Abstract
Acer buergerianum Miq. (Trident maple) is a native species of China with a large distribution, but exist in small population. Water and light are two important factors limiting plant growth and are crucial in the framework of forest regeneration. However, there is no consensus on how shade interacts with drought. Four hypotheses in the recent literature variously predict that shade will have a stronger, weaker or equal impact on seedlings under drought stress. This study investigated the interactive responses of A. buergerianum to light and water focusing on seedling growth, leaf morphology and biomass partitioning by performing a growth experiment in pots with different water supply regimes [15, 35, 55, 75, 95 % of field capacity (FC)] combined with two light regimes (10 and 66 % of full sunlight). After 123 days treatment, the results showed that shade greatly reduced growth and biomass, in contrast enhancing the amount of chlorophyll, the amount of water in the leaves, and the specific leaf area. Drought reduced growth, biomass, and the bulk of the leaves. Most leaf traits and biomass characteristics had strong interactions in their responses to light and water treatments. Allometric analysis revealed that water and light had no effects on root to shoot ratios, main root to lateral root ratios, and root mass ratios. Shade alleviated the negative impact of drought. A. buergerianum successfully adapted to the various light and water conditions. We recommend a water supply above 15 % FC to keep the seedlings vigorous, under both sunlight conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Light is one of the most important ecological factors in plant ecophysiology due to its essential function in autotrophic plants (Humbert et al. 2007). However, the quantity of light required by plants in a specific environment, particularly in their natural habitats varies both spatially and temporarily (Björkman 1981). In addition, water limitation is one of the most general types of stress experienced by plants and is a main factor limiting plant growth (Guo et al. 2007).
The interactive effects of drought and shade on woody seedling have been reported in many previous studies (Holmgren 2000; Quero et al. 2006; Aranda et al. 2005), but no consensus has been reached. The trade-off theory (Smith and Humston 1989) is the most influential (Yang et al. 2008) among the several hypotheses suggested. This theory predicts that shade aggravates the drought stress, and plants in the shade invest more to produce shoots and leaves rather than roots, increasing irradiation capture at the expense of the amount of water obtained.
In contrast to the trade-off theory, the above-ground facilitation hypothesis illustrates that shade is a shelter for the plants from high leaf and air temperatures, high vapour pressure deficits and oxidation, which may aggravate the stress caused by drought. Thus, shade can reduce the impact of drought (Holmgren 2000).
According to the interplay hypothesis, the impact of drought is relatively strong at high irradiance, weaker in moderate shade (because of facilitation), and strong in deep shade (Holmgren et al. 1997). Another hypothesis, the independent effects model put forward by Nobel (1999), predicts the impacts of shade and drought are orthogonal, i.e., dry soil should have a proportional impact across illuminations. The theory has been triumphantly validated in cacti, agaves (Nobel 1999, 1984; Nobel and Hartsoek 1986) and woody seedlings (Sack and Grubb 2002).
Acer buergerianum Miq. (Trident maple) is a native deciduous species covering a large area of China and commonly cultivated in Japan, Korea and the USA. Trident maple is a pioneer species and can grow quickly throughout the whole growth season. Besides distributing in forests, this species is broadly cultivated as landscape plant in urban areas, such as parks, gardens, two sides of the roads, etc. In conclusion, it is important for its ecological and ornamental function. However, trident maple trees are always interspersing among other trees in forests and only rarely can forests of trident maples be found today in China. Moreover, trident maples produce a large number of seeds every year when reaching maturity, indicating its low recruitment success ratio in living environments.
The broad distribution means trident maple trees are likely to be exposed to diverse situations with different levels of irradiation and moisture, which may be the main factors limiting trident maple regeneration and recruitment. The effects of different levels of light and water on the growth of some species of the Aceraceae family treated have been reported (Lei and Lechowicz 1998; Adams et al. 1990; Hanba et al. 2002). However, trident maple trees were often excluded in these studies, and the interaction of light and water was not included. Whether trident maple trees are drought tolerant or shade tolerant is unclear. We hypothesize that the small numbers of this species is at least partly due to its intolerance to drought, as there is little rainfall during the spring in east and north China, which is the distribution area of trident maple trees.
