Abstract
Pericardial effusion (PE) is the presence of an excess of fluid in the pericardial cavity. PE symptoms depend from the rate of fluid accumulation, ranging from mild dyspnea on exertion to shock due to cardiac tamponade. Echocardiography is usually the primary diagnostic tool when PE is suspected, as it is accurate, non-invasive, widely available, and feasible also with pocket size devices. Studies have shown a high degree of sensitivity and specificity in the detection of PE using focused cardiac ultrasound (FOCUS), which can be performed also by non-cardiologist in emergency setting or at bedside. A PE is visualized as an echo-free space between the heart and the parietal layer of the pericardium. A semi-quantification of the PE may be obtained measuring the distance between the two pericardial layers. Once PE diagnosis has been made, characterization of fluid and search for signs of possible cardiac tamponade have to be performed. While unechogenic space is usually associated with serous fluid, hemorrhagic, and purulent effusions may be suspected in the presence of corpuscolated/echogenic fluid. Echocardiography may identify cardiac tamponade before it is clinically evident, and can guide pericardiocentesis. B-mode echocardiographic signs of cardiac tamponade include cardiac chambers collapse (with right chambers collapse occurring at earlier stages), opposite changes in right and left cardiac chamber filling during respiratory cycle, inferior vena cava and hepatic vein plethora. Doppler analysis of tricuspidalic and mitral flow velocities are used for a more detailed analysis of ventricular interdependence, even though more advanced operator expertise is required.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
When to perform bedside ultrasound (US) in suspected pericardial effusion
Pericardial effusion (PE) is the presence of an excess of fluid in the pericardial cavity, which is the virtual space between the layers of the pericardium. The pericardial space in physiologic conditions contains a small amount (<50 ml) of fluid allowing normal heart movement in the chest.
Leading causes of PE include pericarditis (idiopathic, infective, cancer related, autoimmune, uremic), injury to the pericardium or heart from a medical procedure (cardiac surgery or catheterization), myocardial infarction, hypothyroidism, trauma, aortic dissection.
PE collection symptoms range widely mainly in relation to the rate of fluid accumulation. Slow accumulation of large amount of PE are often asymptomatic while even a small quantity of fluid rapidly increasing in the pericardial space leads to intrapericardial pressure rise, thus interfering with normal cardiac filling and function, and producing cardiac tamponade. When PE is caused by pericarditis, the main symptom is chest pain, which can be accompanied by fever, fatigue, and myalgia. In other cases, PE may present with only mild dyspnea on exertion, thoracic discomfort, or even nonspecific symptoms. Signs and symptoms of cardiac tamponade include: hypotension, tachycardia, jugular-venous distension, muffled heart sounds, pulsus paradoxus (a drop of at least 10 mmHg in arterial blood pressure with inspiration), dyspnoea, confusion, or cardiac arrest.
How to perform bedside cardiac US
Echocardiography is usually the primary diagnostic tool when PE is suspected, as it is accurate, non-invasive, widely available, and easily utilized in emergency settings. Its high sensitivity allows the identification of the separation of pericardial layers when the PE exceeds just 15–35 ml. Bedside US performed with miniaturized devices, portable and even pocket size, has shown a good reproducibility in the detection of PE in respect to standard US machines.
Routinely, a phased-array sector transducer (3.5–5 MHz) is used for the evaluation of the pericardium, although rough information (presence/absence of fluid and evaluation of IVC dynamics) can also be obtained with a convex probe from a subcostal view. In the emergency setting, a subcostal scan obtained in the supine patient is usually used, but remember that ≥2 of the basic cardiac ultrasound views should be obtained to avoid missing a loculated fluid collection. The left lateral decubitus position is otherwise preferred, and all views are utilized to perform optimal visualization of the entire pericardial space and its contents.
What to look for: ultrasound signs of PE and cardiac tamponade
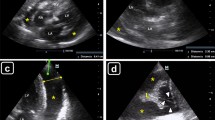
The pericardium appears at echocardiography as a hyperechoic layer surrounding the heart. A PE is visualized as an echo-free space between the heart and the parietal layer of the pericardium: a small PE accumulates mainly in the posterior atrioventricular groove, while a large PE usually surrounds the entire heart. A semi-quantification of the PE may be obtained measuring the distance between the two pericardial layers: small (echo-free space in diastole <10 mm, corresponding approximately to 300 ml); moderate (10–20 mm, corresponding to 500 ml); large (>20 mm, >700 ml) (Fig. 1) [1]. Once a PE diagnosis has been made, characterization of fluid and search for signs of possible cardiac tamponade have to be performed. While unechogenic space is usually associated with serous fluid, hemorrhagic and purulent effusions may be suspected in the presence of corpusculated/echogenic fluid. Hematomas and neoplastic diseases may be characterized by solid masses inside the pericardium (Fig. 1). Echocardiography may identify cardiac tamponade before it is clinically evident, and can guide pericardiocentesis. A pericardial tamponade is a clinical diagnosis that includes the visualization of a PE in addition to typical clinical signs. When intrapericardic pressure exceeds intracardiac pressure, cardiac chambers compression and reduced cardiac output occur. The intracavitary pressure is different in the four cardiac chambers, and it varies during the cardiac cycle. Because of its low instantaneous pressure, right atrium wall collapse during ventricular systole is the earliest echocardiographic finding of compression, followed by early diastolic collapse of the right ventricle. Moreover, respiratory changes in the cardiac chamber, and consequently in right and left cardiac output, are clearly seen in cardiac tamponade: inspiration results in an increase in venous return to the right heart that in case of significant pericardial fluid determines a significant simultaneous decrease in left heart filling. With expiration, opposite changes are observed (i.e., an increase in left heart filling and decrease in right heart filling). This pathologically enhanced ventricular interdependence: the echographic parallel of the ‘pulsus paradoxus,’ is also the cause of the characteristic respiratory movement of the so-called ‘swinging heart.’ Other echocardiographic signs of tamponade include inferior vena cava and hepatic vein plethora. The sensitivity and specificity of echographic signs of cardiac tamponade are reported in Table 1 [2].
