Abstract
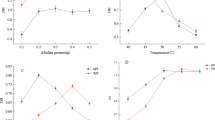
Biopolymers from soy protein isolate (SPI) crosslinked with glutaraldehyde (GA) were prepared. Surface hydrophobicities of SPI-GA biopolymers and SPI were 4.4 and 11.5, respectively. The solubility profile of SPI was slightly higher than that of SPI-GA biopolymers. Foaming capacities of SPI-GA biopolymers (23 mL) were higher than that of SPI (19 mL), but similar to egg white (22 mL). Foaming stabilities of SPI-GA biopolymers (120 min) were significantly higher than those of SPI (40 min) and egg white (98 min). The emulsifying properties of SPI-GA biopolymers were lower than those of SPI and bovine serum albumin (P>0.05). Tensile strength (TS) and elongation at break (ETB) of SPI-GA biopolymer films were significantly higher than those of glycerol-plasticized soy protein films. TS and ETB of SPI-GA biopolymer films increased with increasing GA concentrations. GA treatment intensified yellowness of SPI-GA biopolymer films. SPI-GA biopolymers may have potential use for biodegradable packaging materials.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ly, Y., J. Jane, and L.A. Johnson, Soy Proteins as Biopolymers, in Biopolymers from Renewable Resources, edited by D. Kaplan Springer-Verlag, Heidelberg, 1998, pp. 171–200.
Matsuda, S., H. Iwata, N. Se, and Y. Ikada, Bioadhesion of Gelatin Films Cross-linked with Glutaraldehyde, J. Biomed. Mater. Res. 45:20–27 (1999).
Huang-Lee, L.L.H., D.T. Cheung, and M.E. Nimni, Biomedical Changes and Cytotoxicity Associated with the Degradation of Polymeric Glutaraldehyde-Derived Cross-links, 24:1185–1201 (1990).
Ziegler, K., I. Schmitz, and H. Zahn, Introduction of New Crosslinks into Proteins, Adv. Exp. Med. Biol. 86A:345–354 (1974).
Feeney, R.E., G. Blankenhorn, and H.B.F. Dixon, Carbonylamine Reactions in Protein Chemistry, Adv. Protein Chem. 29:135–203 (1975).
Hood, L.L., Collagen in Sausage Casings, in Advances in Meat Research, edited by A.M. Pearson, T.R. Dutson, and A.J. Bailey, Van Nostrand Reinhold Co., Inc., New York, 1987, Vol. 4, pp. 109–129.
Ghorpade, V.M., H. Li, A. Gennadios, and M.A. Hanna, Chemically Modified Soy Protein Films, Trans. ASAE 38:1805–1809 (1995).
Wolf, W.J., and J.C. Cowan, Soybeans as a Food Source, revised edition, CRC Press, Inc., Cleveland, 1975, pp. 101–105.
Hayakawa, S., and S. Nakai, Relationships of Hydrophobicity and Net Charge to the Solubility of Milk and Soy Protein, J. Food Sci. 50:486–491 (1985).
Pearce, K.N., and J.E. Kinsella, Emulsifying Properties of Proteins: Evaluation of Turbidimetric Techniques, J. Agric. Food Chem. 26:716–723 (1978).
Kato, A., A. Takahashi, N. Matsudomi, and K. Kobayashi, Determination of Foaming Properties of Proteins by Conductivity Measurement. J. Food Sci. 48:62–65 (1983).
ASTM, Standard Test Method for Tensile Properties of Thin Plastic Sheeting, in Annual Book of American Standard Testing Methods, Philadelphia, 1990, D882-90.
Francis, F.J., and F.M. Clydesdale, Food Colorimetry: Theory and Applications, AVI Publishing, Westport, 1975, pp. 171–181.
SAS Institute Inc., SAS/STAT User's Guide, Statistical Analysis System Institute, Cary, NC, 1994.
Darmodaran, S., Structure-Function Relationship of Food Protein, in Protein Functionality in Food Systems, edited by N.S. Hettiarachchy and G.R. Ziegler, Marcel Dekker Inc., New York, 1994, pp. 1–37.
Chtel, J.C., J.L. Cug, and D. Lorient, Amino Acids, Peptides, and Protein, in Food Chemistry, edited by O.W. Fennema, Marcel Dekker Inc., New York, 1985, pp. 245–370.
Yildirim, M., N.S. Hettiarachchy, and U. Kalapathy, Properties of Biopolymers from Cross-Linking Whey Protein Isolate and Soybean 11S Globulin, J. Food Sci. 61:1129–1131 (1996).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Park, S.K., Bae, D.H. & Rhee, K.C. Soy protein biopolymers cross-linked with glutaraldehyde. J Amer Oil Chem Soc 77, 879–884 (2000). https://doi.org/10.1007/s11746-000-0140-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11746-000-0140-3