In this study, the morphological and biomass characteristics of trident maple trees under different levels of light and water were measured to determine (1) whether trident maples are intolerant to drought or shade, (2) which of the four hypotheses explains the responses of trident maples to the interactions of shade and drought, (3) whether drought or lack of light is responsible for the low numbers and provide suggestions for its regeneration.
Materials and methods
Study site and plant materials
The study was conducted at Fanggan Research Station of Shandong University in Laiwu, Shandong Province, China (36°26′N, 117°27′E). The site has a warm temperate monsoon climate, with an average temperature of 13 ± 1 °C, and an average annual precipitation of 700 ± 100 mm, most of which falls during the summer (Zhang et al. 2006). The soil type is yellow cinnamon soil with limestone as parent material (Zhang et al. 2006). The whole experiment was carried out in the greenhouse of the station to maintain a controlled environment. The greenhouse was well ventilated by rolling the plastic side films.
The seeds were purchased from Dacheng Seed Company (Jinan, China), who collected the seeds in its own garden in early winter of 2009. The seeds were stored at 0–4 °C through the winter. Then, the seeds were soaked in water for 24 h and preserved in wet sand at 0–4 °C to stimulate germination in March. The germinated seedlings were transferred into plastic pots (320 mm height × 290 mm diameter) with one seedling per pot. Each pot was loaded with a mixture of 2:1 (v/v) of sandy loam and humic soils. The total weight of the substrate was 6.5 kg.
Experimental design
Two light levels (10 and 66 % of full sunlight, i.e., L1, L2), and a broad range of water regimes from 15 to 95 % of field capacity (FC) were set to simulate the heterogeneous conditions under which trident maples may occur. The light intensity was measured with a micro-quantum sensor (Xu et al. 2009). The FC of the soil was 1.833 kg ± 0.066 (n = 6). Water regimes were applied to simulate the different moisture condition. The two light levels represented the illumination status of understorey and moderate forest gap in northern China. The 10 % (L1) light level was achieved with shade shelters covered with plastic film and the 66 % (L2) level was produced with woven black nylon nets.
At each light level, we arranged five water supply regimes: 15, 35, 55, 75, and 95 % FC (designated W1, W2, W3, W4, and W5). Thus, the experiment was arranged in a completely randomized design with two factors. Seven pots were included for each treatment.
Throughout the experiments, the pots received compensatory irrigation by weighing daily at 17:00 to maintain a constant soil moisture level. Pots were randomly arranged in the glasshouse and re-randomized at regular intervals throughout the experiment. No further fertilization was added to the pot through the experiment. The experiment was carried out from 8 June to 18 October, which is the primary growth period for trident maples in a year and lasted for 123 days.
Measurements
Seedling height was recorded approximately every 15 days from the time the light and water treatments were started. The number of leaves, seedling diameter at ground height (about 0.5 cm above the ground line) and crown area were recorded at the end of the treatment. The crown area was calculated as: crown area = 0.5ab (a and b were the length of diagonal).
Leaf morphology was measured at the end of August. Ten fully expanded leaves (fifth or/and sixth leaves from the tip) per treatment were scanned with a scanner (Epson Perfection V700, Seiko Epson, Japan). Leaf area and leaf perimeter were calculated with an image analyzer (Image-Pro Plus Version 4.5, Media Cybernetic, Inc., Silver Spring, MD, USA) using the scanned images. Detailed information on the morphological measurements is shown in Table 1. The leaves were weighted before and after oven drying at 80 °C for 48 h to calculate the water content of the leaves (LWC) and specific leaf area (SLA).
Leaf elongation was calculated as: leaf elongation = leaf length/leaf width
Chlorophylls a and b were extracted and measured in the middle of August according to the method by (Lichtenthaler and Wellburn 1983), using a spectrophotometer (722S visible light spectrophotometer, Leng Guang, Inc., Shanghai, China). Seven fully expanded leaves (the fifth or sixth from the tip) were sampled for chlorophyll measurement.
