Abstract
In brief, perioperative oral intervention consists of elimination of odontogenic foci and maintenance of oral hygiene in patients undergoing surgery. The importance of oral intervention before, during, and after medical treatments is well-known, especially in cancer patients, because odontogenic foci such as untreated deep dental caries or periodontitis can cause systemic infection in patients with myelosuppression resulting from chemotherapy. Although perioperative oral intervention is currently recommended for patients with cardiovascular disease, its efficacy in this population has not been established. This article consists of three sections: first, we review the current knowledge about the association between dental disease and cardiovascular disease to show the importance of oral hygiene maintenance and the risks of invasive dental procedures in patients with cardiovascular disease; second, we introduce pertinent, but limited evidence concerning the effect of oral care in preventing postoperative pneumonia; and finally, we present the optimal strategy for perioperative oral intervention in cardiovascular surgery patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Perioperative oral intervention generally consists of oral assessment and elimination of odontogenic foci before treatment, oral hygiene maintenance during treatment, and patient education during the post-treatment period. The importance of prechemotherapeutic oral intervention for patients with hematological malignancy is widely known [1]. Myelosuppressive chemotherapy for hematological malignancy or hematopoietic stem cell transplantation places patients in an immunocompromised state [2]. Odontogenic foci such as untreated deep dental caries, periodontitis, or erupted wisdom teeth can cause infection, possibly resulting in interruption of medical treatments or life-threatening septicemia [3]. Before the initiation of chemotherapy, oral surgeons must precisely diagnose and eliminate odontogenic foci that could cause local or systemic infection. During the chemotherapeutic period, good oral hygiene should be maintained to prevent exacerbation of chemotherapy-induced oral mucositis [4].
In addition to its benefits in patients with hematological malignancy, perioperative oral care was recently reported to reduce the risk of postoperative pneumonia (PP) in patients undergoing esophageal cancer surgery [5]. Some systematic reviews have indicated that good oral hygiene helps prevent pneumonia and respiratory tract infection in hospitalized elderly people, elderly nursing home residents [6], and nonventilated individuals [7]. However, it should be noted that the trials included in these systematic reviews have the risk of bias [7] and methodological limitations [8]. Furthermore, the true efficacy of oral hygiene care in ventilated individuals remains controversial. Although oral hygiene care, including chlorhexidine mouthwash or gel, can reduce the risk of ventilator-associated pneumonia (VAP) in critically ill patients, there is no evidence that such care affects mortality, duration of mechanical ventilation, or duration of stay in the intensive care unit [9, 10].
A 2015 article by Jenkins et al. entitled “What is the role of the oral and maxillofacial department in the preoperative management of patients awaiting cardiac operations?” [11] proposed a clinical pathway for dental treatment in patients awaiting cardiac surgery. That report especially focused on the risk of infectious endocarditis (IE) caused by dental disease and its subsequent treatment, while acknowledging the limitations of the evidence and available guidance regarding preoperative dental assessment in these patients.
Although perioperative oral intervention is currently recommended for patients with cardiovascular disease (CVD), its efficacy in this population has not been established. This article consists of three sections. First, we review the current knowledge about the association between dental disease (e.g., periodontitis) and CVD. Second, we introduce the results of recent studies showing the efficacy of oral care in preventing PP. Finally, we describe what we consider to be the optimal strategy for perioperative oral intervention in cardiovascular surgery patients.
Association between dental disease and cardiovascular disease
Although it remains unclear whether the relationship is causal or coincidental, an association between odontogenic foci and CVD has been reported [12]. Epidemiologic evidence has revealed that periodontitis is associated with an increased risk for future CVD [13]. A recent cross-sectional analysis of a large cohort of 60,174 participants in the Netherlands showed an independent association between periodontitis and atherosclerotic CVD [14]. A Swedish study including 5297 individuals reported that individuals who did not respond well to periodontal treatment had an increased risk for future CVD (myocardial infarction, stroke, and heart failure) [15]. A Finnish population-based survey of 8446 subjects with 13 years of follow-up reported that having five or more missing teeth was associated with a 60–140% increased risk of incident coronary heart disease events and acute myocardial infarction [16].
