Abstract
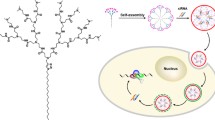
Small interfering RNA (siRNA) therapeutics hold great promise to treat a variety of diseases, as long as they can be delivered safely and effectively into cells. Dendrimers are appealing vectors for siRNA delivery by virtue of their well-defined molecular architecture and multivalent cooperativity. However, the clinical translation of RNA therapeutics mediated by dendrimer delivery is hampered by the lack of dendrimers that are of high quality to meet good manufacturing practice standard. In this context, we have developed small amphiphilic dendrimers that self-assemble into supramolecular structures, which mimic high-generation dendrimers synthesized with covalent construction, yet are easy to produce in large amount and superior quality. Indeed, the concept of supramolecular dendrimers has proved to be very promising, and has opened up a new avenue for dendrimer-mediated siRNA delivery. A series of self-assembling supramolecular dendrimers have consequently been established, some of them out-performing the currently available nonviral vectors in delivering siRNA to various cell types in vitro and in vivo, including human primary cells and stem cells. This short review presents a brief introduction to RNAi therapeutics, the obstacles to their delivery and the advantages of dendrimer delivery vectors as well as our bio-inspired structurally flexible dendrimers for siRNA delivery. We then highlight our efforts in creating selfassembling amphiphilic dendrimers to construct supramolecular dendrimer nanosystems for effective siRNA delivery as well as the related structural alterations to enhance delivery efficiency. The advent of self-assembling supramolecular dendrimer nanovectors holds great promise and heralds a new era of dendrimer-mediated delivery of RNA therapeutics in biomedical applications.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bobbin M L, Rossi J J. RNA interference (RNAi)-based therapeutics: Delivering on the promise? Annual Review of Pharmacology and Toxicology, 2016, 56(1): 103–122
Haussecker D, Kay M A. Drugging RNAi. Science, 2015, 347(6226): 1069–1070
Crunkhorn S. Trial watch: Pioneering RNAi therapy shows antitumour activity in humans. Nature Reviews. Drug Discovery, 2013, 12(3): 178–178
Castanotto D, Rossi J J. The promises and pitfalls of RNAinterference-based therapeutics. Nature, 2009, 457(7228): 426–433
Fire A, Xu S, MontgomeryMK, Kostas S A, Driver S E, Mello C C. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature, 1998, 391(6669): 806–811
Bernstein E, Caudy A A, Hammond S M, Hannon G J. Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature, 2001, 409(6818): 363–366
Ameres S L, Martinez J, Schroeder R. Molecular basis for target RNA recognition and cleavage by human RISC. Cell, 2007, 130(1): 101–112
Hutvágne G, Zamore P D. A microRNA in a multiple-turnover RNAi enzyme complex. Science, 2002, 297(5589): 2056–2060
Yin H, Kanasty R L, Eltoukhy A A, Vegas A J, Dorkin J R, Anderson D G. Non-viral vectors for gene-based therapy. Nature Reviews. Genetics, 2014, 15(8): 541–555
Kanasty R, Dorkin J R, Vegas A, Anderson D. Delivery materials for siRNA therapeutics. Nature Materials, 2013, 12(11): 967–977
Whitehead K A, Langer R, Anderson D G. Knocking down barriers: Advances in siRNA delivery. Nature Reviews. Drug Discovery, 2009, 8(2): 129–138
Liu X, Rocchi P, Peng L. Dendrimers as non-viral vectors for siRNA delivery. New Journal of Chemistry, 2012, 36(2): 256–263