All plants from each treatment were harvested at the end of the experiment, which is after 123 days of treatment. The whole plant was divided into five parts: main root, lateral roots, stem, leaf blades and petioles. The main root was defined as the root that developed directly from the seed, while the lateral roots were the roots that extended from the main root.
The weight of every part was quantified after oven drying at 80 °C for 48 h. The weight of the leaves collected for leaf morphological measurements was added to the final biomass. Fallen leaves were collected every 3 or 4 days beginning September 20. The number of fallen leaves was added to the growth parameters, and the dry mass was added to the biomass of the leaves.
Total biomass, root mass ratios, stem mass ratios and leaf mass ratios were calculated as follows:
total biomass = main root biomass + lateral root biomass + stem biomass + leaf blades biomass + petioles biomass;
root mass ratio = (main root biomass + lateral root biomass)/total biomass.
stem mass ratio = stem biomass/total biomass;
leaf mass ratio = (leaf blades biomass + petioles biomass)/total biomass.
Statistical analysis
Two-way analysis of variance (ANOVA) was used to compare the effects of light and water stress and one-way ANOVA was applied to detect the differences among the 10 treatments for both parameters. All two-way ANOVAs and one-way ANOVAs were accompanied with Duncan’s multiple comparison tests at p ≤ 0.05.
Before ANOVAs, data were checked for the normality and homogeneity of variance, and were log-transformed when necessary. All of the statistical analyses were performed using the SPSS 13.0 software package (SPSS Inc., IL, USA). Figures were drawn using Origin 8.0 software (OriginLab Co., Massachusetts, USA).
Allometric analysis was conducted to investigate the relationships between shoot biomass and root biomass, leaf biomass and total biomass, stem biomass and total biomass, root biomass and total biomass and main root biomass and lateral root biomass, with SMATR 2.0 using a standardized major axis (SMA) method (Warton et al. 2006; Falster et al. 2006).
Results
There was no seedling death recorded during the experiment. All growth parameters exhibited significant differences among light, water and their interactions by two-way ANOVAs (Table 1). Height changes and growth parameters at the end of the experimental period for Acer buergerianum seedlings are shown in Figs. 1 and 2, respectively. All parameters initially increased and then gradually decreased with the increase in humidity with L2 treatment. However, no significant difference was found between W4 and W5 under L2 conditions. With regard to L1 treatment, four parameters increased as moisture increased. The diameter at ground height was significantly higher with the L2 treatment as compared to the L1 treatment at every level of water treatment.
Comparisons of height (a), diameter at ground height (b), number of leaves (c) and crown (d) of A. buergerianum seedlings under different light and water treatments at the end of the experimental period. The data are mean ± SE (n = 7). Different letters indicate significant differences (p ≤ 0.05) with Duncan’s multiple range test
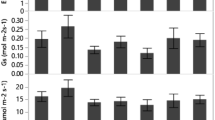
Significant differences were found in leaf morphological features among various light and water treatments by one-way ANOVAs (Fig. 3). Almost all leaf traits exhibited significant differences among light, water and interaction by two-way ANOVAs (Table 1). Only leaf elongation was unaffected by water treatment, light treatment and light and water interaction. SLA was only influenced by light treatment. SLA and LWC were higher at every water level under L1 conditions than under L2 conditions, although the differences were not significant for LWC. Leaf length, leaf width and perimeter were increased first as humidity increased, but descended at the W5 treatment. Interestingly, under W1 conditions, leaf length, leaf width, perimeter and area were lower under the L2 treatment than under the L1 treatment, which was the opposite of the other water conditions. Leaf length to petiole length was affected by humidity rather than illumination and the interaction was significant.