One of major established risk factors for both CVD and periodontal disease is cigarette smoking [17, 18]. A systematic review concluded that a causal association between smoking and tooth loss is highly likely [18]. A large-scale European prospective cohort study including 23,376 participants reported that cigarette smoking was associated with higher prevalence of tooth loss, whereas smoking cessation was consistently associated with a reduction in tooth loss risk, with this risk approaching that of never smokers after about 10–20 years of cessation [17]. The gingival and oropharyngeal flora of smokers contains fewer aerobic and anaerobic organisms with interfering activity against bacterial pathogens and harbors more potential pathogens as compared with the flora of non-smokers [19]. Importantly, the high number of pathogens and the low number of interfering organisms in the nasopharynx of smokers revert to normal levels after complete cessation of smoking [19].
The association between periodontal disease and aortic aneurysm (AA) is well described. Suzuki et al. [20] compared 12 patients with abdominal AA with 24 age- and sex-matched CVD patients without abdominal AA and found that AA patients had deeper periodontal pockets than controls, despite the fact that there was no difference in the number of remaining teeth or in the populations of periodontal bacteria in oral samples. The same authors compared the prevalence of periodontitis among 25 abdominal AA patients and 142 arrhythmia patients and found that the AA patients had significantly fewer remaining teeth and deeper pockets compared with patients with arrhythmia, despite the fact that the periodontal bacteria present in saliva or subgingival plaque and serum antibodies were comparable between the AA and arrhythmia groups [21]. A study by Ding et al. [22] comparing 89 AA patients with 59 age- and sex-matched controls found that the prevalence of periodontal pathogens was higher in the oral samples of AA patients than in the samples of controls; importantly, some oral pathogens were detected in dissected aneurysm samples. A study analyzing histopathological features of AA also demonstrated the existence of Porphyromonas gingivalis (the most common organism linked to adult forms of periodontal disease) on immunochemistry examination of AA specimens [23, 24]. The major characteristic of Porphyromonas gingivalis-positive aortic tissue is smooth muscle cell proliferation, as revealed by the expression of a proliferative phenotypic marker of smooth muscle cells [25]. The expression of this proliferative marker on the surface of smooth muscle cells is found in injured blood vessels [25] and in human atherosclerotic lesions [26], and reportedly influences atherogenesis and its complications [23]. Porphyromonas gingivalis can accelerate atherosclerosis in vitro and in mouse models [24]. Oral hematogenous spread of Porphyromonas gingivalis is considered one of the causative events in the development of aortic hyperplasia in periodontitis patients [25]. These findings indicate that maintaining good oral hygiene may contribute to the prevention of CVD.
The most well-known and problematic CVD that may be caused by odontogenic foci and invasive dental treatments is IE [11]. Oral viridans group streptococci are implicated as causal microorganisms in 35–45% of IE cases [27]. Performing invasive dental treatments for odontogenic foci before heart valve replacement may be advisable to decrease the risk of prosthetic valve IE [28]. The American Heart Association recommends antibiotic prophylaxis to prevent bacteremia from invasive dental procedures only in patients at high-risk (i.e., those with previous IE, prosthetic heart valves, valves repaired with prosthetic material, unrepaired cyanotic congenital heart disease, or some repaired congenital heart defects) [29]. In contrast, in 2008 the UK National Institute for Health and Clinical Excellence (NICE) recommended complete cessation of antibiotic prophylaxis [30]. Prescriptions for antibiotic prophylaxis fell after introduction of the NICE guideline; thereafter the incidence of IE increased significantly in England [27]. Therefore, the NICE guideline has recently been changed [30]. Importantly, excellent oral hygiene and regular dental evaluation are considered important to reduce the risk of IE [11].
The risks of invasive dental procedures in patients with CVD have also been described. Smith et al. [32] investigated the mortality rate among 205 patients who underwent 208 dental extractions before planned cardiac surgery [most commonly valvular surgery or coronary artery bypass graft (CABG)] and reported that the mortality rate within 30 days of dental extraction was 6% (3% died preoperatively and 3% died postoperatively). In contrast, Nakamura et al. [33] investigated the optimal timing of treatment for advanced periodontitis in patients undergoing elective heart valve surgery and found that no complications arose from tooth extraction, either among patients who underwent extraction within 2 weeks before valve surgery or those whose valve surgery was more than 2 weeks after extraction. The authors concluded that dental treatment, including extraction, could be performed safely prior to valve surgery [33].