Mild (a), moderate (b) and severe (c) pericardial effusion from a long axis parasternal view. Of note, the pericardial effusion spreads anterior to the descending aorta (bold arrow). In c, an intrapericardial mass (thin arrow) accompanies the pericardial effusion. d–f Echocardiographic signs of cardiac tamponade, with right atrial collapse (bold arrow), right ventricle collapse (thin arrow), and inferior vena cava plethora, respectively
Pulsed wave doppler analysis of tricuspid and mitral flow velocities are used for a more detailed analysis of ventricular interdependence, even though more sophisticated machines and more advanced operator expertise is required. While in a normal subject, inspiratory increase in Doppler velocities at the tricuspid and pulmonary valves and corresponding decrease in mitral and aortic flow velocities are less than 20 %, in cardiac tamponade mitral and aortic valve flows are reduced by more than 40 % with a corresponding increase observed in right-sided flow velocities.
Time or number of examinations necessary for an operator to learn how to recognize the ultrasound signs of PE
Although it is clear that extensive training and experience are needed to perform and interpret a complete echocardiographic study, a growing body of literature demonstrates that a non-cardiologist can be trained to acquire and interpret echocardiographic imaging in focused cardiac ultrasound (FOCUS) with an acceptable overall level of accuracy. Studies have shown a high degree of sensitivity and specificity in the detection of PE in both medical and trauma patients using FOCUS [3]. A prospective, observational study between emergency physician–performed bedside US and cardiologists shows that the former detects PE with a sensitivity of 96 %, specificity of 98 % and overall accuracy of 97.5 % [4]. In this study, all participating physicians took a 16-h course on emergency ultrasonography that included 1 h of instruction and 4 h of practical training dedicated to echocardiography.
Considering the relevant role of implementing the bedside approach to the patient with US, SIMI, as other scientific societies, has traced an educational program in bedside US [5].
Pitfalls
Anechoic or hypoechoic spaces adjacent to the heart (pleural effusions, ascites, pericardial cysts, pericardial fat, diaphragmatic herniae, left ventricular pseudoaneurysm) can be mistaken for PE. Visualization of the descending aorta in the parasternal long axis view may help in differentiating pericardial from pleural fluid: in the first case, the descending aorta remains posterior to the PE and anterior to pleural fluid (Fig. 1). Ascites collecting just below the diaphragm may simulate PE. However, in most cases, ascites can also be easily detected in other peritoneal spaces. The presence of pericardial fat may be mistaken for an organized PE: the former is normally distributed only in the anterior groove, has a typical echoic aspect, and is found mostly in aged or obese patients.
Collapse of the right heart chambers does not occur even with overt tamponade if the right heart pressures are elevated, such as in acute or chronic cor pulmonare, or in regional tamponade, where loculated pericardial effusions are sufficiently large to impact the filling of the adjacent chamber.
When not trust bedside US, but request a second level examination
Second level examinations (transesophageal echocardiography, computed tomography, and magnetic resonance) should be considered in selected cases of extremely difficult transthoracic visualization, or in loculated effusions. Conventional radiology studies can also help to determine the origin of suspected neoplastic PE and masses. Cardiac catheterization should be reserved for the patient with absent or only minimal PE, but in the clinical context of constrictive pericarditis.
References
Adler Y, Charron P, Imazio M et al (2015) ESC Guidelines for the diagnosis and management of pericardial diseases. Eur Heart J. doi:10.1093/eurheartj/ehv318
Guntheroth WG (2007) Sensitivity and specificity of echocardiographic evidence of tamponade: implications for ventricular interdependence and pulsus paradoxus. Pediatr Cardiol 28:358–362
Labovitz AJ, Noble VE, Bierig M (2010) Focused cardiac ultrasound in the emergent setting: a consensus statement of the American Society of Echocardiography and American College of Emergency Physicians. J Am Soc Echocardiogr 23:1225–1230
Mandavia DP, Hoffner RJ, Mahaney K, Henderson SO (2001) Bedside echocardiography by emergency physicians. Ann Emerg Med 38:377–382
Arienti V, Di Giulio R, Cogliati C, Accogli E, Aluigi L, Corazza GR (2014) Bedside ultrasonography (US), echoscopy and US point of care as a new kind of stethoscope for Internal Medicine Departments: the training program of the Italian Internal Medicine Society (SIMI). Intern Emerg Med 9:805–814
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Statement of human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
No informed consent.
Rights and permissions
About this article
Cite this article
Ceriani, E., Cogliati, C. Update on bedside ultrasound diagnosis of pericardial effusion. Intern Emerg Med 11, 477–480 (2016). https://doi.org/10.1007/s11739-015-1372-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-015-1372-8