Abstract
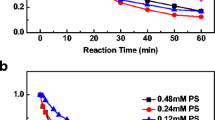
The degradation behavior of ethyl xanthate (EX) salt was the most widely used collector in sulfide mineral flotation and emission of flotation tailings with residual EX was harmful to environment. In this work, hydrogen peroxide (H2O2) was investigated by UV-visible spectroscopy (UV/Vis) at different pH values from 3 to 12. For pH value from 5 to 12, EX was oxidized into ethyl per xanthate (EPX) by H2O2. Then EPX was further oxidized into thiosulfate (TS) salt rather than ethyl thiocarbonate (ETC) and this step was the reaction-limited step. Then depending on pH values, TS was degraded into sulphate and carbonate salts (pH>7) or elemental sulfur (pH<7). The kinetics data show that the degradation rate of EX increases with increasing the H2O2 concentration and is independent on the pH values. Without H2O2, EX is hydrolyzed to carbon disulfide fast at pH value <3.0, but the reaction of hydrolysis is undetectable at pH value >3.0 during test time.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
CHEN J H, ZHAO C H, GONG Z Q. Research on the photocatalytic removal of N-butyl and isobutyl xanthates in the TiO2 suspension system [C]// 4th International Conference on Separation Science and Technology (ICSST-04). Nanning, China: Frontiers on Separation Science and Technology, 2004: 1010–1018.
National Industrial Chemicals Notification and assessmentscheme. Sodium ethyl xanthate. Priority existing chemical No 5 [R]. Canberra, Australia: Australian Government Publishing Service, 1995: 1–66.
CHEN Shao-hua, GONG Wen-qi, MEI Guang-jun, CHEN Xiao-dong, YAN Heng-zhen. Evaluation of biodegradability of alkyl xanthates flotation collecters [J]. Journal of Central South University (Science and Technology), 2011, 42(2): 546–554. (in Chinese)
SHU Sheng-hui, YANG Yong-feng, ZHANG Zhi. Treatment methods and research progress in xanthate-removal from effluent of flotation operation [J]. Multipurpose Utilization of Mineral Resources, 2009, (4): 35–36.
DUAN Hai-xia, LIU Jiong-tian, LANG Xian-ming, WANG Jing-cheng. Study development of interior xanthate wastwater treatment [J]. Mining and Metallurgy, 2009, 18(4): 80–82.
AI Guang-hua, WEI Zong-wu. Study on flotation reagents prevention and control of ecological environmental pollution [J]. Environmental Protection of Xinjiang, 2008, 30(2): 31–34.
MIELCZARSKI J. The role of impurities of sphalerite in the adsorption of ethyl xanthate and its flotation [J]. International Journal of Mineral Processing, 1986, 16(3/4): 179–194.
SILVESTER E, TRUCCOLO D, FU Ping-hao. Kinetics and mechanism of the oxidation of ethyl xanthate and ethyl thiocarbonate by hydrogen peroxide [J]. J Chem Soc Perkin Trans, 2002, 2(9): 1562–1571.
FAGADAR-COSMA G, TARANU I, FAGADAR-COSMA E. Electrochemical oxidation of sodium ethyl xanthate in aqueous solutions [J]. Rev Roum Chim, 2003, 48(2): 131–136.
XU Jin, SUN Shui-yu, ZHANG Ping, CAI He-shan. Degradation of remainder xanthate in flotation wastewater by fenton reagent [J]. Environmental Protection of Chemical Industry, 2005, 25(2): 125–127.
JI Hong-an. Study on using ozone to resolve xanthate and pine oil in the mineral wastewater [J]. Gansu Metallurgy, 2008, 30(3): 70–72. (in Chinese)
VUCINIC D R, LAZIC P M, ROSIC A A. Ethyl xanthate adsorption and adsorption kinetics on lead-modified galena and sphalerite under flotation conditions [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2006, 279(1/2/3): 96–104.
CHENG Wei, ZHANG Qin, MA Wen-qiang. A research on removal of xanthate from floatation wastewater with activated carbon [J]. Acta Mineralogica Sinica, 2010, 30(2): 262–267.
CHEN S H, GONG W Q, MEI G J, ZHOU Q, BAI C P, XU N. Primary biodegradation of sulfide mineral flotation collectors [J]. Miner Eng, 2011, 24(8): 953–955.
CHOCKALINGAM E, SUBRAMANIAN S, NATARAJAN K A. Studies on biodegradation of organic flotation collectors using Bacillus polymyxa [J]. Hydrometallurgy, 2003, 71(1/2): 249–256.
IWASAKI I, COOKE S R B. The decomposition of xanthate in acid solution [J]. J Am Chem Soc, 1958, 80(2): 285–288.
IWASAKI I, COOKE S R B. Dissociation constant of xanthic acid as determined by spectrophotometric method [J]. J Phys Chem, 1959, 63(8): 1321–1322.
FINKELSTEIN N P. Kinetic and thermodynamic aspects of the interaction between potassium ethyl xanthate and oxygen in aqueous solution [J]. Trans Inst Min Metall, 1967, 76: 51–59.
HAO F P, SILVESTER E, SENIOR G D. Spectroscopic characterization of ethyl xanthate oxidation products and analysis by ion interaction chromatography [J]. Anal Chem, 2000, 72(20): 4836–4845.
SUN Z, FORSLING W. The degradation kinetics of ethyl-xanthate as a function of pH in aqueous solution [J]. Mineral Engineering, 1997, 10(4): 389–400.
AI Guang-hua, WANG Yong, TAO Xiu-xiang, CHEN Yu-lin. Removal of xanthate in flotation wastewater by ultrasound and fenton reagent [C]// Electric Technology and Civil Engineering (ICETCE), 2011 International Conference on. Lushan, China: IEEE, 2001: 6106–6110.
RUBINSON K A, RUBINSON J F. Contemporary instrumental analysis [M]. Upper Saddle River, NJ: Prentice Hall, 2000: 653–662.
MIURA Y, KAWAOI A. Determination of thiosulfate, thiocyanate and polythionates in a mixture by ion-pair chromatography with ultraviolet absorbance detection [J]. Journal of Chromatography A, 2000, 884(1/2): 81–87.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: Project(2013AA064102) supported by the National High Technology Research and Development Program of China; Project(B14034) supported by the Program of Introducing Talents of Discipline to Universities, China; Project supported by the 2011 Collaborative Innovation Center for Clean and Efficient utilization of Strategic Metal Mineral Resources, China
Rights and permissions
About this article
Cite this article
Chen, Xh., Hu, Yh., Peng, H. et al. Degradation of ethyl xanthate in flotation residues by hydrogen peroxide. J. Cent. South Univ. 22, 495–501 (2015). https://doi.org/10.1007/s11771-015-2548-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11771-015-2548-0