Some previous studies have indicated the potential risks of dental extraction in patients with AA. Umino et al. [34] reported a case of unexpected atrial fibrillation in response to tooth extraction in a 70-years-old patient with thoracic AA. Those authors advocated that even when a low concentration of epinephrine is used, practitioners should be aware of the possibility of unexpected cardiovascular reactions in elderly patients with severe CVD. They further recommended that an electrocardiogram, blood pressure device, and pulse oximeter should be used in high-risk patients to prevent and detect potentially dangerous cardiovascular emergencies, even if dental treatment is performed under local anesthesia [34]. Peters et al. [35] and Imai et al. [36] reported cases of bleeding from the tooth socket after extraction or spontaneous hemorrhage from the dental alveolus and ecchymosis as manifestations of thoracoabdominal AA-associated disseminated intravascular coagulation. This evidence suggests that dental extractions in patients with AA should be performed by oral surgeons who understand the increased risks of cardiac failure and aortic rupture resulting from a sudden increase in blood pressure caused by pain, mental stress, or local anesthesia during tooth extraction [37].
Oral hygiene and postoperative pneumonia
Another important role of the oral medicine team in the management of cardiovascular surgery patients is the prevention of PP. Bacterial colonization of oropharyngeal tissues and dental plaque are major precursors to the development of respiratory infection [38]. Implementing protocols to prevent aspiration and improve oral hygiene reduces the occurrence of PP in patients after cardiac surgery [39, 40]. Although simple comparisons are not appropriate because of differences in the definition of PP and study populations among previously reported studies, the reported incidence of PP after cardiac surgery is between 2% and 10%, especially during the first week after surgery [41]. Allou et al. [41] reported that the incidence of PP diagnosed during the first 7 days after surgery (the period of highest PP risk) was 3.1% (174/5582). Strobel et al. [42] reported that the incidence of PP among 16,804 patients who underwent CABG was 3.3%. Santos et al. [43] reported that the incidence of PP in the first 10 days after complex heart surgery was 8.1%.
Some studies have evaluated the efficacy of strict perioperative oral management in patients undergoing cardiac surgery. Starks and Harbert [39] reported that the incidence of PP after cardiothoracic surgery was 11% before implementation of an aspiration prevention protocol that included strict oral care; after implementation of the protocol, no patients developed PP. Bergan et al. [40] reported in their single-arm prospective intervention study that the incidence of PP following cardiac surgery was 8% (18/226 patients who underwent valve surgery or CABG) after improvement in perioperative oral health through tooth- and tongue-brushing instruction and routine oral rinsing with chlorhexidine gluconate. In Bergan’s prospective study, the presence of tongue plaque and poor denture hygiene significantly increased the risk of PP [40]. If not properly cleaned, dentures may easily serve as a reservoir for bacteria in edentulous patients. Therefore, all patients, dentate or not, should receive oral evaluation before cardiac surgery [40].
Strategy for perioperative oral intervention in cardiovascular surgery patients
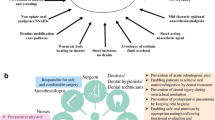
All patients scheduled for surgical intervention for CVD should undergo oral assessment. In each case, the attending cardiovascular surgeon or cardiologist should inform the oral surgeon of the patient’s diagnosis and treatment schedule if possible. An oral surgeon performs a clinical examination of the hard and soft oral tissues with panoramic radiography; a dental hygienist performs a periodontal examination. All dental diseases (caries, apical periodontitis, marginal periodontitis, and impacted wisdom teeth), unfitted dental restorations (inlay, crown, or bridge), and denture use should be recorded. An oral surgeon plans the dental treatment, with consideration of the time limitations until scheduled treatments for CVD. The presence of loose teeth at high-risk of avulsion during intubation for surgery should be carefully evaluated. Teeth with marginal periodontitis with severe gingival inflammation (pain, swelling, redness, purulent discharge) and severe mobility categorized as Miller’s class III [44] are reasonable candidates for prophylactic dental extraction. A dental hygienist can instruct patients with poor oral condition to improve their oral hygiene with the goal of maintaining a Plaque Index less than 20% [45] and can perform professional mechanical tooth cleaning. In patients with valve disease and a risk of IE, antibiotic prophylaxis (amoxicillin, 2 g orally 1 h prior to dental procedures, including tooth extraction or scaling) is recommended, according to the guidelines of the American Heart Association [29]. When patients requiring immediate surgery are referred to an oral surgeon just before surgery (e.g., 1 day preoperatively), invasive procedures such as tooth extraction and scaling should generally be avoided, and only professional mechanical tooth cleaning should be performed. In other words, in patients who lack sufficient time for prophylactic dental extraction, unpreservable teeth should be conservatively treated, even if their condition warrants extraction. This policy should avoid unexpected hemorrhage resulting from dental extraction and prevent the delay of scheduled cardiovascular surgery. However, it may be better for clearly infected teeth with persistent purulent drainage and teeth with severe periodontitis that have a high-risk of avulsion during intubation to be extracted even 1 day before surgery. If an oral surgeon determines that it is best to extract unpreservable teeth, an attending cardiovascular surgeon should be informed of the necessity of dental extraction. After cardiovascular surgery and during the postoperative period, patients should receive routine professional oral management by dental hygienists and daily mouth cleaning by nurses. When the patient’s postoperative condition stabilizes, teeth with an indication for extraction should be extracted.
