Abstract
Cardiovascular (CV) disease is one of the most common causes of death in the western populations and, nowadays, its incidence is increasing even in the developing countries; although CV disease affects both sexes, it is more frequent in males in whom it shortens the average life expectancy. In this regard, this difference has been wrongly attributed for many years to the negative effects of testosterone (T); however, nowadays, a large amount of evidence suggests that this hormone may have protective effects on the CV system and that, indeed, the low levels of T could be associated with an increased CV risk and with an augmentation of morbidity and mortality in males. Such an aspect gains great relevance in light of the consideration that T decrease, besides occurring as a consequence of rare pathological conditions, can often take place with natural aging, causing a state of “male menopause”, also called late-onset hypogonadism. In this review, we aimed to summarize the present state of the art concerning the association between T deficit and CV disease by analyzing the protective role of T on CV system and the relationship of this hormonal lack with metabolic syndrome, CV morbidity and mortality, and with the CV complications, such as ischemic heart disease, heart failure and stroke, that frequently occur in T deficiency.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
For many years, the higher cardiovascular (CV) risk in men has been wrongly attributed to the negative systemic effects of testosterone (T); however, nowadays, a large amount of evidence suggests that, indeed, in males the low levels of T could be associated with morbidity and mortality. Also, the high CV mortality could be due to some physical and behavioral characteristics typical of male sex, such as the central distribution of fat adiposity or the smoking or alcohol consumption habit [1]. In any case T deficiency gains relevance because, besides occurring as a consequence of rare pathological conditions which are almost completely of endocrinological competence, can also take place with natural aging, causing a “male menopause” state, also defined as “late-onset hypogonadism” (LOH). This condition is generally associated with metabolic disturbances and increased CV risk, and often requires testosterone replacement therapy (TRT).
In this short review, we aimed to summarize the present state of the art concerning the association between T deficit and CV disease.
Hypogonadism
Male hypogonadism diagnosis requires unequivocally low levels of serum T (total T < 8 nmol/l and free T < 225 pmol/l) together with signs and symptoms consistent with hypogonadism (e.g., decreased libido, erectile dysfunction (ED), decrease of muscle mass or strength, increase of body fat, low bone mineral density or osteoporosis or decreased vitality) [2]. Free T calculation according to Vermeulen’s formula (at http://www.issam.ch/freetesto.htm) can be helpful in clinical states in which sex hormone-binding globulin might be decreased (e.g., obesity, acromegaly, andhypothyroidism) or increased (e.g., hepatic illness, hyperthyroidism, and use of anticonvulsants) [2]. As far as symptomatology is concerned, all the andrology society guidelines recognize that sexual symptoms are important in defining symptomatic hypogonadism and that the presence of at least one of the hypogonadism-related symptoms is an essential element to identify a clinically significant hypogonadism; among the relevant symptoms, hypoactive sexual desire is considered the one which is most commonly associated with male hypogonadism, while ED is the one that more frequently leads the patient to a medical consultation [2].
In cases of T deficiency, TRT is usually warranted when, in symptomatic men, circulating total T is below 8 nmol/l. When total T is repetitively between 8 and 12 nmol/l, in the presence of typical hypogonadal symptoms, a T treatment trial should be considered. Conversely, when total T is above 12 nmol/l, TRT should not be used [2].
Protective effects of T on CV system
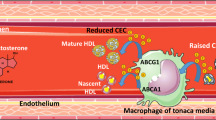
T seems to have several beneficial effects on CV system (Table 1). First of all, a large body of evidence suggests a direct vasodilator effect of this hormone on many vascular districts, including the coronaric one, in addition to a role in endothelium repair [1]. Second, studies carried out in hypogonadal subjects demonstrated that TRT is able to decrease the production of inflammatory cytokines, such as tumor necrosis factor-α, interleukin-1β and interleukin-6, and to increase the level of the antiaterogenic interleukin-10, while, in animal models of hypogonadism, it has been demonstrated that TRT can decrease the occurrence of typical atherosclerotic processes such as neointima formation, fatty streak accumulation and smooth muscle cells proliferation in the intima layer [1]. Third, in vitro studies and evidence collected in patients with low levels of T before and after TRT suggested that androgens could decrease arterial stiffness probably by inhibiting vascular smooth muscle calcification [1]. Finally, T seems to have a weak anticoagulant activity that could consist in stimulating tissue factor pathway inhibitor and tissue plasminogen activator expression and in inhibiting plasminogen activator inhibitor type 1 secretion by the endothelium [1].
