Abstract
Cenchrus (family Poaceae) is an important component of major grass covers of the world. Largely it is apomictic and both annual and perennial species exist in nature. Variations in contents of malondialdehyde, proline, specific leaf area and carbon isotope discrimination for drought tolerance were estimated among eight prominent species of Cenchrus. Simultaneously, genetic variations were also estimated by employing 187 RAPD primers. Of these, 23 primers did not react, 2 performed poorly and 7 produced many non-scorable bands and one primer yielded a single monomorphic band. Rest of the 154 primers generated one or more unambiguously scorable fragments. Twelve hundred and four of the 1,296 putative loci were polymorphic (93%) between at least one pair-wise comparisons among eight species. Dice coefficient and neighbor-joining algorithm analyses showed clustering patterns that fit with the known habitat of the species except perennial, C. myosuroides which formed a node between two annuals species. When these species were subjected to water stress tolerance test, a correlation (r = 0.612) between specific leaf area (SLA) and carbon isotope discrimination (CID) and difference in levels of drought tolerance based parameters among eight species were observed. Of the eight species investigated two annuals viz., C. biflorus and C. echinatus showed highest level of genetic similarity which was also evident from the similar levels of SLA, MDA, proline contents and carbon isotope discrimination values observed in these two species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The grass genus Cenchrus is distributed throughout the tropics but the species reported in the present investigation are primarily from Australia, Africa and of Indian sub-continent. Of the 22 species recognized by Clayton and Renvoize (1982), C. ciliaris L., C. setigerus Vahl. and C. pennisetiformis Hochst and Steud. ex Steud have been largely used as sown pastures with the first being most important in India. The other five species included in the present study are C. prieurri Kunth, C. biflorus Roxb., C. echinatus L. (all annuals), C. glaucus Mudaliar and Sundaraj and C. myosuroides Kunth. Though most of these species are apomictic in nature, some levels of sexuality have been observed in accessions of these species (Dubey 2004). To maximize the potential of sexuality observed in some species molecular characterization of these species hold importance as not much information is available about the genome analysis except classification of 322 accessions of C. ciliaris and C. setigerus using 11 agronomic attributes in six groups largely on the basis of rhizome development, plant maturity and yield (Pengelly et al. 1992). In lack of clear morphological attributes, the identification and grouping of accessions of different species becomes much more difficult when they are collected from their natural habitats. Similar situations have been reported in Dichanthium where complex and intermediates of it with Borthriochloa are found (Chandra et al. 2006).
Information on genetic diversity and relationships within and among crop species and their wild relatives is essential for the efficient utilization of plant genetic resource collections and thus for the management of germplasm and for evolving conservation strategies. Different types of marker systems have been used for genetic analysis and characterization studies. DNA markers are considered the best tools for determining genetic relationships/diversity, as they are highly polymorphic and independent to environmental interactions i.e., highly heritable. Among several markers, random amplified polymorphic DNA (RAPD; Williams et al. 1990) data can be generated faster and with less labor than others. It employs single short primer with an arbitrary sequence to generate genome specific ‘finger print’ of multiple amplification products. Polymorphism found between RAPD profiles can serve as a genetic marker (Williams et al. 1990).
Genetic studies of complex traits such as drought and salt tolerance became easier with the development of informative molecular markers (Pakniyat et al. 1997). In case of drought, transpiration efficiency (TE) is one such trait which contributed tolerance in Stylosanthes scabra and exhibited a significant negative relationship with carbon isotope discrimination (CID) (Thumma et al. 1998). The carbon in atmospheric CO2 is mostly in the form of 12C, but a fraction is also present in the stable isotope form of 13C. During carboxylation, plants discriminate against 13C present in ambient CO2. It has been postulated that plants, which show less discrimination against 13C, would have high TE (Farquhar et al. 1982). The negative relationship between TE and CID has been established in many plant species (Farquhar et al. 1989). Additionally, specific leaf area have been reported to be associated with TE in several plant species (Virgona et al. 1990; Wright et al. 1994; Brown and Byrd 1997), and thus CID and SLA have been used as indirect measures of TE (Johnson and Tieszen 1994; Thumma et al. 1998; Asalatha et al. 1999). At species level, attempts have been made to decipher the mechanism of water stress tolerance and suitable indices like proline accumulation (Hanson et al. 1977), protein metabolism (Thakur and Rai 1982), enzyme defence (Dhindsa and Matowe 1981), solute accumulation (Schwab and Heber 1984), solute leakage (Krishnamani et al. 1984) and H2O2 metabolism including level of lipid peroxidation have been postulated in different crops. The change in level of MDA which generally reflects the lipid peroxidation and consequent changes in the membrane permeability observed differently in species of wheat (Zhang and Kirkham 1994) and jute (Chowdhury and Choudhuri 1985). However, such studies are scanty in major tropical grasses like Cenchrus; despite that such grasses grow and survive under harsh environmental conditions.