Oral care intervention seems to be important for the prevention of PP in cardiovascular surgery patients. Although previous studies have described the detailed procedure of oral care [38, 39], appropriate procedures remain undetermined. A meta-analysis by Klompas et al. [46] comparing cardiac versus non-cardiac surgery patients noted that cautions about the effect of chlorhexidine in preventing VAP are warranted: routine oral care with oral antiseptic (chlorhexidine) prevents nosocomial pneumonia only in cardiac surgery patients and may not decrease the risk of VAP in non-cardiac surgery patients. The reasons for discrepant findings between cardiac versus non-cardiac surgery patients include the fact that most cardiac surgery patients are extubated within 1 day and that the pneumonia that occurs among cardiac surgery patients is lower respiratory tract infection, not VAP [46]. In other words, cardiovascular surgery patients may benefit more than non-cardiac surgery patients from oral care with oral antiseptic. In fact, the guidelines of the Centers for Disease Control and Prevention suggest implementing a 0.12% chlorhexidine gluconate rinse during the perioperative period only for patients who undergo cardiac surgery and not for any other postoperative or critically ill patients [38]. However, further deliberation about the appropriate oral care protocol for the prevention of PP in cardiovascular surgery patients is necessary.
Summary
Current knowledge indicates that maintaining good oral hygiene may contribute to the prevention of CVD. Odontogenic foci should be precisely eliminated by oral surgeons who understand the risk of invasive dental procedures in CVD patients. Cardiovascular surgery patients are the only patient population in which implementation of antiseptic rinsing during the perioperative period is recommended.
References
Elad S, Raber-Durlacher JE, Brennan MT, Saunders DP, Mank AP, Zadik Y, et al. Basic oral care for hematology-oncology patients and hematopoietic stem cell transplantation recipients: a position paper from the joint task force of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) and the European Society for Blood and Marrow Transplantation (EBMT). Support Care Cancer. 2015;23(1):223–36.
Kishimoto M, Akashi M, Tsuji K, Kusumoto J, Furudoi S, Shibuya Y, et al. Intensity and duration of neutropenia relates to the development of oral mucositis but not odontogenic infection during chemotherapy for hematological malignancy. PLoS One. 2017;12(7):e0182021.
Akashi M, Shibuya Y, Kusumoto J, Furudoi S, Inui Y, Yakushijin K, et al. Myelosuppression grading of chemotherapies for hematologic malignancies to facilitate communication between medical and dental staff: lessons from two cases experienced odontogenic septicemia. BMC Oral Health. 2013;13:41.
McGuire DB, Fulton JS, Park J, Brown CG, Correa ME, Eilers J, et al. Systematic review of basic oral care for the management of oral mucositis in cancer patients. Support Care Cancer. 2013;21(11):3165–77.
Soutome S, Yanamoto S, Funahara M, Hasegawa T, Komori T, Yamada SI, et al. Effect of perioperative oral care on prevention of postoperative pneumonia associated with esophageal cancer surgery: A multicenter case-control study with propensity score matching analysis. Medicine (Baltimore). 2017;96(33):e7436.
Sjögren P, Nilsson E, Forsell M, Johansson O, Hoogstraate J. A systematic review of the preventive effect of oral hygiene on pneumonia and respiratory tract infection in elderly people in hospitals and nursing homes: effect estimates and methodological quality of randomized controlled trials. J Am Geriatr Soc. 2008;56(11):2124–30.
Kaneoka A, Pisegna JM, Miloro KV, Lo M, Saito H, Riquelme LF, et al. Prevention of healthcare-associated pneumonia with oral care in individuals without mechanical ventilation: a systematic review and meta-analysis of randomized controlled trials. Infect Control Hosp Epidemiol. 2015;36(8):899–906.
Arpin S. Oral hygiene in elderly people in hospitals and nursing homes. Evid Based Dent. 2009;10(2):46.
Shi Z, Xie H, Wang P, Zhang Q, Wu Y, Chen E, et al. Oral hygiene care for critically ill patients to prevent ventilator-associated pneumonia. Cochrane Database Syst Rev. 2013;(8):CD008367.