Relationship between T and metabolic syndrome
A large amount of evidence suggests a connection between hypogonadism and metabolic syndrome. In this regard, in a recent meta-analysis about the cross-sectional studies evaluating the association between metabolic syndrome and hypogonadism, it has been reported that patients affected by metabolic syndrome showed significantly lower T plasma levels in comparison to age-matched subjects [3]; this relationship was independent from the ED and from the various diagnostic criteria of the metabolic syndrome. Link between hypogonadism and metabolic syndrome/CV complications could be bidirectional: in fact, while some studies suggest that in patients affected by prostate cancer (and, therefore, treated with androgens ablation) an increased prevalence of CV disease and metabolic syndrome occurs, other works show that several chronic diseases, including obesity, metabolic syndrome and type 2 diabetes, exert a suppressing effect on T [1]. The T decrease could entail, through the androgen receptor mainly expressed in visceral fat, a lack of negative regulation both of the differentiation of preadipocytes into mature adipocytes and of the differentiation of mesenchymal stem cells to adipocytes [2]. The increase of visceral fat, a well-known CV risk factor [4–6], could, in turn, provoke the worsening of hypogonadism through several mechanisms that may be represented by the augmentation of insulin-resistance, of adipose tissue aromatasic activity, of leptin or tumor necrosis factor-α secretion together with that of other adipokines [2]. As far as insulin-resistance is concerned, it has been demonstrated that, physiologically, insulin may act both at the hypothalamic and at the testicular level, increasing, in the first case, the synthesis and release of gonadotropin-releasing hormone and, in the second case, directly stimulating T secretion. These effects could be greatly blunted in central obesity, a condition characterized by insulin-resistance [2]. Furthermore, luteinizing hormone (LH) secretion could be inhibited by a mechanism of negative feedback exerted by the increased estrogenic synthesis that occurs as a consequence of the hyperexpression of P450 aromatase in adipose tissue [2]. It is also worth considering that leptin, an adipocyte-derived adipokine that circulates in plasma at concentrations that parallel the amount of fat reserves, has been demonstrated to contribute, in case of excess, to the development of hypogonadism in males [2]. Finally, obesity is also associated with increased circulating levels of tumor necrosis factor-α, a cytokine whose intratesticular augmentation has been associated with blunted T response to the human chorionic gonadotropin stimulation and with decrease of LH release in consequence of its activity at pituitary or hypothalamic level [2].
However, it cannot even be ruled out that reduction of T, as evident in visceral obesity and in several chronic diseases, could represent a protective mechanism in order to temporarily inhibit some of the T dependent functions like reproduction and physical labor that are not desirable when the general physical state is debilitated.
Relationship between T and CV events
Low levels of T have been found to be associated with different types of CV diseases including coronary artery disease (CAD), heart failure (HF) and stroke (Table 1).
Regarding CAD, Phillips et al. [7] have reported an inverse relationship between free T levels and the degree of CAD in 61 men. In addition English et al. [8] have found lower values of T in men with CAD compared with matched controls with normal coronary angiography. These findings have been further confirmed by Rosano et al. [9] who, evaluating the relationship between plasma sex hormone levels and presence/degree of CAD in patients undergoing coronary angiography and in matched controls, found that subjects with CAD had lower T and estradiol levels than healthy controls and that the levels of these hormones were inversely correlated to the degree of CAD.
Concerning HF, some authors reported that, in men affected by this pathology, total and free T are reduced proportionally to the HF severity and, in this condition, the prevalence of male hypogonadism is higher than 40 % [1]. It is also worth noting that low levels of T could play a role in favoring the onset of some peculiar features of the advanced HF like decreased exercise capacity, diminished muscle mass and energy handling, fatigue dyspnea and final cachexia [1].
Production of total T could be linked to the incidence or the severity of stroke or transient ischemic attack, to the 6-month mortality and to the infarct size [10, 11]. This hormonal fall could promote the thickening of carotid artery intima-media, the formation of abdominal aortic aneurism and, even, the onset of lone atrial fibrillation [1].
Furthermore, some epidemiological studies point out a relationship between T decrease and mortality [1]. In this regard, it is important to highlight that in the “Health in men” study, which analyzed 3,443 men older than 70 years, low total and free T concentrations were associated with reduced event-free survival and lower total T predicted increased incidence of stroke or transient ischemic attack after adjustment for many confounding factors like age, waist-hip ratio, waist circumference, smoking, hypertension, dyslipidemia, and medical comorbidity [11]. Furthermore, in a prospective study (European Prospective Investigation Into Cancer in Norfolk) examining the prospective relationship between endogenous T concentrations and mortality in 11,606 men, it has been reported that endogenous T concentrations at baseline were inversely related to mortality due to all causes, CV disease, and cancer [12]. In addition, in patients suffering from ED (a condition strongly associated not only with hypogonadism but even with CV risk as the smaller penile arteries reach critical narrowing with insufficient blood flow earlier than larger vessels) low levels of T have been found to be associated with higher mortality from major adverse CV events [13]. However, recent meta-analyses, although confirming the association between low levels of T with the increased risk of overall and CV mortality, did not find a relationship between low T and incident CV disease, suggesting, therefore, that T decrease could only represent a marker of poor general health [14, 15].
