Abstract
The projected increase in utilization rates of total hip arthroplasty (THA) has created an emphasis on novel technologies that can aid providers in maintaining historically positive outcomes. Widespread utilization of robotic assisted THA (RA-THA) is contingent upon achieving favorable outcomes compared to its traditional manual counterpart (mTHA). Therefore, the purpose of our systematic review was to compare RA-THA and mTHA in terms of the following: (1) functional outcomes and (2) complication rates. The PubMed, Embase, and Cochrane library databases were searched for articles published October 1994 and May 2021 comparing functional outcomes and complication rates between RA-THA and mTHA cohorts. When three or more studies evaluated certain PROMs and complications, a pooled analysis utilizing Mantel–Haenszel (M–H) models was conducted utilizing data from final follow-up. Our final analysis included 18 studies which reported on a total of 2811 patients [RA-THA: n = 1194 (42.48%); mTHA: n = 1617 (57.52%)]. No significant differences were demonstrated for a majority of pooled analyses and when segregating by robotic system. Only WOMAC scores were significantly lower among RA-THA patients (p = 0.0006). For outcomes without sufficient data for a pooled analysis, there were no significant differences reported among included studies. The growing utilization of RA-THA motivates comparisons to its manual counterpart. Collectively, we found comparable functional outcomes and complication profiles between RA-THA and mTHA cohorts. More randomized controlled trials of higher quality and larger sample sizes are necessary to further strengthen these findings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Due in part to the widespread success of the procedure [1, 2], procedural volume for total hip arthroplasty (THA) is projected to surge in the coming decades [3,4,5,6]. Yet, while a majority of patients are satisfied following the operation [7, 8], there remains a subset of this patient population that suffers from various post-operative complications [9, 10]. Therefore, as providers continually attempt to improve the quality care in light of increasing caseloads [11,12,13], there has been an increasing need for novel methods of maintaining and improving THA outcomes. The implementation of robotic assisted total hip arthroplasty (RA-THA) has extensively been explored in this domain.
While THA procedures utilizing robotic-arm assistance developed in the 1980’s [14, 15], consideration for the widespread use of RA-THA has only recently begun to emerge. Support for these procedures has mainly focused on the potential benefits that RA-THA provides in relation to implant placement and orientation [16, 17]. Specifically, current RA-THA modalities utilize preoperative planning software that aids in limiting unnecessary resection while guiding providers regarding proper alignment of the prosthetic joint components through tactile feedback [18, 19]. Yet, while numerous studies have compared radiologic outcomes and implant placement accuracy between RA-THA and manual THA (mTHA) cohorts [20,21,22,23,24,25,26,27,28,29,30], there is a need for additional information regarding differences in functional outcomes and complication rates between the two methods of joint replacement.
As the use of RA-THA continues to emerge, more information regarding how outcomes in RA-THA cohorts compare to manually performed manual THA patients is needed in order to determine whether robotic assistance can be a viable option for widespread use. Therefore, the purpose of our systematic review is to compare differences in early and mid-term outcomes between RA-THA and mTHA in relation to (1) functional outcomes and (2) complication incidence.
Methods
Search strategy
The PubMed, Embase, and Cochrane library databases were comprehensively searched for all articles that compared functional outcomes between RA-THA and mTHA cohorts published between October 1994 and May 2021. In combination with “AND” or “OR” Boolean operators, the following keywords were utilized: “robotic”; “robotic-assisted”; “THA”; “total hip arthroplasty”; “outcomes”; “patient-reported outcome measures (PROMs); “complications”.
Articles were included if they met the following inclusion criteria: (1) full-text English manuscript was available, (2) controlled prospective or retrospective studies, (3) studies that utilized FDA approved robotic systems for RA-THA procedures, (4) studies reporting on functional outcomes or complications following RA-THA and mTHA. Additionally, exclusion criteria applied to studies consisted of the following: (1) case series or case reports without mTHA controls, (2) studies that did not utilize a robotic-arm, (3) cadaveric studies, (4) studies that did not report on functional outcomes or complications.
Two researchers independently conducted the query. If there was a disagreement regarding article inclusion, the senior author was consulted to determine if the article should be included.