Ravina M, Paolicelli P, Seijo B, Sanchez A. Knocking down gene expression with dendritic vectors. Mini-Reviews in Medicinal Chemistry, 2010, 10(1): 73–86
Tomalia D A, Christensen J B, Boas U. Dendrimers, Dendrons, and Dendritic Polymers: Discovery, Applications, and the Future. London: Cambridge University Press, 2012, 100–105
Walter M V, Malkoch M. Simplifying the synthesis of dendrimers: Accelerated approaches. Chemical Society Reviews, 2012, 41(13): 4593–4609
Tomalia D A B H, Dewald J, Hall M, Kallos G, Martin S, Roeck J, Ryder J, Smith P. A new class of polymers: Starburst-dendritic macromolecules. Polymer Journal, 1985, 17(1): 117–132
Haensler J, Szoka F C. Polyamidoamine cascade polymers mediate efficient transfection of cells in culture. Bioconjugate Chemistry, 1993, 4(5): 372–379
Kukowska-Latallo J F, Bielinska A U, Johnson J, Spindler R, Tomalia D A, Baker J R. Efficient transfer of genetic material into mammalian cells using Starburst polyamidoamine dendrimers. Proceedings of the National Academy of Sciences of the United States of America, 1996, 93(10): 4897–4902
Eichman J D, Bielinska A U, Kukowska-Latallo J F, Baker J R Jr. The use of PAMAM dendrimers in the efficient transfer of genetic material into cells. Pharmaceutical Science & Technology Today, 2000, 3(7): 232–245
Guillot-Nieckowski M, Eisler S, Diederich F. Dendritic vectors for gene transfection. New Journal of Chemistry, 2007, 31(7): 1111–1127
Mintzer M A, Simanek E E. Nonviral vectors for gene delivery. Chemical Reviews, 2009, 109(2): 259–302
Behr J P. The proton sponge: A trick to enter cells the viruses did not exploit. CHIMIA International Journal for Chemistry, 1997, 51(1-2): 34–36
Liu X, Liu C, Catapano C V, Peng L, Zhou J, Rocchi P. Structurally flexible triethanolamine-core poly(amidoamine) dendrimers as effective nanovectors to deliver RNAi-based therapeutics. Biotechnology Advances, 2014, 32(4): 844–852
Biswas S, Torchilin V. Dendrimers for siRNA delivery. Pharmaceuticals, 2013, 6(2): 161–183
Kang H, De Long R, Fisher M H, Juliano R L. Tat-conjugated PAMAM dendrimers as delivery agents for antisense and siRNA oligonucleotides. Pharmaceutical Research, 2005, 22(12): 2099–2106
Zhou J, Wu J, Hafdi N, Behr J P, Erbacher P, Peng L. PAMAM dendrimers for efficient siRNA delivery and potent gene silencing. Chemical Communications, 2006, 22: 2362–2364
Wu J, Zhou J, Qu F, Bao P, Zhang Y, Peng L. Polycationic dendrimers interact with RNA molecules: Polyamine dendrimers inhibit the catalytic activity of Candida ribozymes. Chemical Communications, 2005, 3: 313–315
Venkatesh S, Workman J L. Histone exchange, chromatin structure and the regulation of transcription. Nature Reviews. Molecular Cell Biology, 2015, 16(3): 178–189
Liu X, Wu J, Yammine M, Zhou J, Posocco P, Viel S, Liu C, Ziarelli F, Fermeglia M, Pricl S, et al. Structurally flexible triethanolamine core PAMAM dendrimers are effective nanovectors for DNA transfection in vitro and in vivo to the mouse thymus. Bioconjugate Chemistry, 2011, 22(12): 2461–2473
Liu X, Liu C, Laurini E, Posocco P, Pricl S, Qu F, Rocchi P, Peng L. Efficient delivery of sticky siRNA and potent gene silencing in a prostate cancer model using a generation 5 triethanolamine-core PAMAM dendrimer. Molecular Pharmaceutics, 2012, 9(3): 470–481
Posocco P, Liu X, Laurini E, Marson D, Chen C, Liu C, Fermeglia M, Rocchi P, Pricl S, Peng L. Impact of siRNA overhangs for dendrimer-mediated siRNA delivery and gene silencing. Molecular Pharmaceutics, 2013, 10(8): 3262–3273
Shen X C, Zhou J, Liu X, Wu J, Qu F, Zhang Z L, Pang D W, Quelever G, Zhang C C, Peng L. Importance of size-to-charge ratio in construction of stable and uniform nanoscale RNA/dendrimer complexes. Organic & Biomolecular Chemistry, 2007, 5(22): 3674–3681