Comparisons of leaf morphological traits variables of A. buergerianum seedlings under different light and water treatments. The data are mean ± SE (n = 10). The leaf morphological traits include length (a), width (b), length elongation (c), area (d), perimeter (e), LL/PL (f), LAR (g), SLA (h) and LWC (i). Different letters indicate significant differences (p ≤ 0.05) with Duncan’s multiple range test. LL/PL leaf length to petiole length, LAR total leaf area to total dry biomass, SLA specific leaf area, LWC leaf water content
The F values and their significance with regard to the amounts of chlorophyll by two-way ANOVAs are listed in Table 1, while the significance using one-way ANOVAs are shown in Fig. 4. The amounts of chlorophyll a, chlorophyll b and total chlorophyll were higher under L1 conditions than L2 conditions. Under low irradiation, no water treatment exhibited any difference in terms of chlorophyll a, chlorophyll b and total chlorophyll. Under high irradiation, the W1 treatment had the lowest chlorophyll levels. For the chlorophyll a to chlorophyll b ratios, the L2 treatment was higher than L1 at every moisture level except W1.
Comparisons of the amounts of chlorophyll a (a), chlorophyll b (b), the ratio of chlorophyll a to chlorophyll b ratio (c), the total amount of chlorophyll (d) from A. buergerianum seedlings under different light and water treatments at the end of the experimental period. The data are mean ± SE (n = 7). Different letters indicate significant differences (p ≤ 0.05) with Duncan’s multiple range test
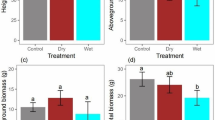
The F values and their significance with regard to biomass production and partitioning by two-way ANOVAs are listed in Table 1. Except for the main root to lateral root ratios, all the other parameters were significantly different under various light and water stress conditions. The light and water interaction was significant for all parameters except for the root to shoot ratios, main root to lateral root ratios and stem mass ratios. Biomass production parameters are shown in Fig. 5. In the L2 treatment, all parameters decreased proportionately with the severity of drought. It should be noted that the W5 treatment was smaller than the W4 treatment, although only the main root parameter was significantly decreased. In the L1 treatment, the tendency was similar, but the decrease was not as large as L2.
Comparisons of total biomass (a), root mass (b), leaf mass (c), stem mass (d) of A. buergerianum seedlings under different light and water treatments at the end of the experimental period. The data are mean ± SE (n = 7). Different letters indicate significant differences (p ≤ 0.05) with Duncan’s multiple range test
Regression models between different biomass parameters are shown in Table 2. p values did not reach the critical level (0.05) indicating there was no evidence that the group slopes were heterogeneous among the ten treatments. Thus, root to shoot ratios, main root to lateral root ratios, and root mass ratios were not affected by water and light conditions. The significance of root to shoot ratios, main root to lateral root ratios, and root mass ratios between different treatments with two ANOVA was due to allometric growth rather than plasticity.
Regression models between shoot biomass and root biomass under two conditions are shown in Table 3. The p values for both light conditions did not reach the critical point, indicating water conditions had no effects on root to shoot ratios.
Sorted pairwise significance matrices for stem biomass and total biomass regression and leaf biomass and total biomass regression are shown in Tables 4, 5, 6, 7, respectively. The slopes for the stem biomass and total biomass regression model were higher in the shade than in high light under all water conditions except the W5 water treatment. The slopes for the leaf biomass and total biomass regression model were higher in the shade than under high light with drought conditions (W1, W2), and the tendency reversed under well-watered conditions (W3, W4, and W5).
Discussion
It was demonstrated in previous research that simulated experiments can achieve similar effects to those in wild habitats (Xu et al. 2008). Based on the greenhouse results, trident maples were able to adapt to diverse light and water conditions, even at the lowest illumination and extreme aridity. Trident maples are relatively a drought- and shade-tolerant species.
The heights of the seedlings approximately did not change during the last 2 weeks, indicating the end of the growth season. Shade and drought inhibited the growth of trident maple seedlings, and the interaction between light and water was significant. As has been reported, shoot height, crown area, diameter at ground height and leaf area would be inhibited by drought (Du et al. 2010) and shade lowered leaf length, leaf width leaf area (Xu et al. 2008), diameter at ground height and number of leaves (Jiménez et al. 2009). However, different from our results, shoot height increased in shade and number of leaves did not influenced by humidity for cork oak (Jiménez et al. 2009), indicating the different sensitivity among species.