Effects of TRT on CV disease and metabolic syndrome
The beneficial effect of TRT on CV risk is a very controversial point. In this regard, although a recent meta-analysis on the effects of TRT on metabolic syndrome found that TRT was associated with a significant reduction of fasting plasma glucose, homeostatic model assessment index, triglycerides, waist circumference and with an increase of high-density lipoprotein cholesterol [3], three meta-analyses failed to find any differences between the group of subjects treated with T and the placebo group either regarding all CV events or a specific type of event [1]. However, Caminiti et al. [16] who analyzed the efficacy of TRT in patients affected by HF, found that TRT was able to improve exercise capacity, muscle strength and baro-reflex sensitivity. The issue remains undefined especially because the trials are often limited by the inadequate design or the insufficient statistical power.
Conclusions
Despite many, in vitro and in vivo, findings indicate that T plays a protective effect on CV system and that, therefore, its decrease could entail an increase of CV risk, several works did not support a relationship either between hypogonadism and CV disease or between TRT and CV events. However, low values of T seem to be associated with increased risk of CV death in community-dwelling men and in men with ED. Great uncertainty remains about the possibility that T deficiency could be the cause or the consequence of the increased risk of overall and CV lethality. Further and larger studies evaluating the impact of TRT on mortality are therefore needed.
Abbreviations
- T:
-
Testosterone
- CV:
-
Cardiovascular
- CAD:
-
Coronary artery disease
- HF:
-
Heart failure
References
Corona G, Rastrelli G, Vignozzi L, Mannucci E, Maggi M (2011) Testosterone, cardiovascular disease and the metabolic syndrome. Best Pract Res Clin Endocrinol Metab 25:337–353. doi:10.1016/j.beem.2010.07.002
Corona G, Rastrelli G, Morelli A, Vignozzi L, Mannucci E, Maggi M (2011) Hypogonadism and metabolic syndrome. J Endocrinol Invest 34:557–567. doi:10.3275/7806
Corona G, Monami M, Rastrelli G, Aversa A, Tishova Y, Saad F, Lenzi A, Forti G, Mannucci E, Maggi M (2011) Testosterone and metabolic syndrome: a meta-analysis study. J Sex Med 8:272–283. doi:10.1111/j.1743-6109.2010.01991.x
Zizzi A, Tirabassi G, Aspriello SD, Piemontese M, Rubini C, Lucarini G (2012) Gingival advanced glycation end-products in diabetes mellitus-associated chronic periodontitis: an immunohistochemical study. J Periodontal Res. doi:10.1111/jre.12007
Marcellini F, Giuli C, Papa R, Tirabassi G, Faloia E, Boscaro M, Polito A, Ciarapica D, Zaccaria M, Mocchegiani E (2009) Obesity and body mass index (BMI) in relation to life-style and psycho-social aspects. Arch Gerontol Geriatr 49(Suppl 1):195–206. doi:10.1016/j.archger.2009.09.029
Faloia E, Tirabassi G, Canibus P, Boscaro M (2009) Protective effect of leg fat against cardiovascular risk factors in obese premenopausal women. Nutr Metab Cardiovasc Dis 19:39–44. doi:10.1016/j.numecd.2008.02.004
Phillips GB, Castelli WP, Abbott RD, McNamara PM (1983) Association of hyperestrogenemia and coronary heart disease in men in the Framingham cohort. Am J Med 74:863–869
English KM, Mandour O, Steeds RP, Diver MJ, Jones TH, Channer KS (2000) Men with coronary artery disease have lower levels of androgens than men with normal coronary angiograms. Eur Heart J 21:890–894
Rosano GM, Sheiban I, Massaro R, Pagnotta P, Marazzi G, Vitale C, Mercuro G, Volterrani M, Aversa A, Fini M (2007) Low testosterone levels are associated with coronary artery disease in male patients with angina. Int J Impot Res 19:176–182
Jeppesen LL, Jørgensen HS, Nakayama H, Raaschou HO, Olsen TS, Winther K (1996) Decreased serum testosterone in men with acute ischemic stroke. Arterioscler Thromb Vasc Biol 16:749–754