Study selection
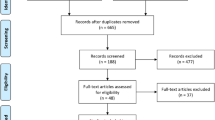
The initial search yielded a total of 526 potentially relevant articles. Following duplication removal, a total of 398 articles remained. After a thorough review of the abstracts, and application of the predetermined inclusion and exclusion criteria, 47 articles were further considered. The full-text manuscripts of these articles were then examined, resulting in 18 articles being further considered. No additional articles were identified following a thorough review of the reference lists of these studies (Fig. 1). Therefore, our final analysis included 18 studies which reported on a total of 2811 patients (RA-THA: n = 1194 (42.48%); mTHA: n = 1617 (57.52%)) (Table 1).
Implemented systems
Two RA-THA modalities were implemented among included studies. ROBODOC (Curexo Tecnology, Fremont, CA) is a fully-active system which autonomously prepares the femoral canal [18]. Conversely, the MAKO system (Stryker Orthopaedics, Mahwah, NJ) is a semi-active system since the surgeon ultimately has control of the resection through tactile and auditory feedback provided by the system [19]. The ROBODOC and MAKO systems were implemented by eight and ten included studies, respectively.
Statistical analysis
When three or more studies evaluated certain PROMs and complications, a pooled analysis utilizing Mantel–Haenszel (M–H) models was conducted utilizing data from final follow-up. Per previous methodology, random-effects models were implemented for pooled analyses whose I2 values were > 50% (high heterogeneity) [31]. Conversely, for articles with either low (I2 = 0–25%) or moderate (I2 = 25–49%), a fixed-effects model was implemented. When possible, pooled analyses were additionally conducted while segregating by the type of implemented robotic system. Significance was determined for p-values < 0.05.
Results
Functional outcomes
Harris Hip Scores
The most commonly reported patient-reported outcome (PRO) was the Harris Hip Score (HSS), with a total of ten studies comparing scores between RA-THA and mTHA cohorts [14, 20, 21, 26, 32,33,34,35,36,37] (Table 2). Pooled analysis demonstrated no significant difference between RA-THA and mTHA (Mean Difference (MD): 5.05, 95% Confidence Interval (CI) − 0.88 to 10.98; p = 0.10) (Fig. 2). No significant differences were similarly demonstrated when evaluating the MAKO (MD: 6.76, 95% CI − 1.23 to 14.75; p = 0.10) and ROBODOC (MD: 1.73, 95% CI − 1.29 to 4.75; p = 0.26) systems independently.
WOMAC
A total of four studies reported patient scores on the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) [20, 21, 26, 36]. Pooled analysis by fixed-effects model demonstrated significantly higher scores among the RA-THA cohorts (MD: − 3.57; 95% CI − 5.62 to − 1.52; p = 0.006) (Fig. 3). There was not enough data to segregate by implemented robotic system.
Forgotten Joint Score
The Forgotten Joint Score (FJS) was implemented by four analyses [35, 37,38,39] (Table 3). All included analyses utilized the MAKO system. There was no significant difference between RA-THA and mTHA patients by random-effects model (MD: 8.73, 95% CI − 4.79 to 22.25; p = 0.21) (Fig. 4).
Pain
A total of four studies evaluated differences in pain between patients undergoing RA- and manual THA utilizing a visual analogy scale (VAS) [20, 35, 37, 38]. A fixed-effects model demonstrated that there were no differences in VAS pain between THA techniques (MD: − 0.19, 95% CI − 0.65 to 0.27; p = 0.41) (Fig. 5). This was similarly demonstrated when isolating cohorts evaluating the MAKO semi-active system (MD: − 0.18, 95% CI − 0.64 to 0.28; p = 0.44).
When utilizing alternative pain measurements, Barger et al. reported pain values significantly higher for the RA-THA patients [20]. Specifically, Health Status Questionnaire (HSQ) pain scores (83.75 ± 20.40 vs. 72.65 ± 16.31; p = 0.019) and modified HHS (mHHS) pain values (41.81 ± 5.05 vs. 39.09 ± 7.37; p = 0.025) were higher for their RA-THA cohort.
Merle d’Aubigné
The Merle d’Aubigné (MDA) hip score was utilized by three studies that evaluated the ROBODOC fully active system [32, 40, 41] (Table 4). No significant difference was seen between THA cohorts by fixed-effects model (MD: 0.24, 95% CI − 0.05 to 0.52; p = 0.10) (Fig. 6).