Adjusting the leaf characteristics was suggested to be the most efficient way to acclimate and forage for light (Rozendaal et al. 2006). Leaf length, leaf width, perimeter and area were restricted by drought and shade, however as reported, leaf elongation was unaffected. The trident maple changed the size of its leaves to acclimatize to the stress, but did not change the shape properties.
The ratio of leaf length to petiole length ratio reflected the architectural balance between lamina and petiole (Noda et al. 2004). The decrease in leaf length to petiole length ratio along the increasing moisture in L2 leaves in our study indicated that the resistance of water flow from roots to lamina under water stress was reduced (Noda et al. 2004). Leaf length to petiole length ratio decreased when soil humidity increased in L2 condition, while did not change in shade. Furthermore, under L1 conditions, the leaf area was remarkably smaller at the W5 level than the W4 level, indicating the W5 level may have inhibited the plant and the W4 level may be the optimal humidity for low illumination. Under L2 irradiation, neither parameter presented such changes. Thus, irradiation had an effect on moisture conditions. Leaf area ratio (m2 g−1), calculated as photosynthetic surface to total respiring plant biomass (Smith and Humston 1989), indicated the balance between photosynthesis and respiration, and could determine C accumulation (μmol g−1 day−1) by multiplying photosynthetic rate (μmol m−2 day−1) (Holmgren 2000). Leaf area ratio of shade leaf decreased when soil humidity increased, however, that of well-illuminated leaf did not change along the water regime, also indicating the remarkable shade effect on drought. Nevertheless, it is not the case in the former study (Holmgren 2000), where leaf area ratio decreased in drought status, regardless of the illumination conditions.
Among the leaf morphological traits, SLA was one of the major contributors to an axis of resource capture, usage and availability (Grime et al. 1997). In the present study, SLA was notably affected by illumination status, but not humidity. Trident maples tended to use less investment by thinning their shaded leaves to capture more irradiation and thicken the leaves in high light to resist overheating, similar to results in previous study (Sack et al. 2003) on understory plants in Spain. The SLA of cork oak was modified by light availability rather than water conditions (Jiménez et al. 2009), which is comparable to results in our study. Changes in SLA with light availability, but not with water conditions, might be interpreted as a homeostatic mechanism to prioritize the optimization of light capture (Aranda et al. 2005; Lambers and Poorter 1992).
Seedlings grown in low light significantly increased their chlorophyll content on a mass basis, as exhibited in previous studies (Aranda et al. 2005; Jiménez et al. 2009). Decreases in chlorophyll b content have been suggested to be an indication of chlorophyll destruction by excess irradiation (Jason et al. 2004), and the marked increase after shade treatment demonstrated the plant’s ability to maximize the light-harvesting capacity under low light conditions (Lei et al. 1996). The chlorophyll a/b ratios were lower for 11 species of maple saplings under low light regimes, including trident maples (Lei and Lechowicz 1998), in accordance with that, the greater investment expectation in chlorophyll b would enhance PSII function in low light environments (Lei et al. 1996; Lei and Lechowicz 1998).
As previous studies (Du et al. 2010; Holmgren 2000) demonstrated, both drought and shade exceedingly reduced the production of seedlings. When resources are limiting, plants usually adapt by allocating more photosynthates to produce organs that are able to acquire the resources (Wang et al. 2008). Here, we certainly found that the seedlings invested more in leaves under shade and drought conditions, but this was not the case under well-watered conditions. The slopes of leaf biomass and total biomass regressions were higher under high illumination in well-watered conditions. This did not agree with the previous results that described a high allocation to above-ground under shade conditions (Poorter and Nagel 2000) and unchanged leaf mass ratios under low light (Navas and Garnier 2002). Furthermore, the unchanged root to shoot ratios among various moisture conditions also indicated seedlings did not allocate more resources to underground organs that are proficient to acquire more water.