Hua F, Xie H, Worthington HV, Furness S, Zhang Q, Li C. Oral hygiene care for critically ill patients to prevent ventilator-associated pneumonia. Cochrane Database Syst Rev. 2016;10:CD008367.
Jenkins GW, Holmes A, Colman-Nally J, Mustafa S. What is the role of the oral and maxillofacial department in the preoperative management of patients awaiting cardiac operations? Br J Oral Maxillofac Surg. 2015;53(5):442–5.
Demmer RT, Desvarieux M. Periodontal infections and cardiovascular disease: the heart of the matter. J Am Dent Assoc. 2006;137:14S–20S (quiz 38S)
Tonetti MS, Van Dyke TE. working group 1 of the joint EFP/AAP workshop. Periodontitis and atherosclerotic cardiovascular disease: consensus report of the Joint EFP/AAP Workshop on Periodontitis and Systemic Diseases. J Periodontol. 2013;84(4 Suppl):24–9.
Beukers NG, van der Heijden GJ, van Wijk AJ, Loos BG. Periodontitis is an independent risk indicator for atherosclerotic cardiovascular diseases among 60,174 participants in a large dental school in the Netherlands. J Epidemiol Community Health. 2017;71(1):37–42.
Holmlund A, Lampa E, Lind L. Poor response to periodontal treatment may predict future cardiovascular disease. J Dent Res. 2017;96(7):768–73.
Liljestrand JM, Havulinna AS, Paju S, Männistö S, Salomaa V, Pussinen PJ. Missing teeth predict incident cardiovascular events, diabetes, and death. J Dent Res. 2015;94(8):1055–62.
Dietrich T, Walter C, Oluwagbemigun K, Bergmann M, Pischon T, Pischon N, et al. Smoking, smoking cessation, and risk of tooth loss: the EPIC-potsdam study. J Dent Res. 2015;94(10):1369–75.
Hanioka T, Ojima M, Tanaka K, Matsuo K, Sato F, Tanaka H. Causal assessment of smoking and tooth loss: a systematic review of observational studies. BMC Public Health. 2011;11:221.
Brook I. The impact of smoking on oral and nasopharyngeal bacterial flora. J Dent Res. 2011;90(6):704–10.
Suzuki J, Aoyama N, Aoki M, Tada Y, Wakayama K, Akazawa H, et al. High incidence of periodontitis in Japanese patients with abdominal aortic aneurysm. Int Heart J. 2014;55(3):268–70.
Suzuki J, Aoyama N, Aoki M, Tada Y, Wakayama K, Akazawa H, et al. Incidence of periodontitis in Japanese patients with cardiovascular diseases: a comparison between abdominal aortic aneurysm and arrhythmia. Heart Vessels. 2015;30(4):498–502.
Ding F, Lyu Y, Han X, Zhang H, Liu D, Hei W, et al. Detection of periodontal pathogens in the patients with aortic aneurysm. Chin Med J. 2014;127(23):4114–8.
Nakano K, Wada K, Nomura R, Nemoto H, Inaba H, Kojima A, et al. Characterization of aortic aneurysms in cardiovascular disease patients harboring porphyromonas gingivalis. Oral Dis. 2011;17(4):370–8.
Gibson FC 3rd, Yumoto H, Takahashi Y, Chou HH, Genco CA. Innate immune signaling and Porphyromonas gingivalis-accelerated atherosclerosis. J Dent Res. 2006;85(2):106–21.
Hokamura K, Umemura K. Roles of oral bacteria in cardiovascular diseases—from molecular mechanisms to clinical cases: Porphyromonas gingivalis is the important role of intimal hyperplasia in the aorta. J Pharmacol Sci. 2010;113(2):110–4.
McCormick MM, Rahimi F, Bobryshev YV, Gaus K, Zreiqat H, Cai H, et al. S100A8 and S100A9 in human arterial wall. Implications for atherogenesis. J Biol Chem. 2005;280(50):41521–9.
Dayer MJ, Jones S, Prendergast B, Baddour LM, Lockhart PB, Thornhill MH. Incidence of infective endocarditis in England, 2000-13: a secular trend, interrupted time-series analysis. Lancet. 2015;385(9974):1219–28.
Lockhart PB, Brennan MT, Cook WH, Sasser H, Lovell RD, Skipper ER, et al. Concomitant surgical treatment of dental and valvular heart diseases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;107(1):71–6.