Short-Form
A Short-Form survey (SF) was implemented by three difference analyses [14, 21, 35]. Similar to other reported PROs, there was no consensus regarding whether manual THA or RA-THA yielded superior outcomes. For the ROBODOC system, Bargar et al. [14] reported no significant difference in Short-Form 36 (SF-36) scores between the robotic THA and the manual THA groups up to 24-month follow-up.
This was similarly demonstrated by Bukowski et al. while utilizing the MAKO system at 1 year post-operatively for SF-12 values [21]. Furthermore, although Domb et al. found significantly higher SF-12 physical scores among their RA-THA cohort at minimum 5-year follow-up (50.30 ± 8.83 vs. mTHA: 45.92 ± 9.44; p = 0.002) [35], no differences were found between cohorts for SF-12 mental scores (p = 0.17).
UCLA
Two studies reported on the University of California Los Angeles (UCLA) physical activity scale [20, 21]. As with the previously discussed outcomes, there was no agreement between these analyses. While Bargar et al. [20] found no difference between RA-THA (6.09 ± 1.89) and mTHA (5.71 ± 1.45; p = 0.417) cohorts, Bukowski et al. [21] contrarily demonstrated that the RA-THA cohort achieved higher mean postoperative UCLA (6.3 ± 1.8 vs. 5.8 ± 1.7; p = 0.033).
Additional PROs
Various additional PROs were reported across the included studies. As a whole, there was mixed evidence regarding the differences between cohort scores. Results of these remaining PROs are available in Table 5.
Gait
While gait pattern and walking speed are universal parameters of assessing the functional capacity [42, 43], only two studies reported on gait-related outcomes [29, 40].
In their study of ROBODOC assisted procedures, Nishihara et al. [40] found no difference between the RA-THA (mean: 14 days, range 7–31 days) and mTHA cohorts (mean: 16 days, range 7–46 days) in the amount of time since surgery required for the patient to achieve independent ambulation of at least 500 m without using a cane (p = 0.0552). Moreover, the number of patients who were able to walk more than 6 blocks without using a cane within 13 days of the procedure was significantly greater for the RA-THA cohort (41 vs. 28; p < 0.05).
Utilizing the MAKO system, Peng et al. [29] analyzed the range of motion, walking speed, and gait mechanics in patients who received robotic and manual THA and compared each to the native contralateral hip mechanics. In the RA-THA cohort, they found no difference in peak range of motion in the frontal or axial planes (p > 0.05). However, net sagittal plane range of motion was significantly reduced (mean (± SD) difference: − 3.0 ± 4.9°; p = 0.043). Contrarily, no significant difference in net range of motion was found for the mTHA cohort in any direction (p > 0.05). The authors additionally demonstrated no significant difference between the two groups in terms of walking speed (RA-THA = 1.91 ± 0.49, range 0.5–2.9; mTHA = 1.76 ± 0.34, range 1.0–2.6 mph; p = 0.262). Finally, there was no difference in the degree of gait asymmetry between the RA-THA and mTHA cohorts.
Complications
Revision rate
Revision rates between RA-THA and mTHA cohorts were comparable for a majority of studies. By pooled analysis, there was no significant difference between all cohorts (MD: 0.73, 95% CI 0.26–2.03; p = 0.54) (Fig. 7). This was similarly demonstrated for studies only evaluating MAKO systems (MD: 0.56, 95% CI 0.23–1.38; p = 0.21). All reported revision rates are available in Table 6.
Dislocation rate
No significant differences in dislocation rates were found between cohorts when including studies evaluating both ROBODOC and MAKO systems (Odds Ratio (OR): 1.81, 95% CI 0.71–4.58; p = 0.02) (Fig. 8; Table 7). This was similarly demonstrated when evaluating MAKO studies independently (OR: 0.66, 95% CI 0.15–3.02; p = 0.60). However, when evaluating only ROBODOC studies, there was a significantly higher rate of dislocations among RA-THA cohorts (OR: 2.87, 95% CI 1.07–7.07; p = 0.04).
Infection
There is a significant scarcity of evidence in literature in terms of comparing the incidence of infection after RA-THA versus mTHA. Within 5-year follow-up, Domb et al. reported two superficial infections in their RA-THA cohort while no infections were found in mTHA patients [35]. Similarly, Perets et al. [37] reported a higher incidence of superficial infections in their RA-THA cohort (n = 6) compared to mTHA controls (n = 2). However, this difference was not significant (p = 0.15).