However, leaves as a water-losing organ, their area became smaller under drought stress. Thus, leaves with small size and high SLA can contribute to drought tolerance and shade conditions simultaneously. Actually, the approximately LWC among various moisture conditions except the W1 level demonstrated the leaves did sustain a well-hydrated situation. Drought levels as low as 15 % FC led to remarkable injuries of the photosynthesis apparatus to the leaves, indicating that, under this moisture condition, A. buergerianum could not adjust its plasticity to maintain the leaves in their natural states.
Most leaf traits, biomass and partitioning characteristics showed strong interactions in their responses to light and water treatments; hence, their variation was not according to the independent-effects model. Under W1 conditions, no biomass parameters showed any significant differences between L1 and L2. However, all biomass parameters in L2 were several-fold higher than L1 under other moisture conditions. As the moisture was enhanced, the distinction between L2 and L1 became larger, and peaked at the W4 treatment level, becoming smaller thereafter. Therefore, our results failed to support the trade-off model (Smith and Humston 1989) as drought alleviated shading effects. According to the above-ground facilitation hypothesis, shading effects decreased under dry conditions and moisture effects decreased under shade conditions. Thus, our results are in agreement with the above-ground facilitation hypothesis. Allometric analysis showed that drought had no effects on root to shoot ratios, despite light conditions. This is in accordance with the above-ground facilitation hypothesis (Holmgren 2000), but not with the trade-off model (Smith and Humston 1989) and some other research (Achten et al. 2010).
Noticeably, total leaf area to total biomass ratios decreased in more severe drought conditions under low irradiation; however, water had no significant effects on the total leaf area to total biomass ratios under high irradiation. This did not agree with the trade-off model (Smith and Humston 1989) nor the above-ground facilitation (Holmgren 2000) hypothesis since the two models expect the total leaf area to total biomass ratios to decrease under drought conditions for shaded plants. Our results definitively demonstrated that shade and drought effects had positive interactions.
The presented results could not be arbitrarily classified as supporting the interplay hypothesis (Holmgren et al. 1997), since the two light regimes (10 and 66 % of full sunlight) we applied were not enough to recognize which was high illumination, moderate shade or deep shade, nor could it be shown how much the light intensity should be regarded as the criterion to judge the three different degrees for trident maples.
As a native species of China, A. buergerianum has a large distribution throughout the world. However, the quantity of trees of this species in China is much fewer than expected. The present study showed that the seedlings can tolerate water stress through morphological and physiological responses even when facing extreme drought stress. Thus, neither drought nor shade conditions can decrease numbers of this species.
Drought and shade had a positive interaction to the seedlings. The effects of drought or shade cannot be considered solely without also considering their interaction. The seedlings under W1 conditions had almost no increase in height through the 123 days, indicates the seedlings had little or no growth after a whole growth season. Thus, approximately 15 % FC is likely the critical point for keeping A. buergerianum seedlings alive under pot conditions, which excludes the stress of competition and insect pests. A critical point may increase with competition from other vegetation, when considering natural regeneration of maples in the field. Thus, water supplies above 15 % FC are recommended to keep seedlings vigorous, under all sunlight conditions. In addition, as compared to gap conditions (66 % of full sunlight), A. buergerianum seedlings grow slowly in the understory (10 % of full sunlight). Regulation of canopy density, e.g., release cutting and thinning and control of competitors, especially tall rapidly growing weeds, would enhance seedlings growth and promote regeneration.
Based on the above-ground facilitation hypothesis, plants under dry conditions and high irradiance had lower daily photosynthetic rates than plants under low irradiance, and the whole-shoot light compensation point tended to decrease under more severe drought conditions (Holmgren 2000). Further measurements including daily photosynthetic rates and the whole-shoot light compensation point are needed to confirm our conclusions.
Author contribution
Xiao Guo, Weihua Guo and Renqing Wang were responsible for the design of the experiment and manuscript preparation. Xiao Guo, Yujie Luo and Xiangfeng Tan performed the experiment. Xiao Guo, Weihua Guo and Ning Du analyzed the data. Weihua Guo and Renqing Wang revised the manuscript as well.