In the present paper we report the genetic relationships among eight species of Cenchrus constituting three annuals (C. prieurri, C. biflorus, C. echinatus) and five perennial (C. ciliaris, C. setigerus, C. glaucus, C. pennisetiformis, and C. myosuroides) species and also the clustering patterns of these species in view of their habitat employing neighbor-joining algorithm module. The differing nature of these species towards drought tolerance was assessed by measuring the level of MDA, proline content, specific leaf area (SLA) and carbon isotope discrimination.
Materials and methods
Experimental material and extraction of DNA
Except C. ciliaris IG-69-3108 cultivar, seeds of other accessions of seven species were received from the collections assembled at IGER, Aberystwyth, UK through NBPGR, New Delhi, India (Table 1). C. ciliaris (IG-69-3108), is a native collection and has been released in India based on the production potential of the accession. Seedlings (1-month-old) were transplanted in three replications in bigger plots in experimental fields of the Indian Grassland and Fodder Research Institute, Jhansi (25°27′N, 78°35′E). Genetic purity of each species was maintained as they were replicated from single plant originally raised from pure seed. Fresh and young leaves of three plants of each species were used to isolate the genomic DNA. Due to apomictic nature of the crop it was possible without any problem, especially of homogeneity. Genomic DNA was isolated following the methodology of Iqbal et al. (1997) with suitable modifications (Chandra et al. 2004). For each species, DNA was extracted from 2 g of fresh, young and green leaves. It was ground in liquid nitrogen and the resultant fine powder was mixed with CTAB total DNA extraction buffer [CTAB 2% (w/v), NaCl 1.4%, TRIS–HCl 100 mM, EDTA 20 mM and 2-mercaptoethanol 100 mM (added freshly) in a 1:2 ratio (w/v)] and incubated at 65°C for 1 h with occasional swirling. After incubation, the mixture was emulsified with an equal volume of 24:1 (v/v) chloroform-isoamyl alcohol and centrifuged at 5,000 g for 15 min. The process was repeated once. The aqueous phase was removed and DNA was precipitated with 0.6 volume of isopropanol by keeping at −20°C for 2–3 h. The DNA pellet was dissolved in 10 mM TE buffer (pH 8.0) and kept at 4°C overnight. The nucleic acid solution was treated with RNase and, after incubation for 30 min at 37°C, it was extracted twice with phenol-chloroform and finally with chloroform. DNA was precipitated in 100% ethanol and pelleted at 5,000 g for 10 min and finally washed with 70% ethanol. After a brief period of air-drying, DNA was suspended in 2 ml TE buffer (pH 8.0). The presence and quality of genomic DNA was confirmed by electrophoresis on a 0.7 % (w/v) agarose gel (Sigma). The concentration was adjusted to 5 ng μl−1 for use in PCR analysis.
DNA amplification
Each PCR amplification was performed in a final volume of 20 μl of reaction mixture containing 67 mM TRIS–HCl (pH 8.0), 16.6 mM (NH4)2SO4, 0.45% (v/v) Triton X-100, 4 μg BSA, 3.5 mM MgCl2, 150 μM of each of dATP, dCTP, dGTP and dTTP, 7.5 pmol (15 ng) primer, 25 ng genomic DNA template and 0.5 unit Taq polymerase (Bangalore Genei, India), and finally it was overlaid by 5–10 μl light mineral oil. Primers were procured from Operon Inc. USA. Amplifications were performed on a DNA thermal cycler PTC-200 (MJ Research, USA) with the cycling program consisting of 94°C for 1 min, 37°C for 1 min and 72°C for 2 min for 40 cycles followed by 41st cycle at 37°C for 1 min and finally at 72°C for 10 min extension. The amplified products were kept at 4°C until loaded on to the gel. Amplification products were separated by electrophoresis on 1.6% agarose gel in 0.5× TBE buffer (pH 8.0) to which ethidium bromide was added for visualization with UV light. Along with the unknown samples 100 base pair DNA ladder was also loaded to know the size of the amplified products. Gels were photographed using SLR and Polaroid cameras. PCR reactions were repeated at least once to establish reproducibility of results under strict control of the reaction conditions.
