Abstract
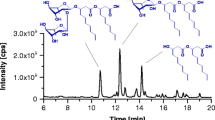
Biosurfactants are produced as a mixture of different homologs. The application of liquid chromatography-electrospray mass spectrometry in negative mode for the analysis of rhamnolipid mixtures AT10 and 47T2 has been studied. Working at low (up to −35V) extraction voltages, the [M-H]− for each compound was obtained. Increasing this potential to −75V produced an increase in the fragmentation of compounds and enabled the co-eluting isomers of rhamnolipids to be distinguished and their proportions in the sample to be calculated. In this work, the physicochemical and biological properties of two different rhamnolipid mixtures produced by two Pseudomonas strains RLAT10 (Rha-Rha-C10-C10; Rha-Rha-C10-C12:1; Rha-Rha-C10-C12; Rha-C10-C12:1; Rha-C10-C10; RhaC10-C12:1; Rha-C10-C12; Rha-C8:1; Rha-C12:2) and RL47T2 (Rha-Rha-C8-C10; Rha-Rha-C8-C12:1; Rha-Rha-C10-C10; Rha-Rha-C10-C12:1; Rha-Rha-C10-C12; Rha-Rha-C12-C10; Rha-Rha-C12:1-C12; Rha-Rha-C10-C14:1; Rha-C8-C10; Rha-C10-C8; Rha-C10-C10; Rha-C10-C12:1; Rha-C10-C12; Rha-C12-C10, where Rha=rhamnose moiety) are compared. The surface tensions found were 26.8 and 32.8 mN/m for RLAT10 and RL47T2, respectively. These two products differ in their antimicrobial properties, as based on their minimal inhibition concentrations (MIC). RLAT10 was effective against the fungal species (MIC) Aspergillus niger (16 μg/mL); Gliocadium virens (16 μg/mL); Penicillium chrysogenum (32 μg/mL); Botrytis cinerea (18 μg/mL); and Rhizoctonia solani (18 μg/mL), whereas RL47T2 was more effective against the bacteria (MIC) Enterobacter aerogenes (4 μg/mL); Serratia marcescens (8 μg/mL); Bacillus subtilis (16 μg/mL); and Staphylococcus aureus (32 μg/mL).
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- CID:
-
collison-induced dissociation
- ES:
-
electrospray
- FFA:
-
free fatty acids
- LC:
-
fleuid chromatography
- MIC:
-
minimal inhibition concentration
- MS:
-
mass spectrometry
- M.W.:
-
molecular weight
- Rha:
-
rhamnose moiety
- RL:
-
rhamnolipi
References
Linhardt, R.J., Microbially Produced Rhamnolipid as a Source of Rhamnose, Biotechnol. Bioeng. 33:365 (1989).
Parra, J., J. Pastor, F. Comelles, A. Manresa, and M. Bosch, Studies of Biosurfactants Obtained from Olive Oil, Tenside Surfactants Deterg. 27:302 (1990).
Olvera, C., J. Goldberg, R. Sánchez, and G. Soberón-Chávez, The Pseudomonas aeruginosa algC Gene Product Participates in Rhamnolipid Biosynthesis, FEMS Microbiol. Lett. 179:85 (1999).
Mercadé, M.E., A. Manresa, M. Robert, M.J. Espuny, C. de Andrés, and J. Guinea, Olive Oil Mill Effluent (OOME). New Substrate for Biosurfactant Production, Bioresource Technol. 43:1 (1993).
Schenk, T., I. Schuphan, and B. Schmidt, High-Performance Liquid Chromatographic Determination of the Rhamnolipids Produced by Pseudomonas aeruginosa, J. Chromatogr. A 693:7 (1994).
Déziel, E., F. Lépine, D. Dennie, D. Boismenu, O. Mamer, and R. Villemur, Liquid Chromatography/Mass Spectrometry Analysis of Mixtures of Rhamnolipids Produced by Pseudomonas aeruginosa Strain 57RP Grown on Mannitol or Naphthalene, Biochim. Biophys. Acta 1440:244 (1999).
Mata-Sandoval, J., J. Karns, and A. Torrents, High-Performance Liquid Chromatography Method for the Characterization of Rhamnolipid Mixtures Produced by Pseudomonas aeruginosa UG2 on Corn Oil, J. Chromatogr. 864:211 (1999).
Déziel, E., F. Lépine, S. Milot, and R. Villemur, Mass Spectrometry Monitoring of Rhamnolipids from a Growing Culture of Pseudomonas aeruginosa Strain 57RP, Biochim. Biophys. Acta 1485:145 (2000).
Lepine, F., E. Déziel, S. Milot, and R. Villemur, Liquid Chromatographic/Mass Spectrometric Detection of the 3-(3-Hydroxyalkanoyloxy) Alkanoic Acid Precursor of Rhamnolipids in Pseudomonas aeruginosa Cultures, J. Mass Spectrom. 37:41 (2002).
Woods, G.L., and J.A. Washington, Antibacterial Susceptibility Test: Dilution and Disk Diffusion Methods, in Manual of Clinical Microbiology, 6th edn. edited by P.R. Murray, ASM Press, Washington, DC, 1995, p. 1327.
Espinel-Ingroff, A., and M. Pfaller, M., Antifungal Agents and Susceptibility Testing, in Manual of Clinical Microbiology, 6th edn., edited by P.R. Murray, pp. 1405, ASM Press, Washington, DC, 1995, p. 1405.
Guerra-Santos, L., O. Käppeli, and A. Fiechter, Pseudomonas aeruginosa Biosurfactant Production in Continuous Culture with Glucose as Carbon Source. Appl. Environ. Microbiol. 48:301 (1984).
Manresa, A., J. Bastida, M.E. Mercadé, M. Robert, C. de Andrés, M.J. Espuny, and J. Guinea, Kinetic Studies on Surfactant Production by Pseudomonas aeruginosa 44T1, J. Ind. Microbiol. 8:133 (1991).
Desai, J., and I. Banat, Microbial Production of Surfactants and Their Commercial Potential, Microbiol. Mol. Biol. Rev. 61:47 (1997).
Maier, R., and G. Soberón-Chávez, Pseudomonas aeruginosa Rhamnolipids: Biosynthesis and Potential Applications, Appl. Microbiol. Biotechnol. 54:625 (2000).
Mata-Sandoval, J., J. Karns, and A. Torrens, Effect of Nutritional and Environmental Conditions on the Production and Composition of Rhamnolipids by P. aeruginosa UG2, Microbiol. Res. 155:249 (2001).
Rosen, M., Relationship of Structure Properties in Surfactants: II. Efficiency in Surfaces or Interfacial Tension Reduction, J. Am. Oil Chem. Soc. 51:461 (1974).
Lang, S., and F. Wagner, Bioconversion of Oils and Sugars to glycolipids, in Biosurfactants: Production, Properties, and Applications, edited by N. Kosaric, Marcel Dekker, New York, Surfactant Science Series Vol. 48, 1993, p. 251.
Kitamoto, D., H. Yanagishita, T. Shinbo, T. Nakane, C. Kamisawa, and T. Nakahara, Surface Active Properties and Antimicrobial Activities of Mannosylerythritol Lipids as Biosurfactants Produced by Candida antarctica, J. Biotechnol. 29:91 (1993).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Haba, E., Abalos, A., Jáuregui, O. et al. Use of liquid chromatography-mass spectroscopy for studying the composition and properties of rhamnolipids produced by different strains of Pseudomonas aeruginosa . J Surfact Deterg 6, 155–161 (2003). https://doi.org/10.1007/s11743-003-0260-7
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11743-003-0260-7