Conversely, while one mTHA patient suffered from infection in the analysis by Kong et al., no infection was reported in their RA-THA cohort [34]. Similarly, Honl et al. [32] reported two revision surgeries due to deep infection within their mTHA cohort while none of the patients in the RA-THA cohort developed an infection requiring reoperation. This was similarly demonstrated by Kamara et al. [30] who reported two reoperations for infection in their mTHA cohort; one for a superficial soft tissue infection that was resistant to local wound care and the other a deep hematogenous infection over a year post operatively.
Nerve injury
Reports of nerve injury were similarly not common in the identified comparative studies included in this review since most of the included studies did experience any cases of nerve injury in either cohorts. Honl et al. [32] reported an incidence of partial lesion of the peroneal division of the sciatic nerve in four cases (7%) in the RA-THA cohort. An insignificantly lower rate of nerve injury was identified in their mTHA cohort with only one case of partial femoral nerve injury identified (p = 0.31). Additionally, Domb et al. reported one case of sciatic nerve injury among mTHA patients as well as three cases of thigh numbness [35]. Furthermore, Perets et al. reported two cases of lateral femoral cutaneous (LFCN) injury and one case of incisional numbness in their mTHA cohort [37]. No complications related to nerve injury were reported for RA-THA patients in either of these studies.
Intraoperative femoral fractures
Bargar et al. [14] reported three intraoperative femoral fractures (7%) in their mTHA group. No fractures were seen in their RA-THA cohort. Similar results were observed by Hananouchi et al. [41], who demonstrated insignificant differences in fracture incidence between the RA-THA (0, 0%) and mTHA (2, 7%) cohorts (p = 0.21). This was additionally found by Lim et al. [26] (RA-THA: 0% vs. mTHA: 8%; p < 0.05) and Kamara et al. [30] (RA-THA: 0 (0%) vs. mTHA: 3 (2%); p > 0.05). Conversely, Nishihara et al. [40] reported a significantly lower intraoperative fractures rate their RA-THA cohort (n = 0, 0%) compared to their mTHA cohort (n = 5, 7%; p < 0.05).
Periprosthetic fractures
In their 14-year follow-up, Bargar et al. [20], reported one periprosthetic fracture in their RA-THA cohort that occurred 2 years postoperatively and one periprosthetic fracture 3 years postoperatively in their mTHA cohort. The only othe periprosthetic factor reported among included studies occurred in the mTHA cohort of Nakamura et al. [27]
Heterotopic ossification (HO)
Three studies evaluated the incidence of heterotopic ossification (HO) following THA, with all studies agreeing there was no difference found between RA-THA and mTHA cohorts.
Within a follow-up period of 24 months, Honl et al. [32] reported a 10% prevalence of Brooker [44] grade 2 or 3 HO in both the RA-THA and mTHA cohorts (p = 0.31). Similarly, Nakamura et al. [27] did not find any significant difference in the incidence of HO between their RA-THA and mTHA cohorts within their 67-month follow-up period. Specifically, a rate of HO for Grade 1 (11% vs. 11%), 2 (7% vs. 1%), and 3 (0% vs. 3%) was observed for the RA-THA and mTHA cohorts, respectively (p = 0.1). These patients similarly demonstrated insignificant differences in HO rates at longer follow-up (p = 0.2) [45].
Persistent pain
Nakamura et al. [27] found one case (1%) of persistent thigh pain and two cases (3%) of persistent knee pain at the ROBODOC navigation pin insertion site for the RA-THA cohort. However, these resolved at 1 year and 1 month, respectively. In their mTHA cohort, persistent thigh pain was reported in four cases (5.6%) with spontaneous resolution at 1 year.
Additionally, Nishihara et al. [40] reported that four (5.1%) patients in their RA-THA cohort had persistent thigh pain, with two cases resolving within the 3-month postoperative period. On the other hand, the mTHA cohort had 11 (14%) patients who reported persistent thigh pain at 1 month postoperatively (p = 0.0573), a number that decreased to three (4%) patients at 3 months (p = 0.6494).