Leaf water potential and drought stress parameters
By withholding water for a period of 1 week, relative water content and water potential were determined which was indicative of the level of stress (data not shown). Relative water content (RWC) was determined using the method of Barrs and Weatherley (1962): RWC = (FW − DW)/(TW − DW) × 100, where FW is the leaf fresh weight, DW is leaf dry weight and TW is turgid weight of tissue after being soaked in water for 6 h at room temperature. Water potential was measured in both controlled and stressed leaves using HR 33T due point micro voltmeter and C-52 sample chambers (Wescor, USA). Middle portion of the leaf was cut into the size of 2.5 × 2.5 mm length and breadth and was placed in sample chamber. Water potential of it was recorded which is a linear function of electro magnetic force produced by the temperature difference between the junction at the dew point temperature and the ambient temperature. The recorded value was further divided by proportionality constant (−0.75 μmoles bar−1) to get the value of water potential in bar which was finally converted into mega Pascal (MPa). Leaf area was measured by the portable leaf area meter LI-3000A. Ten leaves of each species in three replications were used to measure the leaf area and then they were dried in oven at 60°C for dry weight. Specific leaf area was calculated by dividing the leaf area by the dry weight of the leaf and expressed in cm2 g−1.
Malondialdehyde (MDA) was estimated in leaf by following the procedure of Heath and Packer (1968). Two hundred and fifty milligram samples were ground in 5 ml distilled water. In the same solution 5 ml TBA-TCA reagent (0.5% thiobarbituric acid and 20% TCA dissolved in 100 ml of distilled water) was added. The slurry was kept in water bath at 95°C for 30 min. Cooled solution was obtained by placing in ice bath and centrifuged at 10,000 g for 10 min. Optical density of supernatant was recorded at 600 and 535 nm. The difference in optical density gave the actual intensity of color developed by malondialdehyde. The concentration of MDA was calculated from its extinction coefficient of 155 mM−1 cm−1. Proline was extracted in 3% sulphosalicylic acid and estimated by the colorimetric method of Bates et al. (1973).
Transpiration efficiency and CID analysis
The relationship between transpiration efficiency and isotope discrimination was established by designing the experiment as described for Stylosanthes scabra (Thumma et al. 1998). Seedlings (15-day-old) of each of the eight species were transplanted in eight pots. The soil of each pot was prepared with farmyard manure and in each pot, 2 kg soil was placed before transplanting the seedlings. In each pot, two or three seedlings were transplanted. Six pots of each species were finally maintained. After growing the plants for 30 days, two treatments were imposed. In control and stress pots, water was given at field and half of the field capacity, respectively. Pots belonging to each species were regularly randomized to avoid the space effects. In each pot thick layer (2–3 cm) of vermiculite was placed to reduce the amount of evaporation. Three pots without any plants were also maintained in each treatment. Both controlled and stressed plants were watered every second and third day, and on each watering day pots were weighed manually using an electronic balance and the amount of water loss was calculated. Similar amounts of water were provided to unplanted pots too. Temperature and humidity of the glasshouse recorded during experimental periods were 34.08 and 36.4%, respectively. After 60 days of water treatment, plants were harvested (three pots in each species in each treatment) by carefully removing plants from the pots and separating them into roots and shoots. Roots were thoroughly cleaned and soils were removed. Both shoots and roots were oven-dried at 80°C for 48 h. Transpiration was calculated by deducting evaporation (estimated from the unplanted pots) from the total water used of the plants of each pot. Transpiration efficiency (TE) was calculated as total biomass produced per unit of water transpired. At the time of final harvest, SLA was measured by harvesting six of the youngest fully expanded leaves. For isotope discrimination analysis leaf samples used for specific leaf area measurement were finely ground and 10 mg of leaf material was used for the measurement of carbon isotope composition by using isotopes ratio mass spectrometer (IRMS) as described by Farquhar et al. (1989).