References
Achten WMJ, Maes WH, Reubens B, Mathijs E, Singh VP, Verchot L, Muys B (2010) Biomass production and allocation in Jatropha curcas L. seedlings under different levels of drought stress. Biomass and Bioenergy 34(5):667–676
Adams W, Demmig-Adams B, Winter K, Schreiber U (1990) The ratio of variable to maximum chlorophyll fluorescence from photosystem II, measured in leaves at ambient temperature and at 77 K, as an indicator of the photon yield of photosynthesis. Planta 180:166–174
Aranda I, Castro L, Pardos M, Gil L, Pardos J (2005) Effects of the interaction between drought and shade on water relations, gas exchange and morphological traits in cork oak (L.) seedlings. For Ecol Manage 210(1–3):117–129
Björkman O (1981) Responses to different quantum flux densities. In: Lange OL, Nobel PS, Osmond CB, Zeigler H (eds) Encyclopedia of Plant Physiology, New Series, Springer, Berlin, 12A: pp 57–107
Du N, Guo WH, Zhang XR, Wang RQ (2010) Morphological and physiological responses of Vitex negundo L. var. heterophylla (Franch.) rehd. to drought stress. Acta Physiol Plant 32(5):839–848
Falster DS, Warton DI, Wright IJ (2006) User’s guide to SMATR: standardized major axis tests and routines, version 2.0. http://wwwbiomqeduau/ecology/SMATR/SMATR_users_guidepdf
Grime JP, Thompson K, Hunt R, Hodgson JG, Cornelissen JHC, Rorison IH, Hendry GAF, Ashenden TW, Askew AP, Band SR, Booth RE, Bossard CC, Campbell BD, Cooper JEL, Davison AW, Gupta PL, Hall W, Hand DW, Hannah MA, Hillier SH, Hodkinson DJ, Jalili A, Liu Z, Mackey JML, Matthews N, Mowforth MA, Neal AM, Reader RJ, Reiling K, Ross-Fraser W, Spencer RE, Sutton F, Tasker DE, Thorpe PC, Whitehouse J (1997) Integrated screening validates primary axes of specialization in plants. Oikos 79:259–281
Guo W, Li B, Zhang X, Wang R (2007) Architectural plasticity and growth responses of Hippophae rhamnoides and Caragana intermedia seedlings to simulated water stress. J Arid Environ 69(3):385–399
Hanba YT, Kogami H, Terashima I (2002) The effect of growth irradiance on leaf anatomy and photosynthesis in Acer species differing in light demand. Plant Cell Environ 25(8):1021–1030
Holmgren M (2000) Combined effects of shade and drought on tulip poplar seedlings: trade-off in tolerance or facilitation? Oikos 90(1):67–78
Holmgren M, Scheffer M, Huston MA (1997) The interplay of facilitation and competition in plant communities. Ecology 78:1966–1975
Humbert L, Gagnon D, Kneeshaw D, Messier C (2007) A shade tolerance index for common understory species of northeastern North America. Ecol Indic 7(1):195–207
Jason JG, Thomas GR, Pharr DM (2004) Photosynthesis, chlorophyll fluorescence, and carbohydrate content of Illicium taxa grown under varied irradiance. J Am Soc Hort Sci 129(1):46–53
Jiménez MD, Pardos M, Puértolas J, Kleczkowski LA, Parados JA (2009) Deep shade alters the acclimation response to moderate water stress in Quercus suber L. Forestry 82(3):285–298
Lambers H, Poorter H (1992) Inherent variation in growth rate between higher plants: a search for physiological causes and ecological consequences. Adv Ecol Res 23:187–261
Lei TT, Lechowicz MJ (1998) Diverse responses of maple saplings to forest light regimes. Ann Bot 82(1):9–19
Lei TT, Tabuchi R, Kitao M, Koike T (1996) Functional relationship between chlorophyll content and leaf reflectance, and light capturing efficiency of Japanese forest species. Physiol Plant 96:411–418
Lichtenthaler HK, Wellburn AR (1983) Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem Soc Trans 11:591–592
Navas M-L, Garnier E (2002) Plasticity of whole plant and leaf traits in Rubia peregrina in response to light, nutrient and water availability. Acta Oecologica 23:375–383
