Abstract
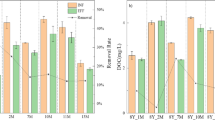
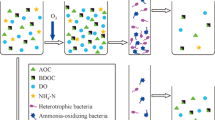
Optimizing the characteristics of granular activated carbon (GAC) can improve the performance of biologically activated carbon (BAC) filters, and iodine value has always been the principal index for GAC selection. However, in this study, among three types of GAC treating the same humic acid-contaminated water, one had an iodine value 35% lower than the other two, but the dissolved organic carbon removal efficiency of its BAC was less than 5% away from the others. Iodine value was found to influence the removal of different organic fractions instead of the total removal efficiency. Based on the removal and biological characteristics, two possible mechanisms of organic matter removal during steady-state were suggested. For GAC with poor micropore volume and iodine value, high molecular weight substances (3500–9000 Da) were removed mainly through degradation by microorganisms, and the biodegraded organics (soluble microbial by-products, < 3500 Da) were released because of the low adsorption capacity of activated carbon. For GAC with higher micropore volume and iodine value, organics with low molecular weight (< 3500 Da) were more easily removed, first being adsorbed by micropores and then biodegraded by the biofilm. The biomass was determined by the pore volume with pore diameters greater than 100 µm, but did not correspond to the removal efficiency. Nevertheless, the microbial community structure was coordinate with both the pore structure and the organic removal characteristics. The findings provide a theoretical basis for selecting GAC for the BAC process based on its pore structure.

Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Boon N, Pycke B F, Marzorati M, Hammes F (2011). Nutrient gradients in a granular activated carbon biofilter drives bacterial community organization and dynamics. Water Research, 45(19): 6355–6361
Chen M, Liu W, Tan G, Han L (2013). Study on the influential factors on expansion curves of up-flow GAC process in waterworks. Water and Wastewater Engineering, 39(3): 115–120 (in Chinese)
Chen S, Zhou Y, Chen Y, Gu J (2018). Fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics (Oxford, England), 34(17): i884–i890
Chen W, Westerhoff P, Leenheer J, Booksh K (2003). Fluorescence excitation-emission matrix regional integration to quantify spectra for dissolved organic matter. Environmental Science & Technology, 37(24): 5701–5710
Chiang Y C, Chiang P C, Huang C P (2001). Effects of pore structure and temperature on VOC adsorption on activated carbon. Carbon, 39(4): 523–534
Du Z, Jia R, Li C, Cui P, Song W, Liu J (2020). Pilot-scale UV/H2O2-BAC process for drinking water treatment — Analysis and comparison of different activated carbon columns. Chemical Engineering Journal, 382: 123044
Dussert W B, Stone V R G (1994). Biological activated carbon process for water purification. Water-Engineering & Management, 141(12): 22–24
Edgar R C (2013). UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nature Methods, 10(10): 996–998
Franz M, Arafat H A, Pinto N G (2000). Effect of chemical surface heterogeneity on the adsorption mechanism of dissolved aromatics on activated carbon. Carbon, 38(13): 1807–1819
Gibert O, Lefevre B, Fernandez M, Bernat X, Paraira M, Pons M (2013). Fractionation and removal of dissolved organic carbon in a full-scale granular activated carbon filter used for drinking water production. Water Research, 47(8): 2821–2829
Han L, Liu W, Chen M, Zhang M, Liu S, Sun R, Fei X (2013). Comparison of NOM removal and microbial properties in up-flow/down-flow BAC filter. Water Research, 47(14): 4861–1868
Herzberg M, Dosoretz C G, Kuhn J, Klein S, Green M (2006). Visualization of active biomass distribution in a BGAC fluidized bed reactor using GFP tagged Pseudomonas putida F1. Water Research, 40(14): 2704–2712
Hidayah E N, Chou Y C, Yeh H H (2016). Using HPSEC to identify NOM fraction removal and the correlation with disinfection by-product precursors. Water Science and Technology: Water Supply, 16(2): 305–313
Hou L, Zhou Q, Wu Q, Gu Q, Sun M, Zhang J (2018). Spatiotemporal changes in bacterial community and microbial activity in a full-scale drinking water treatment plant. Science of the Total Environment, 625: 449–459
