Abstract
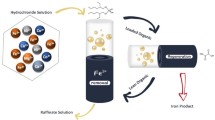
The solvent extraction of copper and zinc from the bioleaching solutions of low-grade sulfide ores with LIX984 and D2EHPA was investigated. The influences of extractant content, aqueous pH value, phase ratio and equilibration time on metals extraction were studied. The results show that LIX984 has a higher selectivity for copper than for iron, zinc and other metals, and has the copper extraction rate above 97%, while the zinc and iron extraction rate is less than 1.6% respectively. Zinc extraction is carried out following the copper extraction from the raffinate. The zinc extraction with di(2-ethylhexyl) phosphoric acid (D2EHPA) is low due to its poor cation exchange. A sodium salt of D2EHPA is used and the zinc extraction rate is enhanced to above 98%. Though iron (III) is strongly extracted before the extraction of zinc by D2EHPA, it is difficult to strip iron from the organic phase by sulfuric acid. The zinc stripping rate is above 99% with 100 g/L sulfuric acid, while that of iron is 0.16%. Hence, the separation of zinc from iron can be achieved by the selective stripping.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arbiter N, Fletcher A W. Copper hydrometallurgy—evolution and milestones[J]. Mining Engineering, 1994, 46(2): 118–123.
Kordosky G A. Copper solvent extraction: the state of the art[J]. JOM, 1992, 44 (5): 40–45.
George O. Selective extractions of Zn and Cd from Zn-Cd-Co-Ni sulphate solution using di-2-ethylhexyl phosphoric acid extractant[J]. Hydrometallurgy, 1998, 47(2): 205–215.
CHENG Chu-yong. Purification of synthetic laterite leach solution by solvent extraction using D2EHPA [J]. Hydrometallurgy, 2000, 56(3): 369–386.
Kopacz S. Extraction of zinc(II) ions from sulfate solutions with mixtures of n-hexanoic acid and pentanol in n-decane[J]. Hydrometallurgy, 1994, 35(3): 313–319.
Alguacil F J, Alonso M. The effect of ammonium sulfate and ammonia on the liquid-liquid extraction of zinc using LIX54[J]. Hydrometallurgy, 1999, 53(2): 203–209.
Sainz-Diaz C I, Klocker H, Marr R, et al. New approach in the modeling of the extraction equilibrium of zinc with bis-(2-ethylhexyl) phosphoric acid[J]. Hydrometallurgy, 1996, 42(1): 1–11.
Amer S, Figueiredo J M, Luis A. The recovery of zinc from the liquors of the CHENIM-LENTI process by solvent extraction with di (2-ethylhexyl) phosphoric acid[J]. Hydrometallurgy, 1995, 37(3): 323–337.
Svendsen H F, Schei G O M. Kinetics of extraction of zinc by di (2-ethylhexyl) phosphoric acid in cumene [J]. Hydrometallurgy, 1990, 25(2): 197–212.
Ritcey G M, Lucas B H, Price K T. Evaluation and selection of extractants for the separation of copper and zinc from chloride leach liquor[J]. Hydrometallurgy, 1982, 8(3): 197–222.
Dukov I L. Solvent extraction of zinc and copper with mixtures of LIX34 and versatic 911 in kerosene[J]. Hydrometallurgy, 1982, 8(1): 77–82.
Vazarlis H, Neou-Syngoyan P. A study of the leaching of copper and zinc from a Greek copper concentrate: liquid-liquid extraction for the separation of copper, zinc and iron from the leach solutions[J]. Hydrometallurgy, 1984, 12(3): 365–373.
George O. Selective extraction of copper from acidic zinc sulfate leach solution using LIX 622[J]. Hydrometallurgy, 1999, 51(1): 1–8.
Devi N B, Nathsarma K C, Chakravortty V. Extraction and separation of Mn and Zn from sulfate solution by sodium salt of Cyanex 272[J]. Hydrometallurgy, 1997, 45(1–2): 169–179.
GU H, CHANG C M, Barrera-Godinez J A. Preliminary design of a solvent extraction process for the galvanic stripping of iron from D2EHPA[J]. Minerals and Metallurgical Processing, 2000, 17(1): 16–22.
Author information
Authors and Affiliations
Additional information
Foundation item: Project (50321402) supported by the National Natural Science Foundation of China
Rights and permissions
About this article
Cite this article
Lan, Zy., Hu, Yh., Liu, Js. et al. Solvent extraction of copper and zinc from bioleaching solutions with LIX984 and D2EHPA. J Cent. South Univ. Technol. 12, 45–49 (2005). https://doi.org/10.1007/s11771-005-0201-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11771-005-0201-z