Yeap BB, Hyde Z, Almeida OP, Norman PE, Chubb SA, Jamrozik K, Flicker L, Hankey GJ (2009) Lower testosterone levels predict incident stroke and transient ischemic attack in older men. J Clin Endocrinol Metab 94:2353–2359. doi:10.1210/jc.2008-2416
Khaw KT, Dowsett M, Folkerd E, Bingham S, Wareham N, Luben R, Welch A, Day N (2007) Endogenous testosterone and mortality due to all causes, cardiovascular disease, and cancer in men: European prospective investigation into cancer in Norfolk (EPIC-Norfolk) prospective population study. Circulation 116:2694–2701
Corona G, Monami M, Boddi V, Cameron-Smith M, Fisher AD, de Vita G, Melani C, Balzi D, Sforza A, Forti G, Mannucci E, Maggi M (2010) Low testosterone is associated with an increased risk of MACE lethality in subjects with erectile dysfunction. J Sex Med. 7:1557–1564. doi:10.1111/j.1743-6109.2009.01690.x
Corona G, Rastrelli G, Monami M, Guay A, Buvat J, Sforza A, Forti G, Mannucci E, Maggi M (2011) Hypogonadism as a risk factor for cardiovascular mortality in men: a meta-analytic study. Eur J Endocrinol 165:687–701. doi:10.1530/EJE-11-0447
Araujo AB, Dixon JM, Suarez EA, Murad MH, Guey LT, Wittert GA (2011) Clinical review: endogenous testosterone and mortality in men: a systematic review and meta-analysis. J Clin Endocrinol Metab 96:3007–3019. doi:10.1210/jc.2011-1137
Caminiti G, Volterrani M, Iellamo F, Marazzi G, Massaro R, Miceli M, Mammi C, Piepoli M, Fini M, Rosano GM (2009) Effect of long-acting testosterone treatment on functional exercise capacity, skeletal muscle performance, insulin resistance, and baroreflex sensitivity in elderly patients with chronic heart failure a double-blind, placebo-controlled, randomized study. J Am Coll Cardiol 54:919–927. doi:10.1016/j.jacc.2009.04.078
Webb CM, Adamson DL, de Zeigler D, Collins P (1999) Effect of acute testosterone on myocardial ischemia in men with coronary artery disease. Am J Cardiol 83:437–439
Deenadayalu VP, White RE, Stallone JN, Gao X, Garcia AJ (2001) Testosterone relaxes coronary arteries by opening the large-conductance, calcium-activated potassium channel. Am J Physiol Heart Circ Physiol 281:1720–1727
Jones RD, English KM, Pugh PJ, Morice AH, Jones TH, Channer KS (2002) Pulmonary vasodilatory action of testosterone: evidence of a calcium antagonistic action. J Cardiovasc Pharmacol 39:814–823
Corrales JJ, Almeida M, Burgo R, Mories MT, Miralles JM, Orfao A (2006) Androgen-replacement therapy depresses the ex vivo production of inflammatory cytokines by circulating antigen-presenting cells in aging type-2 diabetic men with partial androgen deficiency. J Endocrinol 189:595–604
Nettleship JE, Jones TH, Channer KS, Jones RD (2007) Physiological testosterone replacement therapy attenuates fatty streak formation and improves high-density lipoprotein cholesterol in the Tfm mouse: an effect that is independent of the classic androgen receptor. Circulation 116:2427–2434
Tharp DL, Masseau I, Ivey J, Ganjam VK, Bowles DK (2009) Endogenous testosterone attenuates neointima formation after moderate coronary balloon injury in male swine. Cardiovasc Res 82:152–160. doi:10.1093/cvr/cvp038
Maddali KK, Korzick DH, Tharp DL, Bowles DK (2005) PKCdelta mediates testosterone-induced increases in coronary smooth muscle Cav1.2. J Biol Chem 280:43024–43029
Jin H, Lin J, Fu L, Mei YF, Peng G, Tan X, Wang DM, Wang W, Li YG (2007) Physiological testosterone stimulates tissue plasminogen activator and tissue factor pathway inhibitor and inhibits plasminogen activator inhibitor type 1 release in endothelial cells. Biochem Cell Biol 85:246–251
Buvat J, Maggi M, Gooren L, Guay AT, Kaufman J, Morgentaler A, Schulman C, Tan HM, Torres LO, Yassin A, Zitzmann M (2010) Endocrine aspects of male sexual dysfunctions. J Sex Med 7:1627–1656. doi:10.1111/j.1743-6109.2010.01780.x
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tirabassi, G., Gioia, A., Giovannini, L. et al. Testosterone and cardiovascular risk. Intern Emerg Med 8 (Suppl 1), 65–69 (2013). https://doi.org/10.1007/s11739-013-0914-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-013-0914-1