Abstract
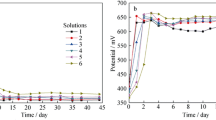
Bioleaching and electrochemical experiments were conducted to evaluate pyrrhotite dissolution in the presence of pure L. ferriphilum and mixed culture of L. ferriphilum and A. caldus. The results indicate that the pyrrhotite oxidation behavior is the preferential dissolution of iron accompanied with the massive formation of sulfur in the presence of L. ferriphilum, which significantly hinders the leaching efficiency. Comparatively, the leaching rate of pyrrhotite distinctly increases by 68% in the mixed culture of L. ferriphilum and A. caldus at the 3rd day. But, the accumulated ferric ions and high pH value produced by bioleaching process can give rise to the rapid formation of jarosite, which is the primary passivation film blocking continuous iron extraction during bioleaching by the mixed culture. The addition of A. caldus during leaching by L. ferriphilum can accelerate the oxidation rate of pyrrhotite, but not change the electrochemical oxidation mechanisms of pyrrhotite. XRD and SEM/EDS analyses as well as electrochemical study confirm the above conclusions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
ZHAO Kai-le, GU Guo-hua, LI Shuang-ke. Electrochemistry of oxidation of pyrrhotite in presence and absence of Acidthiobacillus ferrooxidans [J]. Journal of Central South University: Science and Technology, 2011, 42(10): 2887–2892. (in Chinese)
GU Guo-hua, ZHAO Kai-le, QIU Guan-zhao, HU Yu-hua, SUN Xiao-jun. Effects of Leptospirillum ferriphilum and Acidithiobacillus caldus on surface properties of pyrrhotite [J]. Hydrometallurgy, 2009, 100(1/2): 72–75.
WANG Hong-mei, BIGHAM J Y, TUOVINEN O H. Oxidation of marcasite and pyrite by iron-oxidizing bacteria and archaea [J]. Hydrometallurgy, 2007, 88(1/2/3/4): 127–131.
LIU Yun, DANG Zhi, LU Gui-ning, WU Ping-xiao, FENG Chun-hua, YI Xiao-yun. Utilization of electrochemical impedance spectroscopy for monitoring pyrite oxidation in the presence and absence of Acidithiobacillus ferrooxidans [J]. Minerals Engineering, 2011, 24(8): 833–838.
GLEISNER M, HERBERT R. Pyrite oxidation by Acidithiobacillus ferrooxidans at various concentrations of dissolved oxygen [J]. Chemical Geology, 2006, 225(1/2): 16–29.
YANG Hong-ying, YANG Li. Bacterial metallurgy [M]. Beijing: Chemical Industry Press, 2006: 10–18. (in Chinese)
SMITH R W, MISRA M, DUBEL J. Mineral bioprocessing and the future [J]. Minerals Engineering, 1991, 4(7/8/9/10/11): 1127–1141.
PAN Hao-dan, YANG Hong-ying, TONG Lin-lin, ZHONG Congbin, ZHAO Yu-shan. Control method of chalcopyrite passivation in bioleaching [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(9): 2256–2260.
STOTT M B, WATLING H R, FRANZMANN P D, SUTTON D. The role of iron-hydroxy precipitates in the passivation of chalcopyrite during bioleaching [J]. Minerals Engineering, 2000, 13(10): 1117–1127.
DEVECI H, AKCIL A, ALP I. Bioleaching of complex zinc sulphide using mesophilic and thermophilic bacteria: Comparative importance of pH and iron [J]. Hydrometallurgy, 2004, 73(3/4): 293–303.
PAN Hao-dan, YANG Hong-ying, CHEN Shi-dong, LI Wei-tao. Study of passivation phenomenon on chalcopyrite surface under different conditions [C]// Memoir of the 8th Annual Conference of Transactions of Nonferrous Metals Society of China. Beijing: The Nonferrous Metals Society of China, 2010: 146–151. (in Chinese)
YANG Hong-ying, PAN Hao-dan, TONG Lin-lin, LIU Yuan-yuan. Formation process of biological oxide film on chalcopyrite crystal surface [J]. Acta Metallurgica Sinica, 2012, 48(9): 1145–1152. (in Chinese)
STEGER H F. Oxidation of sulfide minerals: VII. Effect of temperature and relative humidity on the oxidation of pyrrhotite [J]. Chemical Geology, 1982, 35: 281–295.
SANTOS L R G, BARBOSA A F, SOUZA A D. Bioleaching of a complex nickel-iron concentrate by mesophile bacteria [J]. Minerals Engineering, 2006, 19(12): 1251–1258.
BUCKLEY A N, HAMILTON I C, WOODS R. Studies of the surface oxidation of pyrite and pyrrhotite using X-ray photoelectron spectroscopy and linear potential sweep voltammetry [J]. The Electrochemical Society, 1988, 88: 234–246.
BUCKLEY A N, WOODS R. X-ray photoelectron spectroscopy of oxidized pyrrhotite surfaces: I. Exposure to air [J]. Appl Surf Sci, 1985, 22: 280–287.
BUCKLEY A N, WOODS R. X-ray photoelectron spectroscopy of oxidized pyrrhotite surfaces: I. Exposure to aqueous solutions [J]. Appl Surf Sci, 1985, 20: 472–480.
ALMEIDA C M V B, GIANNETTI B F. The electrochemical behavior of pyrite-pyrrhotite mixtures [J]. Journal of Electroanalytical Chemistry, 2003, 553: 27–34.
ZHOU Hong-bo, ZENG Wei-ming, QIU Guan-zhou. Bioleaching of chalcopyrite concentrate by amoderately thermophilic culture in a stirred tank reactor [J]. Bioresource Technology, 2009, 100: 515–520.
BIEGLER T, HORNE M D. The electrochemistry of surface oxidation of chalcopyrite [J]. Journal of the Electrochemical Society, 1985, 132(6): 1363–1369
NAVA J L, OROPEZA M T, GONZALEZ I. Electrochemical characterisation of sulfur species formed during anodic dissolution of galena concentrate in perchlorate medium at pH 0 [J]. Electrochimica Acta, 2002, 47(10): 1513–1525.
EGHBALNIA M, DIXON D G. Electrochemical study of leached chalcopyrite using solid paraffin-based carbon paste electrodes [J]. Hydrometallurgy, 2011, 110(1/2/3/4): 1–12.
ZENG Wei-ming, QIU Guan-zhou, ZHOU Hong-bo, CHEN Miao. Electrochemical behaviour of massive chalcopyrite electrodes bioleached by moderately thermophilic microorganisms at 48 °C [J]. Hydrometallurgy, 2011, 105(3/4): 259–263.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: Project(2010CB630903) supported by the National Basic Research Program of China; Project(51374249) supported by the National Natural Science Foundation of China
Rights and permissions
About this article
Cite this article
Gu, Gh., Yang, Hs., Hu, Kt. et al. Formation of passivation film during pyrrhotite bioleached by pure L. ferriphilum and mixed culture of L. ferriphilum and A. caldus . J. Cent. South Univ. 22, 880–886 (2015). https://doi.org/10.1007/s11771-015-2597-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11771-015-2597-4