Data analysis
A locus was considered as polymorphic if the band was present in some species and absent in others and monomorphic if the band was present in all species. RAPD markers input binary data matrix of eight species of Cenchrus was developed by entering the data by assigning 1 to presence or 0 to absence of bands. Only reproducible and unambiguous RAPD fragments were used for analysis. The NTSYS program version 2.0 was used to produce the similarity matrix (Simqual function). Dice similarity coefficient was used to estimate the genetic similarity. The resulting data were further processed with neighbor-joining algorithm for clustering and generation of dendrogram (Saitou and Nei 1987). The MIDPOINT method of rooting was employed.
Results and discussion
Of the 187 decamer oligonucleotide primers (Operon Technologies, Inc., USA) used for amplification of eight species of Cenchrus genomic DNA, 23 (12.3%) primers did not react, two performed poorly and produced faint bands or smears, seven produced many non-scorable bands and one primer gave single monomorphic band (Dubey and Chandra 2004). Each of the rest i.e., 154 primers (82.4%) generated one or more unambiguously scorable fragments (Fig. 1). Depending on the primer species combination and the amplification condition used, the number of amplification products varied between 2 produced by OPE03, OPF06, OPF07 and OPQ01 primers and 18 produced by OPU01. The fragment size ranged from 0.2 to 1.5 kb. Bands of higher molecular weight (> 2.0 kb), which could not be reproduced consistently were not included in the data analysis. A total of 1,296 DNA fragments were scored. Of these, 1,204 (92.9%) showed polymorphism between at least one pair-wise comparison among eight species of Cenchrus, and the remaining 92 (7.1q%) bands were monomorphic produced by sixty-five primers (Table 2). Of these, forty-nine produced one monomorphic band, nine generated two and rest seven produced more than two monomorphic bands.
RAPD patterns obtained from individual genomic DNA samples of Cenchrus species. a Amplified fragments with primers OPB1, OPB5 and OPB15 where most of the bands were scored except very faint. b Primers like OPF9, OPF10 and OPF11 produced less number of bands and also indicating similar RAPD patterns by more than one species. Lane 1: C. ciliaris; lane 2: C. setigerus; lane 3: C. pennisetiformis; lane 4: C. prieurri; lane 5: C. biflorus; lane 6: C. echinatus; lane 7: C. myosuroides and lane 8: C. glaucus. Arrow indicates the threshold band intensity for scoring of the bands with all primers. M 100 bp DNA ladder as molecular weight marker
RAPD markers have been reported for its wide applications due to the easiness of performing the assay and also being less time consuming and inexpensive. The level of polymorphism demonstrated that RAPD technology is quite useful to identify DNA polymorphisms among the cultivars and accessions of agronomically important Cenchrus species. The problem often encountered and questioned regarding RAPD analysis is the reproducibility of banding patterns. However, this has been minimized by thoroughly optimizing PCR reaction conditions and following the same protocol each and every time. Our experience demonstrated that an assay could be made satisfactorily reproducible with sufficient repetitions and by systematically disregarding faint amplified bands by keeping threshold band intensity (Chandra et al. 2004). Weeden et al. (1994) have reported that by disregarding faint staining bands the error rate can be reduced from 8 to 0%. We also observed loss in number of bands when annealing temperature was increased to strengthen the stringency (Dubey 2004) as reported by Levi et al. (1993). For more accurate analysis, reaction should be performed twice or thrice, scoring only those bands that are reproducible in each reaction (Chandra et al. 2004). The high level of polymorphism as observed here by RAPD markers could be valuable in studies that require high numbers of polymorphic loci, such as population association genetics study to construct genetic linkage map or marking a single gene.
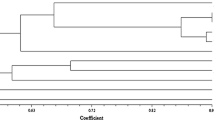
One thousand two hundred and ninety six fragments were used to generate the input matrix and further by using the routine SIMQUAL, similarity coefficients (DICE) was generated among eight species of Cenchrus. Neighbor-joining algorithm was used for clustering (Fig. 2). Cenchrus biflorus formed an isolated node whereas C. setigerus and C. pennisetiformis clustered in one group. Four species namely C. echinatus, C. myosuroides, C. prieurri and C. glaucus occupy a position between these two groups in dendrogram developed based on RAPD data. Interestingly, among these four species, C. myosuroides of the collection of Paraguay being perennial surrounded by two annual species. Our earlier study has indicated more closeness of perennial C. myosuroides species to the annuals when they were analyzed for different drought tolerance biochemical parameters (Dubey 2004). The C. ciliaris, which is the only accession of India formed a shared node with two other species namely C. setigerus of Uganda and C. pennisetiformis of Kenya. These three species are perennial and were close to the node made by another perennial species namely C. glaucus (Fig. 2), however, the closest node was made by C. biflorus and C. echinatus indicated highest level of genetic similarity among them which was also evident from the similar levels of SLA, MDA, proline contents and carbon isotope discrimination values (Table 3).