Liu X, Rocchi P, Qu F Q, Zheng S Q, Liang Z C, Gleave M, Iovanna J, Peng L. PAMAM dendrimers mediate siRNA delivery to target Hsp27 and produce potent antiproliferative effects on prostate cancer cells. ChemMedChem, 2009, 4(8): 1302–1310
Liu C, Liu X, Rocchi P, Qu F, Iovanna J L, Peng L. Arginineterminated generation 4 PAMAM dendrimer as an effective nanovector for functional siRNA delivery in vitro and in vivo. Bioconjugate Chemistry, 2014, 25(3): 521–532
Liu X, Liu C, Chen C, Bentobji M, Cheillan F A, Piana J T, Qu F, Rocchi P, Peng L. Targeted delivery of Dicer-substrate siRNAs using a dual targeting peptide decorated dendrimer delivery system. Nanomedicine; Nanotechnology, Biology, and Medicine, 2014, 10(8): 1627–1636
Reebye V, Sætrom P, Mintz P J, Huang K W, Swiderski P, Peng L, Liu C, Liu X, Lindkær-Jensen S, Zacharoulis D, et al. Novel RNA oligonucleotide improves liver function and inhibits liver carcinogenesis in vivo. Hepatology (Baltimore, MD.), 2014, 59(1): 216–227
Kala S, Mak A S C, Liu X, Posocco P, Pricl S, Peng L, Wong A S T. Combination of dendrimer-nanovector-mediated small interfering RNA delivery to target akt with the clinical anticancer drug paclitaxel for effective and potent anticancer activity in treating ovarian cancer. Journal of Medicinal Chemistry, 2014, 57(6): 2634–2642
Cui Q, Yang S, Ye P, Tian E, Sun G, Zhou J, Sun G, Liu X, Chen C, Murai K, et al. Downregulation of TLX induces TET3 expression and inhibits glioblastoma stem cell self-renewal and tumorigenesis. Nature Communications, 2016, 7: 10637–10651
Lang MF, Yang S, Zhao C, Sun G, Murai K, Wu X, Wang J, Gao H, Brown C E, Liu X, et al. Genome-wide profiling identified a set of miRNAs that are differentially expressed in glioblastoma stem cells and normal neural stem cells. PLoS One, 2012, 7(4): e36248–e36251
Zhou J, Neff C P, Liu X, Zhang J, Li H, Smith D D, Swiderski P, Aboellail T, Huang Y, Du Q, et al. Systemic administration of combinatorial dsiRNAs via nanoparticles efficiently suppresses HIV-1 infection in humanized mice. Molecular Therapy, 2011, 19(12): 2228–2238
Svenson S. The dendrimer paradox—High medical expectations but poor clinical translation. Chemical Society Reviews, 2015, 44(12): 4131–4144
Yu T, Liu X, Bolcato-Bellemin A L, Wang Y, Liu C, Erbacher P, Qu F, Rocchi P, Behr J P, Peng L. An amphiphilic dendrimer for effective delivery of small interfering RNA and gene silencing in vitro and in vivo. Angewandte Chemie International Edition in English, 2012, 51(34): 8478–8484
Chen C, Posocco P, Liu X, Cheng Q, Laurini E, Zhou J, Liu C, Wang Y, Tang J, Col V D, et al. Mastering dendrimer self-assembly for efficient siRNA delivery: From conceptual design to in vivo efficient gene silencing. Small, 2016, 12(27): 3667–3676
Márquez-Miranda V, Araya-Durán I, Camarada M B, Comer J, Valencia-Gallegos J A, González- Nilo F D. Self-assembly of amphiphilic dendrimers: The role of generation and alkyl chain length in siRNA interaction. Scientific Reports, 2016, 6: 29436–29451
Liu X, Liu C, Zhou J, Chen C, Qu F, Rossi J J, Rocchi P, Peng L. Promoting siRNA delivery via enhanced cellular uptake using an arginine-decorated amphiphilic dendrimer. Nanoscale, 2015, 7(9): 3867–3875
Nakase I, Akita H, Kogure K, Gräslund A, Langel Ü, Harashima H, Futaki S. Efficient intracellular delivery of nucleic acid pharmaceuticals using cell-penetrating peptides. Accounts of Chemical Research, 2012, 45(7): 1132–1139
