Abstract
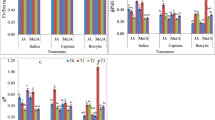
In Arabidopsis thaliana leaves a strong increase of H2O2 content was induced by application of methyl jasmonate (JAMe) through the root system, but the induction only slightly depended on JAMe concentration. The activity of superoxide dismutase and ascorbic acid peroxidase increased at lower JAMe concentrations and decreased at higher ones. Catalase activity decreased proportionally to JAMe concentration (in comparison with control plants). The sum of ascorbic acid and dehydroascorbate content at 10−6 M JAMe was similar to the control, but at higher concentrations it increased, especially due to a higher ascorbate accumulation. Methyl jasmonate applied directly to the extract of leaves (in vitro experiment) also induced a strong increase in H2O2 level, even at a low concentration (10−8 M). Since lower JAMe concentrations induced weak superoxide dismutase and did not change catalase and peroxidase activity, it is suggested that in this case a high level of hydrogen peroxide was not the result of the activity of the mentioned enzymes. JAMe-induction of H2O2 increase at the highest JAMe concentration resulted from SOD activity. Our in vivo and in vitro experiments suggest that jasmonate can influence oxidative stress not only through gene expression but also by its direct effect on enzyme activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aebi H. 1984. Catalase in vitro. Meth. Enzymol., 105: 121–126.
Arnott T., Murphy T.M. 1991. A comparison of the effects of a fungal elicitor and ultraviolet radiation on ion transport and hydrogen peroxide synthesis by rose cells. Environ. Exp. Bot., 31: 209–216.
Auh Ch-K., Murphy T.M. 1995. Plasma membrane redox enzyme involved in the synthesis of O −2 and H2O2 Phytophthora elicitor-stimulated rose cells. Plant Physiol., 107: 1241–1247.
Beauchamp C.O., Fridovich I. 1971. Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Anal. Biochem., 44: 276–287.
Bell E., Creelman R.A., Mullet J.E. 1995. A chloroplast lipoxygenase is required for wound-induced jasmonic acid accumulation in Arabidopsis. Proc. Natl. Acad. Sci. USA, 92: 8675–8679.
Biemelt S., Keetma U., Mock H-P., Grimm B. 2000. Expression and activity of isoenzymes of superoxide dismutase in wheat roots in response to hypoxia and anoxia. Plant Cell Environ., 23: 135–144.
Bostock R.M. 1999. Signal conflicts and synergies in induced resistance to multiple attackers. Physiol. Molec. Plant Pathol., 55: 99–109.
Bradford M.M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye-binding. Anal. Biochem., 72: 248–254.
Czapski J., Saniewski M. 1988. The effect of methyl jasmonate on polyphenol oxidase and peroxidase activities in tomato fruit. Bull. Acad. Pol. Sci. Biol., 36: 127–132.
Chain H.B., Doke N. 1987. Superoxide anion generation: a response of potato leaves to infection with Phytophthora infectans. Phytopathology, 77: 645–649.
Creelman R.A., Tierney M.L., Mullet J. 1992. Jasmonic acid/methyl jasmonate accumulate in wounded soybean hypocotyls and modulate wound gene expression. Proc. Natl. Acad. Sci. USA, 89: 4938–4941.
Creelman R.A., Mullet J. 1997. Oligosaccharins, brassinolides, and jasmonates: nontraditional regulators of plant growth, development, and gene expression. Plant Cell, 9: 1211–1223.
Cuypers A., Vangronsveld J., Clijsters H. 2000. Biphasic effect of copper on ascorbate-glutathione pathway in primary leaves of Phaseolus vulgaris seedlings during the early stages of metal assimilation. Physiol. Plant., 110: 512–517.
Depège N., Varenne M., Boyer N. 2000. Induction of oxidative stress and GPH-like protein activation in tomato plants after mechanical stimulation. Physiol. Plant., 110: 209–214.
Doke N. 1997. The oxidative burst: roles in signal transduction and plant stress. In: Oxidative Stress and the Molecular Biology of Antioxidant Defense, ed. by J.G. Scandalios, Cold Spring Harbor Laboratory Press, New York: 785–813.
Foyer Ch., Rowell J., Walker D. 1983. Measurement of the ascorbate content of spinach leaf protoplasts and chloroplasts during illumination. Planta, 157: 239–244.
Gönner M.V., Schl’sser E. 1993. Oxidative stress in interactions between Avena sativa L. and Drechslera spp. Physiol. Mol. Plant. Pathol., 42: 221–234.
Kar M., Streb P., Hertwig B., Feierabend L. 1993. Sensitivity to photodamage increases during senescence in excised leaves. J. Plant Physiol., 141: 538–544.
Kuźniak E., Skłodowska M. 2001. Ascorbate, glutathione and related enzymes in chloroplasts of tomato leaves infected by Botrytis cinerea. Plant Sci., 160: 723–731.
Lamb C., Dixon R. 1997. The oxidative burst in plant disease resistance. Annu. Rev. Plant Physiol. Plant Mol. Biol., 48: 251–275.
León J., Rojo E., Sánchez-Serrano J.J. 2001. Wound signalling in plants. J. Exp. Bot., 52: 1–9.