RAPD markers have been used earlier to study the taxonomic and phylogenetic relationships (Millan et al. 1996; Singh et al. 2004; Zhang et al. 2005; Padmesh et al. 2006) and depicted high polymorphism than RFLP and AFLP markers (Thormann et al. 1994; Das et al. 1999). RAPD markers in the present study also revealed a high degree of polymorphism (93%) among the eight species of Cenchrus as evident from the fact that of the 187 decamer oligonucleotide primers only one gave a single monomorphic band. Being apomictic in nature, this level of polymorphism (up to 93%) observed between eight Cenchrus species demonstrated that genetic base is quite broad and it is more in annual (52 % dissimilarity) than perennial (40% dissimilarity) species. Moreover, high level of genetic variations as observed in other tropical grass i.e. Dichanthium, further corroborated that some of the species of Cenchrus are wild in form and origins and still maintains heterozygosity as apomixis is one system that has evolved to preserve heterozygosity.
The mean decrease in SLA was 6.7% in stress over control. The carbon isotope discrimination values decreased from a mean value of 13.9 in control to 13.3 in the stress treatment (Fig. 3). Species namely C. myosuroides maintained highest TE (lowest CID) under both well-watered and water-stressed treatments (Fig. 4). Apart from C. myosuroides, C. glaucus, C. prieurri, C. setigerus and C. echinatus maintained lower level of CID. Of these four species, higher level of TE was observed only with C. glaucus and C. setigerus whereas C. prieurri maintained least TE (Fig. 4). This might be due to growing nature of the plant which is very short in height and largely exists in the dormant stage. In control after C. myosuroides, specific leaf area which was lowest in C. ciliaris indicated thicker leaves which signified the drought tolerant nature of the species which was further supported by more TE and proline content (Table 3). Greathead et al. (1998) have also reported the drought tolerant nature of C. ciliaris species. Even after imposing stress, decrease in SLA was minimal in C. ciliaris, which further indicated its stability under stress. Contrary to this, C. prieurri showed maximum decrease in SLA under stress. In general, level of MDA decreased whereas proline content increased when plants were exposed to water stress. Among eight species, MDA content ranged from 16.12 to 44.11 [nmol g−1 (FM)]) and proline from 0.55 to 0.88 [μmol g−1 (FM)] under control. Under drought conditions, a significant decrease in MDA content was observed in C. ciliaris indicating low peroxidation of lipid when plants were exposed to water stress. Proline content increased significantly in all species under stress. Among eight species, maximum increase in proline content under stress was noticed in C. pennisetiformis whereas minimum in C. prieurri (Table 3). Thus, results indicated variations in MDA and proline content among eight species of Cenchrus belonging to habitat, i.e. annual and perennial.
A strong positive relationship was found between SLA and CID in both control (r = 0.658) and stress (r = 0.612) (Fig. 5). SLA has been shown to be negatively related to transpiration efficiency in many crops (Virgona et al. 1990; Brown and Byrd 1997; Thumma et al. 1998), thus could be used as a surrogate of CID in screening large numbers of accessions/segregating lines in the early phase of breeding programs, whereas CID may be more suitable for precise analysis of the advanced lines as suggested by Richards (1996). The drought-induced changes in the level of lipid peroxidation (malondialdehyde content) in eight species indicated increase in the level of MDA under water stress, however, rate of increase of lipid peroxidation observed was different between species. The level of proline increased in all species and was close to each other except C. prieurri (Table 3).
