Abstract
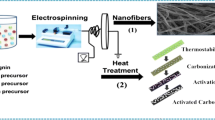
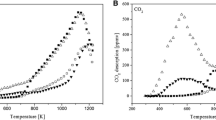
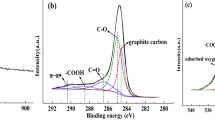
Two types of lignin-based carbon fibers were prepared by electrospinning method. The first was activated with Fe3O4 (LCF-Fe), and the second was not activated with Fe3O4 (LCF). Gas phase adsorption isotherms for toluene on LCF-Fe and LCF were studied. The gas phase adsorption isotherm for 0% RH showed LCF-Fe have about 439 mg/g adsorption capacity which was close to that of commercially available activated carbon (500 mg/g). The Dubinin-Radushkevich equation described the isotherm data very well. Competitive adsorption isotherms between water vapor and toluene were measured for their RH from 0 to 80%. The effect of humidity on toluene gas-phase adsorption was predicted by using the Okazaki et al. model. In addition, a constant pattern homogeneous surface diffusion model (CPHSDM) was used to predict the toluene breakthrough curve of continuous flow-packed columns containing LCF-Fe, and its capacity was 412 mg/g. Our study, which included material characterization, adsorption isotherms, kinetics, the impact of humidity and fixed bed performance modeling, demonstrated the suitability of lignin-based carbon fiber for volatile organic compound removal from gas streams.

Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Lillo-Ródenas M A, Cazorla-Amorós D, Linares-Solano A. Behaviour of activated carbons with different pore size distributions and surface oxygen groups for benzene and toluene adsorption at low concentrations. Carbon, 2005, 43(8): 1758–1767
Gupta V K, Verma N. Removal of volatile organic compounds by cryogenic condensation followed by adsorption. Chemical Engineering Science, 2002, 57(14): 2679–2696
Das D, Gaur V, Verma N. Removal of volatile organic compound by activated carbon fiber. Carbon, 2004, 42(14): 2949–2962
Long C, Liu P, Li Y, Li A, Zhang Q. Characterization of hydrophobic hypercrosslinked polymer as an adsorbent for removal of chlorinated volatile organic compounds. Environmental Science & Technology, 2011, 45(10): 4506–4512
Sullivan P, Moate J, Stone B, Atkinson J D, Hashisho Z, Rood M J. Physical and chemical properties of PAN-derived electrospun activated carbon nanofibers and their potential for use as an adsorbent for toxic industrial chemicals. Adsorption, 2012, 18(3–4): 265–274
Hashisho Z, Emamipour H, Rood M J, James Hay K, Kim B J, Thurston D. Concomitant adsorption and desorption of organic vapor in dry and humid air streams using microwave and direct electrothermal swing adsorption. Environmental Science & Technology, 2008, 42(24): 9317–9322
Foster K L, Fuerman R G, Economy J, Larson S M, Rood M J. Adsorption characteristics of trace volatile organic compounds in gas streams onto activated carbon fibers. Chemistry of Materials, 1992, 4(5): 1068–1073
Qiao WM, Huda M, Song Y, Yoon S H, Korai Y, Mochida I, Katou O, Hayashi H, Kawamoto K. Carbon fibers and films based on biomass resins. Energy & Fuels, 2005, 19(6): 2576–2582
Carrott P J M, Carrott M M L R. Lignin-from natural adsorbent to activated carbon: A review. Bioresource Technology, 2007, 98(12): 2301–2312
Hayashi J, Kazehaya A, Muroyama K, Watkinson A P. Preparation of activated carbon from lignin by chemical activation. Carbon, 2000, 38(13): 1873–1878
Ruiz-Rosas R, Bedia J, Lallave M, Loscertales I G, Barrero A, Rodríguez-Mirasol J, Cordero T. The production of submicron diameter carbon fibers by the electrospinning of lignin. Carbon, 2010, 48(3): 696–705
Ahmad J J, Babak K, Nemat J, Roshanak R K, Mehdi A, Ali A B. Fenton-like catalytic oxidation of tetracycline by AC@Fe3O4 as a heterogeneous persulfate activator: Adsorption and degradation studies. Journal of Industrial and Engineering Chemistry, 2017, 45: 323–333
Chiang Y C, Chiang P C, Huang C P. Effects of pore structure and temperature on VOC adsorption on activated carbon. Carbon, 2001, 39(4): 523–534
Lordgooei M, Kim M S. Modeling volatile organic compound sorption in activated carbon. I: Dynamics and single-component equilibrium. Journal of Environmental Engineering, 2004, 130(3): 212–222