Lin J.-N., Kao Ch.H. 1998. Effect of oxidative stress caused by hydrogen peroxide on senescence of rice leaves. Bot. Bull. Acad. Sin., 39: 161–165.
Maksymiec W., Krupa Z. 2002. Jasmonate and heavy metals in Arabidopsis plants — a similar physiological response to both stressors? J. Plant Physiol., 159: 509–515.
Møller S.G., Chua N-H. 1999. Interactions and intersections of plant signalling pathways. J. Mol. Biol., 293: 219–234.
Nakano Y., Asada K. 1981. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol., 22: 867–880.
Orendi G., Zimmermann P., Baar C., Zentgraf U. 2001. Loss of stress-induced expression of catalase3 during leaf senescence in Arabidopsis thaliana is restricted to oxidative stress. Plant Sci., 161: 301–314.
Orozco-Cárdenas M.L., Ryan C.A. 1999. Hydrogen peroxide is generated systemically in plant leaves by wounding and systemin via the octadecanoid pathway. Proc. Natl. Acad. Sci. USA, 96: 6553–6557.
Orozco-Cárdenas M.L., Narváez-Vásquez J., Ryan C.A. 2001. Hydrogen peroxide acts as a second messenger for the induction of defense genes in tomato plants in response to wounding, systemin, and methyl jasmonate. Plant Cell, 13: 179–191.
Penninckx I., Eggermont K., Terras F., Thomma B., Samblanx G., Buchala A., Metraux J-P., Manners J., Broekaert W. 1996. Pathogen-induced systemic activation of jasmonate and ethylene response pathways is required for induction of a plant defensin gene in Arabidopsis. Plant Cell, 8: 2309–2323.
Peña-Cortés H., Albrecht T., Prat S., Weiler E.W., Willmitzer L. 1993. Aspirin prevents wound-induced gene expression in tomato levels by blocking jasmonic acid biosynthesis. Planta, 191: 123–128.
Pick E. 1986. Microassays for superoxide and hydrogen peroxide production and nitroblue tetrazolium reduction using an enzyme immunoassay microplate reader. Methods Enzymol., 132: 407–421.
Rao M.V., Lee H.I., Creelman R.A., Mullet J.E., Davis K.R. 2000. Jasmonic acid signaling modulates ozone-induced hypersensitive cell death. Plant Cell Physiol., 12: 1633–1646.
Ratajczak R., Feussner I., Hause B., Böhm A., Parthier B., Wasternack C. 1998. Alteration of V-type H+-ATPase during methyljasmonate-induced senescence in barley (Hordeum vulgare L. cv. Salome). J. Plant Physiol., 152: 199–206.
Rojo E., Leü J., Sánchez-Serrano J.J. 1999. Cross-talk between wound signalling pathways determines local versus systemic gene expression in Arabidopsis thaliana. Plant J., 20: 135–142.
Saniewski M., Ueda J., Miyamoto K. 1999. Interaction of ethylene with jasmonates in the regulation of some physiological processes in plants. In: Biology and Biotechnology of the Plant Hormone Ethylene II. Ed. By A.K. Kanellis, C. Chang, H. Klee, A.B. Bleecker, J.C. Pech, D. Grierson, Kluwer Academic Publ., Dordrecht: 173–180.
Sembdner G., Parthier B. 1993. The biochemistry and the physiological and molecular actions of jasmonates. Annu. Rev. Plant Physiol. Plant Mol. Biol., 44: 569–589.
Shoji T., Nakajima K., Hashimoto T. 2000. Ethylene suppresses jasmonate-induced gene expression in nicotine biosynthesis. Plant Cell Physiol., 41: 1072–1076.
Wasternack C., Parthier B. 1997. Jasmonate-signalled plant gene expression. Trends Biochem. Sci., 2: 302–307.
Titarienko E., Rojo E., León J., Sánchez-Serrano J.J. 1997. JA-dependent and independent signalling pathways control wound-induced gene activation in Arabidopsis thaliana. Plant Physiol., 115: 817–826.
Ueda J., Kato J. 1980. Isolation and identification of senescence-promoting substances from wormwood (Artemisia absinthium L.). Plant Physiol., 66: 246–249.
Véronési Ch., Pouénta M.-L., Riskauer M. and Esquerré J Tugay J M.-T. 1999. Regulation of tobacco lipoxygenase by methyl jasmonate and fatty acids. Plant Biol. Pathol., 322: 491–497.
Weckx J.E.J., Clijsters H.M.M. 1997. Zn phytotoxicity induces oxidative stress in primary leaves of Phaseolus vulgaris. Plant Physiol. Biochem., 35: 405–410.
Xiang C., Oliver D.J. 1998. Glutathione metabolic genes coordinately respond to heavy metals and jasmonic acid in Arabidopsis. Plant Cell, 10: 1539–1550.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maksymiec, W., Krupa, Z. The in vivo and in vitro influence of methyl jasmonate on oxidative processes in Arabidopsis thaliana leaves. Acta Physiol Plant 24, 351–357 (2002). https://doi.org/10.1007/s11738-002-0029-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11738-002-0029-1