Relationship between CID with TE and SLA. a Relationship between CID and TE in eight species of Cenchrus (a) control (b) stress treatment. Though association was non-significant it was negative. b Relationship between CID and SLA in eight species of Cenchrus (a) control (b) stress treatment. Association was significant in both control and stress conditions
In summary, RAPD markers clearly divided perennial from annual species of Cenchrus. However, C. myosuroides was the only exception which clustered with annual though being perennial. Among annuals it depicted more closeness with C. biflorus and C. echinatus than C. prieurri. Both specific leaf area and CID was least in C. myosuroides whereas TE was highest. Some of the attributes of drought tolerance for this species observed close to annual while for others it inclined towards perennial. This makes us to speculate that C. myosuroides may be a bridging species depicting genetically more closeness with annuals. Differing nature of the level of peroxidation (membrane permeability) as depicted by MDA level and many fold increase in proline content possibly led to the variations for drought tolerance of Cenchrus species. The significant association between specific leaf area and CID can be exploited in selecting drought tolerant genotypes. Therefore, using CID, rather than markers associated with it may be sufficient in breeding programs to improve TE. Thus, the study provided information that could be exploited in screening large number of accessions on the basis of established drought tolerance characters and polymorphic RAPD primers for diversity analysis with the presented benchmark data.
Abbreviations
- CID:
-
Carbon isotope discrimination
- MDA:
-
Malondialdehyde
- RAPD:
-
Random amplified polymorphic DNA
- SLA:
-
Specific leaf area
- TE:
-
Transpiration efficiency
References
Asalatha M, Reddy PV, Rao R (1999) Relationship of specific leaf area and carbon isotope discrimination with dry matter production and pod yield in groundnut genotype under moisture stress. Indian J Plant Physiol 4:161–166
Barrs HD, Weatherley PE (1962) A re-examination of relative turgidity technique for estimating water deficit in leaves. Aust J Biol Sci 15:413–428
Bates LS, Walbren RP, Teare TD (1973) Rapid determination of free proline water stress studies. Plant Soil 39:205–207
Brown RH, Byrd GT (1997) Transpiration efficiency, specific leaf weight, and mineral concentration in peanut and pearl millet. Crop Sci 37:475–480
Chandra A, Saxena R, Roy AK, Pathak PS (2004) Estimation of genetic variation in Dichanthium annulatum genotypes by RAPD technique. Trop Grasslands 38:245–252
Chandra A, Saxena R, Roy AK (2006) Polymorphism and genotype-specific markers for Dichanthium identified by random amplified polymorphic DNA. Genet Resour Crop Evol 53:1521–1529
Chowdhury SR, Choudhuri MA (1985) Hydrogen peroxide metabolism as an index of water stress tolerance in jute. Physiol Plant 65:503–507
Clayton WD, Renvoize SA (1982) Cenchrus. In: Polhill RM (ed) Flora of tropical East Africa, Rotterdam, pp 691–695
Das S, Rajgopal J, Bhatia S, Srivastava PS, Lakshmikumaran M (1999) Assessment of genetic variation within Brassica compestris cultivars using amplified fragment length polymorphism and random amplification of polymorphic DNA markers. J Biosci 24:433–440
Dhindsa RS, Matowe W (1981) Drought tolerance in two mosses: Correlated with enzymatic defence against lipid peroxidation. J Exp Bot 32:79–91
Dubey A (2004) Moisture stress induced biochemical changes in Cenchrus species. Ph.D. Thesis, Bundelkhand University, India
Dubey A, Chandra A (2004) Identification of primers for DNA fingerprints in genus Cenchrus. Range Manage Agrofor 25:36–40
Farquhar GD, Bernhart D, Waines JG (1982) On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentrations in leaves. Aust J Plant Physiol 9:121–137
Farquhar GD, Ehleringer JR, Hubick KT (1989) Carbon isotope discrimination and photosynthesis. Annu Rev Plant Physiol 40:388–397
Greathead K, Sanford P, Cransberg L (1998) Perennial grasses for animal production in the high rainfall areas of Western Australia. Miscellaneous Publication no. 2/98, New South Wales, Agnote; Department of Primary Industries, Queensland
Hanson AD, Nelson CE, Everson EH (1977) Evolution of proline accumulation as an index of drought resistance using two contrasting barley cultivars. Crop Sci 17:720–726