Discussion
As the implementation of RA-THA continues to be considered among adult reconstructive surgeons, information regarding how robotic assistance influences surgical outcomes has become increasingly important. While RA-THA may yield superior implant placement, evidence regarding its impact on functional outcomes and complication rates should be considered when evaluating the practicality of RA-THA use. Our systematic review and meta-analysis demonstrated that, as a whole, no significant differences were found between mTHA and RA-THA groups in terms of functional outcomes and gait comparison. Specifically, no significant differences were demonstrated for a majority of pooled analyses and when segregating by robotic system. For outcomes without sufficient data for a pooled analysis, there were no significant differences reported among included studies. Additionally, reported complication rates between both cohorts were comparable.
The findings of the present study suggest that functional outcomes may not necessarily be the differentiating factor when considering RA-THA implementation. Rather, consideration of complications such as revision occurrence and dislocation may yield a stronger understanding of RA-THA viability given the economic burden and patient dissatisfaction associated with these adverse events [19, 46]. While there was mixed evidence regarding the rates of these complications between mTHA and RA-THA cohorts, the study by Bargar et al. may shed important insight into true RA-THA outcomes given that their follow-up period (14 years) was the longest among included studies [20]. Specifically, the authors reported no significance difference between cohorts in terms of survivability/revision and dislocation. Since revision may not occur until years after the index procedure [47], the comparable rates demonstrated by this longer term follow-up may more adequately demonstrate the efficacy of RA-THA compared to other shorter-term analyses. However, further studies are needed to better understand this relationship.
Given comparable functional outcomes between cohorts, as well as the low rate of complications reported across studies, evaluating other metrics may shed more light on which THA technique should be considered superior. For example, multiple previous meta-analyses have demonstrated superior radiographic outcomes, including higher rates within the Lewinnek and Callanan safe zones, among RA-THA cohorts [47, 48]. Superior implant placement may mitigate the risk of dislocation and subsequently, the need for early revision THA [16, 17, 49, 50]. Similarly, although reported complications were comparable between cohorts, operative time was found to be significantly longer among patients undergoing RA-THA for a large majority of studies [31, 39]. As prolonged operative times in total joint arthroplasty (TJA) have been associated with an increased risk of adverse outcomes [51,52,53,54,55,56], these findings suggest that RA-THA may carry a higher perioperative complication risk, especially during the learning curve associated with RA-THA implementation. Therefore, while operative time should be considered when evaluating the safety of RA-THA procedures, surgeon proficiency, as well as other confounding factors should be additionally evaluated.
Our study has some limitations. Although it remains unclear how much conflicts of interests (COIs) influenced included studies, there has been a rising concern across the literature regarding how COIs among authors comparing RA-THA and manual THA may bias reported outcomes. Notably, DeFrance et al. found that articles reporting positive findings for RA-THA were more likely to contain authors with COIs related to RA-THA manufacturers [56]. Given the nature of the reported data and the heterogeneity regarding which robotic system was utilized by our included studies, we were unable to conduct a pooled analysis of certain data points. Similarly, instruments utilized to measure PROMs following THA have varied over the past few decades, and thus, there was a large variation in which tools were utilized in each study [57]. Therefore, it remains unclear if certain instruments (i.e. joint-specific vs. joint-agnostic) better serve to detect subtle differences between THA cohorts. As for the included studies themselves, as a whole, there was a limited number of patients included in a majority of the studies. This is especially problematic for studies with longer follow-up intervals that had high rates of patients lost to follow-up [20]. Relatedly, limited longer term follow-up seen in included studies limited our inability to compare early and late revision risk between cohorts. Furthermore, a large proportion of studies failed to control for differences in patient demographics between cohorts that may influence functional outcomes and complication risk, such as comorbidity burden, or baseline PROM values (Table 1).
While RA-THA implementation continues to be considered among orthopedic providers, the present analysis demonstrated a lack of significant improvement regarding functional outcomes following robotic assistance. Similarly, complication risk did not differ compared to manual THA patients. Future studies should continue to evaluate RA-THA in larger cohorts in order to further determine its efficacy and to ensure patient safety for those undergoing RA-THA procedures. These studies should continue to consider outside factors, such as surgeon experience and familiarity with new instrumentation, in these evaluations. The costs associated with RA-THA implementation, from installation of the robotic device to servicing of the computer software, should additionally be considered.