Jiang W, Xia S, Liang J, Zhang Z, Hermanowicz S W (2013). Effect of quorum quenching on the reactor performance, biofouling and biomass characteristics in membrane bioreactors. Water Research, 47(1): 187–196
Jørgensen N O G, Brandt K K, Nybroe O, Hansen M (2010). Vogesella mureinivorans sp. nov., a peptidoglycan-degrading bacterium from lake water. International Journal of Systematic and Evolutionary Microbiology, 60(10): 2467–2472
Kaarela O E, Harkki H A, Palmroth M R T, Tuhkanen T A (2015). Bacterial diversity and active biomass in full-scale granular activated carbon filters operated at low water temperatures. Environmental Technology, 36(6): 681–692
Karanfil T, Kilduff J (1999). Role of granular activated carbon surface chemistry on the adsorption of organic compounds. 1. Priority pollutants. Environmental Science & Technology, 33(18): 3217–3224
Klimenko N, Winther-Nielsen M, Smolin S, Nevynna L, Sydorenko J (2002). Role of the physico-chemical factors in the purification process of water from surface-active matter by biosorption. Water Research, 36(20): 5132–5140
Korotta-Gamage S M, Sathasivan A (2017). A review: Potential and challenges ofbiologically activated carbon to remove natural organic matter in drinking water purification process. Chemosphere, 167: 120–138
Lautenschlager K, Hwang C, Ling F, Liu W T, Boon N, Koster O, Egli T, Hammes F (2014). Abundance and composition of indigenous bacterial communities in a multi-step biofiltration-based drinking water treatment plant. Water Research, 62: 40–52
Liao X, Chen C, Wang Z, Wan R, Chang C H, Zhang X, Xie S (2013a). Changes of biomass and bacterial communities in biological activated carbon filters for drinking water treatment. Process Biochemistry, 48(2): 312–316
Liao X, Chen C, Wang Z, Wan R, Chang C H, Zhang X, Xie S (2013b). Pyrosequencing analysis of bacterial communities in drinking water biofilters receiving influents of different types. Process Biochemistry, 48(4): 703–707
Lillo-Ródenas M A, Cazorla-Amorós D, Linares-Solano A (2005). Behaviour of activated carbons with different pore size distributions and surface oxygen groups for benzene and toluene adsorption at low concentrations. Carbon, 43(8): 1758–1767
Liu C, Yang J, Li C, Zhou K, Liu Y, Gao Z, Chen W (2019). The variation of the biological activated carbon during the application and the judgement criterion of its invalidation. Water & Wastewater Engineering, 45(2): 9–16 (in Chinese)
Liu S T, Yang H W, Liu W J, Zhao Y, Wang X M, Xie Y F F (2016). Evaluation of backwash strategies on biologically active carbon filters by using chloroacetic acids as indicator chemicals. Process Biochemistry, 51(7): 886–894
Lu Z, Sun W, Li C, Ao X, Yang C, Li S (2019). Bioremoval of non-steroidal anti-inflammatory drugs by Pseudoxanthomonas sp. DIN-3 isolated from biological activated carbon process. Water Research, 161: 459–472
Lu Z, Sun W, Li C, Cao W, Jing Z, Li S, Ao X, Chen C, Liu S (2020). Effect of granular activated carbon pore-size distribution on biological activated carbon filter performance. Water Research, 177: 115768
Magoč T, Salzberg S L (2011). FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics (Oxford, England), 27(21): 2957–2963
McKie M J, Bertoia C, Taylor-Edmonds L, Andrews S A, Andrews R C (2019). Pilot-scale comparison of cyclically and continuously operated drinking water biofilters: Evaluation of biomass, biological activity and treated water quality. Water Research, 149: 488–495
Moore B C, Cannon F S, Westrick J A, Metz D H, Shrive C A, DeMarco J, Hartman D J (2001). Changes in GAC pore structure during full-scale water treatment at Cincinnati a comparison between virgin and thermally reactivated GAC. Carbon, 39(6): 789–807
Morlay C, Joly J P (2010). Contribution to the textural characterisation of Filtrasorb 400 and other commercial activated carbons commonly used for water treatment. Journal of Porous Materials, 17(5): 535–543
Newcombe G, Drikas M, Hayes R (1997). Influence of characterised natural organic material on activated carbon adsorption effect on pore volume distribution and adsorption of 2-methylisoborneol. Water Research, 31(5): 1065–1073
Oh S, Hammes F, Liu W T (2018). Metagenomic characterization of biofilter microbial communities in a full-scale drinking water treatment plant. Water Research, 128: 278–285