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplast. I. Kinetics and stoichiometry of fatty acids peroxidation. Arch Bioch Biophys 125:189–198
Iqbal MJ, Aziz N, Saeed NA, Zafar Y, Malik KA (1997) Genetic diversity evaluation of some elite cotton varieties by RAPD analysis. Theor Appl Genet 94:139–144
Johnson RC, Tieszen LL (1994) Variation for water use efficiency in alfalfa germplasm. Crop Sci 34:452–458
Krishnamani MRS, Yopp JH, Myers O Jr (1984) Leaf solute leakage as a drought tolerance indicator in soybean. Phyton 44:43–49
Levi A, Rowland IJ, Hartung J (1993) Production of reliable randomly amplified polymorphic DNA (RAPD) markers from DNA of woody plants. Hort Sci 28:1188–1190
Millan T, Osuna F, Cobos S, Torres AM, Cukero JI (1996) Using RAPDs to study phylogenetic relationships in Rosa. Theor Appl Genet 92:273–277
Padmesh P, Reji JV, Jinish Dhar M, Seeni S (2006) Estimation of genetic diversity in varieties of Mucuna pruriens using RAPD. Biol Plant 50:367–372
Pakniyat H, Powell W, Baird E, Handley LL, Robinson D, Scrimgeour CM, Nevo E, Hackett CA, Caligari PDS, Forster BP (1997) AFLP variation in wild barley (Hordeum spontaneum C. Koch) with reference to salt tolerance and associated ecogeography. Genome 40:332–341
Pengelly BC, Hacker JB, Eagler DA (1992) The classification of a collection of buffer grasses and related species. Trop Grasslands 76:6–11
Richards RA (1996) Defining selection criteria to improve yield under drought. Plant Growth Regul 20:157–166
Saitou N, Nei M (1987) The neighbour-joining method: a new method for reconstructing phylogenic tree. Mol Biol Evol 4:406–425
Schwab KB, Heber U (1984) Thylakoid membrane stability in drought tolerant and drought sensitive plants. Planta 161:37–45
Singh RK, Sharma RK, Singh AK, Singh VP, Singh NK, Tiwari SP, Mohapatra T (2004) Suitability of mapped sequence tagged microsatellites site markers for establishing distinctness, uniformity and stability in aromatic rice. Euphytica 135:135–143
Thakur PS, Rai VK (1982) Effect of water stress on protein content in two (Zea mays) cultivars differing in drought resistance. Biol Plant 24:96–100
Thormann CE, Ferreira ME, Camargo LEA, Tivang JG, Osborn TC (1994) Comparison of RFLP and RAPD markers to estimating genetic relationships within and among cruciferous species. Theor Appl Genet 88:973–980
Thumma BR, Naidu PB, Cameron DF, Bahnisch LM (1998) Transpiration efficiency and its relationship with carbon isotope discrimination under well watered and water–stressed conditions in Stylosanthes scabra. Aust J Agril Res 49:1039–1045
Virgona JM, Hubick KT, Rawson HM, Farquhar GD, Downes RW (1990) Genotypic variation in transpiration efficiency, carbon isotope discrimination and carbon allocation during early growth in sunflower. Aust J Plant Physiol 17:207–214
Weeden NF, Hemmat M, Lawson DM, Lodhi M, Bell RL, Manganaris AG, Resch BI, Brown SK, Ye GN (1994) Development and application of molecular markers linkage map in woody fruit crops. Euphytica 77:71–75
Williams JGK, Kubehk AR, Livak KI, Rafalski JA, Tingey SV (1990) DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res 18:6531–6535
Wright GC, Rao RCN, Farquhar GD (1994) Water use efficiency and carbon isotope discrimination in peanuts under water deficit conditions. Crop Sci 34:92–97
Zhang Z, Kirkham MB (1994) Drought stress induced changes in activities of superoxide dismutase, catalase and peroxidase in wheat species. Plant Cell Physiol 35:785–791
Zhang HY, Liu X.Z, He CS, Zheng (2005) Random amplified DNA polymorphism of Nicotiana tabacum L. cultivars. Biol Plant 49:605–607
Acknowledgments
Authors are thankful to Director and Head of the Division for providing the necessary facilities to carry out the work. The seed of Cenchrus species provided by Mr. U.S. Mishra is duly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Stobiecki.
Rights and permissions
About this article
Cite this article
Chandra, A., Dubey, A. Evaluation of genus Cenchrus based on malondialdehyde, proline content, specific leaf area and carbon isotope discrimination for drought tolerance and divergence of species at DNA level. Acta Physiol Plant 30, 53–61 (2008). https://doi.org/10.1007/s11738-007-0090-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-007-0090-x