References
Jonsson B, Larsson SE (1991) Functional improvement and costs of hip and knee arthroplasty in destructive rheumatoid arthritis. Scand J Rheumatol 20:351–357. https://doi.org/10.3109/03009749109096811
Liang MH, Cullen KE, Larson MG et al (1986) Cost-effectiveness of total joint arthroplasty in osteoarthritis. Arthritis Rheum 29:937–943. https://doi.org/10.1002/art.1780290801
Kurtz S, Ong K, Lau E et al (2007) Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Jt Surg 89:780. https://doi.org/10.2106/JBJS.F.00222
Singh JA (2011) Epidemiology of knee and hip arthroplasty: a systematic review. Open Orthop J 5:80–85. https://doi.org/10.2174/1874325001105010080
Maradit Kremers H, Larson DR, Crowson CS et al (2015) Prevalence of total hip and knee replacement in the United States. J Bone Jt Surg Am 97:1386–1397. https://doi.org/10.2106/JBJS.N.01141
Sloan M, Premkumar A, Sheth NP (2018) Projected volume of primary total joint arthroplasty in the U.S., 2014 to 2030. J Bone Jt Surg 100:1455–1460. https://doi.org/10.2106/JBJS.17.01617
Krushell R, Bhowmik-Stoker M, Kison C et al (2016) Characterization of patient expectations and satisfaction after total hip arthroplasty. J Long Term Eff Med Implants 26:123–132. https://doi.org/10.1615/JLongTermEffMedImplants.2016012621
Mancuso CA, Salvati EA, Johanson NA et al (1997) Patients’ expectations and satisfaction with total hip arthroplasty. J Arthroplast 12:387
Mahomed NN, Barrett JA, Katz JN et al (2003) Rates and outcomers of primary and revision total hip replacement in the United States medicare population. J Bone Jt Surg Am 85:27–32. https://doi.org/10.2106/00004623-200301000-00005
Abbas K, Murtaza G, Umer M et al (2012) Complications of total hip replacement. J Coll Physicians Surg Pak 22:575–578
Zhan C, Miller MR (2003) Excess length of stay, charges, and mortality attributable to medical injuries during hospitalization. JAMA 290:1868. https://doi.org/10.1001/jama.290.14.1868
Waters TM, Daniels MJ, Bazzoli GJ et al (2015) Effect of medicare’s nonpayment for hospital-acquired conditions. JAMA Intern Med 175:347. https://doi.org/10.1001/jamainternmed.2014.5486
Duchman KR, Pugely AJ, Martin CT et al (2017) Operative time affects short-term complications in total joint arthroplasty. J Arthroplasty 32:1285–1291. https://doi.org/10.1016/j.arth.2016.12.003
Bargar WL, Bauer A, Börner M (1998) Primary and revision total hip replacement using the ROBODOC® system. Clin Orthop Relat Res. https://doi.org/10.1097/00003086-199809000-00011
Bargar WL (2007) Robots in orthopaedic surgery: past, present, and future. Clin Orthop Relat Res 463:31–36
Lewinnek GE, Lewis JL, Tarr R et al (1978) Dislocations after total hip-replacement arthroplasties. J Bone Jt Surg Am 60:217–220
Callanan MC, Jarrett B, Bragdon CR et al (2011) The John Charnley Award: risk factors for cup malpositioning: quality improvement through a joint registry at a tertiary hospital. Clin Orthop Relat Res 469:319–329. https://doi.org/10.1007/s11999-010-1487-1
Bargar WL, Bauer A, Borner M (1998) Primary and revision total hip replacement using the Robodoc system. Clin Orthop Relat Res. https://doi.org/10.1097/00003086-199809000-00011
Subramanian P, Wainwright TW, Bahadori S, Middleton RG (2019) A review of the evolution of robotic-assisted total hip arthroplasty. HIP Int 29:232
Bargar WL, Parise CA, Hankins A et al (2018) Fourteen year follow-up of randomized clinical trials of active robotic-assisted total hip arthroplasty. J Arthroplasty 33:810–814. https://doi.org/10.1016/j.arth.2017.09.066
Bukowski BR, Anderson P, Khlopas A et al (2016) Improved functional outcomes with robotic compared with manual total hip arthroplasty. Surg Technol Int 29:303–308
Domb BG, Redmond JM, Louis SS et al (2015) Accuracy of component positioning in 1980 total hip arthroplasties: a comparative analysis by surgical technique and mode of guidance. J Arthroplasty 30:2208–2218. https://doi.org/10.1016/j.arth.2015.06.059