Cosnier F, Celzard A, Furdin G, Bégin D, Marêché J F. Influence of water on the dynamic adsorption of chlorinated VOCs on active carbon: Relative humidity of the gas phase versus pre-adsorbed water. Adsorption Science and Technology, 2006, 24(3): 215–228
Kim T Y, Kim S J, Cho S Y. Effect of relative humidity on the adsorption characteristics of carbon tetrachloride in a fixed bed. Journal of Industrial and Engineering Chemistry, 2004, 10(2): 188–195
Russell B P, LeVan M D. Coadsorption of organic compounds and water vapor on BPL activated carbon.3. Ethane, propane, and mixing rules. Industrial & Engineering Chemistry Research, 1997, 36(6): 2380–2389
Marban G, Fuertes A B. Co-adsorption of n-butane/water vapour mixtures on activated carbon fibre-based monoliths. Carbon, 2004, 42(1): 71–81
Kaplan D, Nir I, Shmueli L. Effects of high relative humidity on the dynamic adsorption of dimethyl methylphosphonate (DMMP) on activated carbon. Carbon, 2006, 44(15): 3247–3254
Ryu Y K, Lee H J, Yoo H K, Lee H C. Adsorption equilibria of toluene and gasoline vapors on activated carbon. Journal of Chemical & Engineering Data, 2002, 47(5): 1222–1225
El-Sharkawy I I, He J M, Ng K C, Yap C, Saha B B. Adsorption equilibrium and kinetics of gasoline vapors onto carbon-based adsorbents. Journal of Chemical & Engineering Data, 2007, 53(1): 41–47
Brosillon S, Manero M H, Foussard J N. Mass transfer in VOC adsorption on zeolite: Experimental and Theoretical Breakthrough Curves. Environmental Science & Technology, 2001, 35(17): 3571–3575
Díez N, Álvarez P, Granda M, Blanco C, Santamaría R, Menéndez R. A novel approach for the production of chemically activated carbon fibers. Chemical Engineering Journal, 2015, 260: 463–468
Song M, Jin B, Xiao R, Yang L, Wu Y M, Zhong Z P, Huang Y J. The comparison of two activation techniques to prepare activated carbon from corn cob. Biomass and Bioenergy, 2013, 48: 250–256
Yun J H, Hwang K Y, Choi D K. Adsorption of benzene and toluene vapors on activated carbon fiber at 298, 323, and 348 K. Journal of Chemical & Engineering Data, 1998, 43(5): 843–845
Wood G O. Affinity coefficients of the Polanyi/Dubinin adsorption isotherm equations: A review with compilations and correlations. Carbon, 2001, 39(3): 343–356
Stoeckli F, Lavanchy A. The adsorption of water by active carbons, in relation to their chemical and structural properties. Carbon, 2000, 38(3): 475–477
Crittenden J C, Rigg T J, Perram D L, Tang S R, Hand D W. Predicting gas-phase adsorption equilibria of volatile organics and humidity. Journal of Environmental Engineering, 1989, 115(3): 560–573
Okazaki M, Tamon H, Toei R. Prediction of binary adsorption equilibria of solvent and water vapor on activated carbon. Journal of Chemical Engineering of Japan, 1978, 11(3): 209–215
Defay R, Prigogine I, Bellemans A. Surface Tension and Adsorption. New York: Longman, 1966
Crittenden J C, Cartright P D, Rick B, Tang S R, Perram D L. An Evaluation of the Technical Feasibility of the Air-Stripping Solvent Recovery Process. Denver: American Water Works Association Research Foundation, 1987
Acknowledgements
The authors are grateful for the National Natural Science Foundation of China (Grant No. 51576048). Thanks to the financial support of Jiangsu Province Environmental Protection Foundation (2015013), the Industry, Education, and Research Prospective Project of Jiangsu Province (BY2015060-04) and Fok Ying Tong Education Foundation (142026). This research was also supported by the Brook Byers Institute for Sustainable Systems, Hightower Chair, and the Georgia Research Alliance at the Georgia Institute of Technology.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Song, M., Zhang, W., Chen, Y. et al. The preparation and performance of lignin-based activated carbon fiber adsorbents for treating gaseous streams. Front. Chem. Sci. Eng. 11, 328–337 (2017). https://doi.org/10.1007/s11705-017-1646-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11705-017-1646-y