1981, Marine phycoculture in China. Proc. Int. Seaweed Symp., 10: 123–152.
Vigneron, T., S. Arbault, and R. Kaas, 1997. Cryopreservation of gametophytes of Laminaria digitata (L) Lamouroux by encapsulation dehydration. Cryo-Lett., 18: 93–98.
Wang, Q. H., E. D. Zhang, Y. P. Gao, and D. P. Li, 2005a. Cryopreservation of marin golden brown algae (Chrysophyceae) used as aquaculture diet by encapsulation-dehydration. Qceanologia et Limnologia Sinica., 36(2): 172–177 (in Chinese).
Wang, Q. H., Y. P. Liu, E. D. Zhang, D. P. Li, and L. C. Zhang, 2005b. Cryopreservation of clonal gametophytes of Undaria pinnatifida by encapsulation-dehydration. Acta Oceano-logica Sinica, 27(2): 154–159 (in Chinese).
Wang, Q. H., M. Liu, and A. H. Cheng, 2000. Cryopreservation of the free living conchocelis of Porphyra haitanensis by encapsulation-dehydration. J. Liaoning Normal Univ., 23(4): 87–390 (in Chinese).
Yi, H. L., and X. X. Deng, 1999. Advances in the techniques for in vitro preservation of plant germplasm. Chin. Bull. Botany., 16(5): 574–581 (in Chinese).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Q., Cong, Y., Qu, S. et al. Cryopreservation of gametophytes of Laminaria japonica (Phaeophyta) using encapsulation-dehydration with two-step cooling method. J. Ocean Univ. China 7, 65–71 (2008). https://doi.org/10.1007/s11802-008-0065-6
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11802-008-0065-6