Domb BG, El Bitar YF, Sadik AY et al (2014) Comparison of robotic-assisted and conventional acetabular cup placement in THA: a matched-pair controlled study hip. Clin Orthop Relat Res 472:329–336. https://doi.org/10.1007/s11999-013-3253-7
El Bitar YF, Stone JC, Jackson TJ et al (2015) Leg-length discrepancy after total hip arthroplasty: comparison of robot-assisted posterior, fluoroscopy-guided anterior, and conventional posterior approaches. Am J Orthop (Belle Mead NJ) 44:265–269
Haddad FS, Kayani B, Huq SS et al (2019) Assuring the long-term total joint arthroplasty. Bone Jt J 101-B:11–18. https://doi.org/10.1302/0301-620x.101b1.bjj-2018-0377.r1
Lim SJ, Ko KR, Park CW et al (2015) Robot-assisted primary cementless total hip arthroplasty with a short femoral stem: a prospective randomized short-Term outcome study. Comput Aided Surg 20:41–46. https://doi.org/10.3109/10929088.2015.1076044
Nakamura N, Sugano N, Nishii T et al (2010) A comparison between robotic-assisted and manual implantation of cementless total hip arthroplasty. Clin Orthop Relat Res 468:1072–1081. https://doi.org/10.1007/s11999-009-1158-2
Kwon Y-M, Tsai T-Y, Dimitriou D, Li J-S (2016) Does haptic robot-assisted total hip arthroplasty better restore native acetabular and femoral anatomy? Int J Med Robot Comput Assist Surg 12:288–295. https://doi.org/10.1002/rcs
Peng Y, Arauz P, Desai P et al (2019) In vivo kinematic analysis of patients with robotic-assisted total hip arthroplasty during gait at 1-year follow-up. Int J Med Robot 15:e2021. https://doi.org/10.1002/rcs.2021
Kamara E, Robinson J, Bas MA et al (2017) Adoption of robotic vs fluoroscopic guidance in total hip arthroplasty: Is acetabular positioning improved in the learning curve? J Arthroplast 32:125–130. https://doi.org/10.1016/j.arth.2016.06.039
Cochrane Training Cochrane Handbook for Systematic Reviews of Interventions | Cochrane Training. https://training.cochrane.org/cochrane-handbook-systematic-reviews-interventions. Accessed 23 Feb 2021
Honl M, Dierk O, Gauck C et al (2003) Comparison of robotic-assisted and manual implantation of a primary total hip replacement: a prospective study. J Bone Jt Surg Ser A 85:1470–1478. https://doi.org/10.2106/00004623-200308000-00007
Kong X, Yang M, Jerabek S et al (2020) A retrospective study comparing a single surgeon’s experience on manual versus robot-assisted total hip arthroplasty after the learning curve of the latter procedure: a cohort study. Int J Surg 77:174–180. https://doi.org/10.1016/j.ijsu.2020.03.067
Kong X, Yang M, Li X et al (2020) Impact of surgeon handedness in manual and robot-assisted total hip arthroplasty. J Orthop Surg Res 15:1–8. https://doi.org/10.1186/s13018-020-01671-0
Domb BG, Chen JW, Lall AC et al (2020) Minimum 5-year outcomes of robotic-assisted primary total hip arthroplasty with a nested comparison against manual primary total hip arthroplasty: a propensity score-matched study. J Am Acad Orthop Surg 28:847–856. https://doi.org/10.5435/JAAOS-D-19-00328
Hadley C, Grossman E, Mont M et al (2020) Robotic-assisted versus manually implanted total hip arthroplasty: a clinical and radiographic comparison - Pubmed. Surg Technol Int 28:371–376
Perets I, Walsh JP, Mu BH et al (2020) Short-term clinical outcomes of robotic-arm assisted total hip arthroplasty: a pair-matched controlled study. Orthopedics. https://doi.org/10.3928/01477447-20201119-10
Clement ND, Gaston P, Bell A et al (2020) Robotic arm-assisted versus manual total hip arthroplasty a propensity score matched cohort study. Bone Jt Res 10:22–30. https://doi.org/10.1302/2046-3758.101.BJR-2020-0161.R1
Singh V, Realyvasquez J, Simcox T et al (2021) Robotics versus navigation versus conventional total hip arthroplasty: does the use of technology yield superior outcomes? J Arthroplasty. https://doi.org/10.1016/j.arth.2021.02.074