Wilson W, Taubert KA, Gewitz M, Lockhart PB, Baddour LM, Levison M, et al. Prevention of infective endocarditis: guidelines from the American Heart Association: a guideline from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee, Council on Cardiovascular Disease in the Young, and the Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and the Quality of Care and Outcomes Research Interdisciplinary Working Group. Circulation. 2007;116(15):1736–54.
Richey R, Wray D, Stokes T, Guideline Development Group. Prophylaxis against infective endocarditis: summary of NICE guidance. BMJ. 2008;336(7647):770–1.
Thornhill MH, Dayer M, Lockhart PB, McGurk M, Shanson D, Prendergast B, et al. A change in the NICE guidelines on antibiotic prophylaxis. Br Dent J. 2016;221(3):112–4.
Smith MM, Barbara DW, Mauermann WJ, Viozzi CF, Dearani JA, Grim KJ. Morbidity and mortality associated with dental extraction before cardiac operation. Ann Thorac Surg. 2014;97(3):838–44.
Nakamura Y, Tagusari O, Seike Y, Ito Y, Saito K, Miyamoto R, et al. Prevalence of periodontitis and optimal timing of dental treatment in patients undergoing heart valve surgery. Interact Cardiovasc Thorac Surg. 2011;12(5):696–700.
Umino M, Ohwatari T, Shimoyama K, Nagao M. Unexpected atrial fibrillation during tooth extraction in a sedated elderly patient. Anesth Prog. 1994;41(3):77–80.
Peters KA, Triolo PT, Darden DL. Disseminated intravascular coagulopathy: manifestations after a routine dental extraction. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005;99(4):419–23.
Imai T, Michizawa M, Shimizu H, Yura Y, Doi Y. Spontaneous intraoral hemorrhage as manifestation of thoracoabdominal aortic aneurysm-associated disseminated intravascular coagulation: case report and review. J Oral Maxillofac Surg. 2010;68(1):195–200.
Seto M, Matsuda M, Narihira K, Kikuta T. Oral surgery under local anesthesia with dexmedetomidine sedation in a morbidly obese patient with aortic dissection. J Korean Assoc Oral Maxillofac Surg. 2016;42(2):162–5.
Garcia R. A review of the possible role of oral and dental colonization on the occurrence of health care-associated pneumonia: underappreciated risk and a call for interventions. Am J Infect Control. 2005;33(9):527–41.
Starks B, Harbert C. Aspiration prevention protocol: decreasing postoperative pneumonia in heart surgery patients. Crit Care Nurse. 2011;31(5):38–45.
Bergan EH, Tura BR, Lamas CC. Impact of improvement in preoperative oral health on nosocomial pneumonia in a group of cardiac surgery patients: a single arm prospective intervention study. Intensive Care Med. 2014;40(1):23–31.
Allou N, Bronchard R, Guglielminotti J, Dilly MP, Provenchere S, Lucet JC, et al. Risk factors for postoperative pneumonia after cardiac surgery and development of a preoperative risk score*. Crit Care Med. 2014;42(5):1150–6.
Strobel RJ, Liang Q, Zhang M, Wu X, Rogers MA, Theurer PF, et al. A preoperative risk model for postoperative pneumonia after coronary artery bypass grafting. Ann Thorac Surg. 2016;102(4):1213–9.
Santos M, Braga JU, Gomes RV, Werneck GL. Predictive factors for pneumonia onset after cardiac surgery in Rio de Janeiro, Brazil. Infect Control Hosp Epidemiol. 2007;28(4):382–8.
Miller PD Jr. A classification of marginal tissue recession. Int J Periodontics Restorative Dent. 1985;5(2):8–13.
O’Leary TJ, Drake RB, Naylor JE. The plaque control record. J Periodontol. 1972;43(1):38.
Klompas M, Speck K, Howell MD, Greene LR, Berenholtz SM. Reappraisal of routine oral care with chlorhexidine gluconate for patients receiving mechanical ventilation: systematic review and meta-analysis. JAMA Intern Med. 2014;174(5):751–61.
Acknowledgements
We thank Rebecca Tollefson, DVM, from Edanz Group (http://www.edanzediting.com/ac) for editing a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no disclosures.
Rights and permissions
About this article
Cite this article
Akashi, M., Nanba, N., Kusumoto, J. et al. Perioperative intervention by oral medicine team in cardiovascular surgery patients. Gen Thorac Cardiovasc Surg 67, 197–202 (2019). https://doi.org/10.1007/s11748-018-1020-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11748-018-1020-0