Nobel PS (1984) Productivity of Agave deserti: measurement by dry weight and monthly prediction using physiological responses to environmental parameters. Oecologia 64(1):1–7
Nobel PS (1999) Physicochemical and environmental plant physiology, 2nd edn. Academic Press, San Diego
Nobel PS, Hartsoek TL (1986) Temperature, water, and PAR influences on predicted and measured productivity of Agave deserti at various elevations. Oecologia, 181–185
Noda H, Muraoka H, Washitani I (2004) Morphological and physiological acclimation responses to contrasting light and water regimes in Primula sieboldii. Ecol Res 19:331–340
Poorter H, Nagel O (2000) The role of biomass allocation in the growth response of plants to different levels of light, CO2, nutrients and water: a quantitative review. Aust J Plant Physiol 27:595–607
Quero JL, Villar R, Marañón T, Zamora R (2006) Interactions of drought and shade effects on seedlings of four Quercus species: physiological and structural leaf responses. New Phytol 170(4):819–833
Rozendaal DMA, Hurtado VH, Poorter L (2006) Plasticity in leaf traits of 38 tropical tree species in response to light; relationships with light demand and adult stature. Funct Ecol 20(2):207–216
Sack L, Grubb P (2002) The combined impacts of deep shade and drought on the growth and biomass allocation of shade-tolerant woody seedlings. Oecologia 131(2):175–185
Sack L, Grubb PJ, Marañón T (2003) The functional morphology of juvenile plants tolerant of strong summer drought in shaded forest understories in southern Spain. Plant Ecol 168(1):139–163
Smith T, Humston M (1989) A theory of the spatial and temporal dynamics of plant communities. Vegetatio 83:49–69
Wang ML, Jiang YS, Wei JQ, Wei X, Qi XX, Jiang SY, Wang ZM (2008) Effects of irradiance on growth, photosynthetic characteristics, and artemisinin content of Artemisia annua L. Photosynthetica 46(1):17–20
Warton DI, Wright IJ, Falster DS, Westoby M (2006) Bivariate line-fitting methods for allometry. Biol Rev 81(2):259–291
Xu F, Guo W, Xu W, Wei Y, Wang R (2009) Leaf morphology correlates with water and light availability: what consequences for simple and compound leaves? Prog Nat Sci 19(12):1789–1798
Xu F, Guo WH, Xu WH, Wang RQ (2008) Habitat effects on leaf morphological plasticity in Quercus Acutissima. Acta Biol Cracov Bot 50(2):19–26
Yang Y, Han C, Liu Q, Lin B, Wang J (2008) Effect of drought and low light on growth and enzymatic antioxidant system of Picea asperata seedlings. Acta Physiol Plant 30(4):433–440
Zhang X, Liu J, Welham C, Liu C, Li D, Chen L, Wang R (2006) The effects of clonal integration on morphological plasticity and placement of daughter ramets in black locust (Robinia pseudoacacia). Flora 201(7):547–554
Acknowledgments
We express our deep gratitude to Yinghua Wei, Chong Fang, Huan He, Yifu Yuan, Chengdong Wang, Wenjuan Ding, Penglin Lu and Hua Chen for their help during the experiments. Thanks to Dr. Jian Liu for his valuable comments and suggestions on the submission, to Dr. Roberta Greenwood of Shandong University for linguistic advice. We also thank two anonymous reviewers and the editor for their insightful and helpful comments. The research was supported by National Science Foundation of China (No.30970166; 31270374), Ministry of Education on Doctorial Discipline (No.20090131110066) and Independent Inovation Project of Shandong University (No.2010JC004; 2011DX008).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J. Franklin.
Rights and permissions
About this article
Cite this article
Guo, X., Guo, W., Luo, Y. et al. Morphological and biomass characteristic acclimation of trident maple (Acer buergerianum Miq.) in response to light and water stress. Acta Physiol Plant 35, 1149–1159 (2013). https://doi.org/10.1007/s11738-012-1154-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-012-1154-0