Pharand L, Van Dyke M I, Anderson W B, Huck P M (2014). Assessment of biomass in drinking water biofilters by adenosine triphosphate. Journal- American Water Works Association, 106(10): E433–E444
Qi W, Li W, Zhang J, Wu X, Zhang J, Zhang W (2018). Effect of biological activated carbon filter depth and backwashing process on transformation of biofilm community. Frontiers of Environmental Science & Engineering, 13(1): 1–11
Rameshkumar N, Lang E, Tanaka N (2016). Description of Vogesella oryzae sp. nov., isolated from the rhizosphere of saline tolerant pokkali rice. Systematic and Applied Microbiology, 39(1): 20–24
Ross P S, van der Aa L T J, van Dijk T, Rietveld L C (2019). Effects of water quality changes on performance of biological activated carbon (BAC) filtration. Separation and Purification Technology, 212: 676–683
Servais P, Billen G, Bouillot P (1994). Biological colonization of granular activated carbon filters in drinking- water treatment. Journal of Environmental Engineering, 120(4): 888–899
Sheu S Y, Chen J C, Young C C, Chen W M (2013). Vogesella fluminis sp. nov., isolated from a freshwater river, and emended description of the genus Vogesella. International Journal of Systematic and Evolutionary Microbiology, 63(Pt_8): 3043–3049
Stackebrandt E, Goebel B M (1994). Taxonomic note: A place for DNA-DNA reassociation and 16S rRNA sequence analysis in the present species definition in bacteriology. International Journal of Systematic Bacteriology, 44(4): 846–849
Subhash Y, Tushar L, Sasikala C, Ramana C V (2013).Vogesella alkaliphila sp. nov., isolated from an alkaline soil, and emended description of the genus Vogesella. International Journal of Systematic and Evolutionary Microbiology, 63(Pt_6): 2338–2343
Wang Q, Garrity G M, Tiedje J M, Cole J R (2007). Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Applied and Environmental Microbiology, 73(16): 5261–5267
Wu Z, Zhang P, Zeng G, Zhang M, Jiang J (2012). Humic acid removal from water with polyaluminum coagulants: Effect of sulfate on aluminum polymerization. Journal of Environmental Engineering, 138(3): 293–298
Xu B, Gao N Y, Sun X F, Xia S J, Simonnot M O, Causserand C, Rui M, Wu H H (2007). Characteristics of organic material in Huangpu River and treatability with the O3-BAC process. Separation and Purification Technology, 57(2): 348–355
Yang J, Ma J, Song D, Zhai X, Kong X (2016). Impact of preozonation on the bioactivity and biodiversity of subsequent biofilters under low temperature conditions; A pilot study. Frontiers of Environmental Science & Engineering, 10(4): 5–10
Yang S D, Liao L H, Liu Z D (2014). Effect of temperature and the altitude of filler on biological activated carbon performance. Applied Mechanics and Materials, 621: 13–18
Yapsaklı K, Çeçen F, Aktaş Ö, Can Z S (2009). Impact of surface properties of granular activated carbon and preozonation on adsorption and desorption of natural organic matter. Environmental Engineering Science, 26(3): 489–500
Yu Y (2015). Impacts of Activated Carbon Porosity Feature on the Removal and Microbiological Characteristics of BAC. Dissertation for the Master’s Degree. Beijing: Tsinghua University (in Chinese)
Zheng J, Lin T, Chen W, Tao H, Tan Y, Ma B (2018). Removal of precursors of typical nitrogenous disinfection byproducts in ozonation integrated with biological activated carbon (O3/BAC). Chemosphere, 209: 68–77
Acknowledgements
This study was supported by the National Key R&D Program of China (No. 2019YFC0408700), the funds from the National Natural Science Foundation of China (Grant Nos. 51778323 and 51761125013), and the National Science and Technology Major Projects of China (Nos. 2012ZX07404-002, 2017ZX07108-002, and 2017ZX-07502003).
Author information
Authors and Affiliations
Corresponding author
Additional information
Highlights
• Pore structure affects biologically activated carbon performance.
• Pore structure determines organic matter (OM) removal mechanism.
• Microbial community structure is related to pore structure and OM removal.
Supporting Materials
Rights and permissions
About this article
Cite this article
Xu, Y., Lu, Z., Sun, W. et al. Influence of pore structure on biologically activated carbon performance and biofilm microbial characteristics. Front. Environ. Sci. Eng. 15, 131 (2021). https://doi.org/10.1007/s11783-021-1419-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11783-021-1419-1