Liu X, Zhou J, Yu T, Chen C, Cheng Q, Sengupta K, Huang Y, Li H, Liu C, Wang Y, et al. Adaptive amphiphilic dendrimer-based nanoassemblies as robust and versatile siRNA delivery systems. Angewandte Chemie International Edition in English, 2014, 53(44): 11822–11827
Percec V, Wilson D A, Leowanawat P, Wilson C J, Hughes A D, Kaucher M S, Hammer D A, Levine D H, Kim A J, Bates F S, et al. Self-assembly of Janus dendrimers into uniform dendrimersomes and other complex architectures. Science, 2010, 328(5981): 1009–1014
Liu X, Wang Y, Chen C, Tintaru A, Cao Y, Liu J, Ziarelli F, Tang J, Guo H, Rosas R, et al. A fluorinated bola-amphiphilic dendrimer for on-demand delivery of siRNA, via specific response to reactive oxygen species. Advanced Functional Materials, 2016, 26(47): 8594–8603
Gorrini C, Harris I S, Mak TW. Modulation of oxidative stress as an anticancer strategy. Nature Reviews. Drug Discovery, 2013, 12(12): 931–947
Trachootham D, Alexandre J, Huang P. Targeting cancer cells by ROS-mediated mechanisms: A radical therapeutic approach? Nature Reviews. Drug Discovery, 2009, 8(7): 579–591
Valentine D L. Adaptations to energy stress dictate the ecology and evolution of the Archaea. Nature Reviews. Microbiology, 2007, 5(4): 316–323
Chen H, Viel S, Ziarelli F, Peng L. 19F NMR: A valuable tool for studying biological events. Chemical Society Reviews, 2013, 42(20): 7971–7982
Acknowledgements
We acknowledge support from La Ligue Nationale Contre le Cancer (LP), Fondation pour la Recherche Médicale (SPF20150934261, YC), Association pour la Recherche sur les Tumeurs de la Prostate (LP, XL), Association Française contre les Myopathies (XL), the international ERA-Net EURONANOMED European Research projects “DENANORNA”, “Target4Cancer” and “NANOGLIO” (LP), Agence Nationale de la Recherche, CNRS, Aix-Marseille University and China Pharmaceutical University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dr. Ling Peng is currently a research director at the French National Scientific Research Center (CNRS) and a group leader at the Interdisciplinary Center of Nanoscience in Marseille (CINaM), France. She carried out her undergraduate study in polymer science with Prof. Chen Rongshi at Nanjing University in China, her Ph.D. program in organic chemistry with Prof. Albert Eschenmoser at the Swiss Federal Institute of Technology in Zurich, Switzerland, and her postdoctoral research in pharmaceutical science with Prof. Maurice Goeldner at the Louis Pasteur University of Strasbourg in France. She was recruited as a research scientist at CNRS in 1997, and promoted to CNRS research director in 2008. She is working actively at the interface of chemistry and biology, and mainly focused on developing functional dendrimers for drug delivery, and molecular probes for understanding and exploring biological events for drug discovery. In particular, she has pioneered bio-inspired structurally flexible dendrimers for nucleic acid delivery in gene therapy, and inaugurated the concept of self-assembling supramolecular dendrimers for drug delivery. Dr. Ling Peng is currently a member of the Editorial Advisory Board of Molecular Pharmaceutics, section editor of drug delivery for Current Medicinal Chemistry and Assistant Editor of New Journal of Chemistry.
Rights and permissions
About this article
Cite this article
Cao, Y., Liu, X. & Peng, L. Molecular engineering of dendrimer nanovectors for siRNA delivery and gene silencing. Front. Chem. Sci. Eng. 11, 663–675 (2017). https://doi.org/10.1007/s11705-017-1623-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11705-017-1623-5