Nishihara S, Sugano N, Nishii T et al (2006) Comparison between hand rasping and robotic milling for stem implantation in cementless total hip arthroplasty. J Arthroplasty 21:957–966. https://doi.org/10.1016/j.arth.2006.01.001
Hananouchi T, Sugano N, Nishii T et al (2007) Effect of robotic milling on periprosthetic bone remodeling. J Orthop Res 25:1062–1069. https://doi.org/10.1002/jor.20376
Colgan G, Walsh M, Bennett D et al (2016) Gait analysis and hip extensor function early post total hip replacement. J Orthop 13:171–176. https://doi.org/10.1016/j.jor.2016.03.005
Middleton A, Fritz SL, Lusardi M (2015) Walking speed: the functional vital sign. J Aging Phys Act 23:314–322. https://doi.org/10.1123/japa.2013-0236
Brooker AF, Bowerman JW, Robinson RA, Riley LHJ (1973) Ectopic ossification following total hip replacement. Incidence and a method of classification. J Bone Jt Surg Am 55:1629–1632
Nakamura N, Sugano N, Sakai T, Nakahara I (2018) Does robotic milling for stem implantation in cementless THA result in improved outcomes scores or survivorship compared with hand rasping? results of a randomized trial at 10 years. Clin Orthop Relat Res 476:2169–2173. https://doi.org/10.1097/CORR.0000000000000467
Bozic KJ, Kurtz SM, Lau E et al (2009) The epidemiology of revision total hip arthroplasty in the United States. J Bone Jt Surg Am 91:128–133. https://doi.org/10.2106/JBJS.H.00155
Ulrich SD, Seyler TM, Bennett D et al (2008) Total hip arthroplasties: what are the reasons for revision? Int Orthop 32:597–604. https://doi.org/10.1007/s00264-007-0364-3
Kessler S, Kinkel S, Käfer W et al (2003) Influence of operation duration on perioperative morbidity in revision total hip arthroplasty. Acta Orthop Belg 69:328–333
Namba RS, Inacio MCS, Paxton EW (2013) Risk factors associated with deep surgical site infections after primary total knee arthroplasty: an analysis of 56,216 knees. J Bone Jt Surg Am 95:775–782. https://doi.org/10.2106/JBJS.L.00211
Peersman G, Laskin R, Davis J et al (2006) Prolonged operative time correlates with increased infection rate after total knee arthroplasty. HSS J 2:70–72. https://doi.org/10.1007/s11420-005-0130-2
Horlocker TT, Hebl JR, Gali B et al (2006) Anesthetic, patient, and surgical risk factors for neurologic complications after prolonged total tourniquet time during total knee arthroplasty. Anesth Analg 102:950–955. https://doi.org/10.1213/01.ane.0000194875.05587.7e
Kurtz SM, Ong KL, Lau E et al (2010) Prosthetic joint infection risk after TKA in the medicare population. Clin Orthop Relat Res 468:52–56. https://doi.org/10.1007/s11999-009-1013-5
Pulido L, Ghanem E, Joshi A et al (2008) Periprosthetic joint infection: the incidence, timing, and predisposing factors. Clin Orthop Relat Res 466:1710–1715. https://doi.org/10.1007/s11999-008-0209-4
O’Malley NT, Fleming FJ, Gunzler DD et al (2012) Factors independently associated with complications and length of stay after hip arthroplasty. J Arthroplasty 27:1832–1837. https://doi.org/10.1016/j.arth.2012.04.025
Russo MW, Macdonell JR, Paulus MC et al (2015) Increased complications in obese patients undergoing direct anterior total hip arthroplasty. J Arthroplasty 30:1384–1387. https://doi.org/10.1016/j.arth.2015.03.002
DeFrance M, Yayac M, Courtney PM, Squire M (2020) The impact of author financial conflicts on robotic-assisted joint arthroplasty research. J Arthroplasty. https://doi.org/10.1016/j.arth.2020.10.033
Lan RH, Bell JW, Samuel LT, Kamath AF (2021) Outcome measures in total hip arthroplasty: have our metrics changed over 15 years? Arch Orthop Trauma Surg 1:1–10. https://doi.org/10.1007/S00402-021-03809-Z
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A.F.K. reports the following disclosures: research support (Signature Orthopaedics), paid presenter or speaker (DePuy Synthes and Zimmer Biomet), paid consultant (DePuy Synthes and Zimmer Biomet), stock or stock options (Zimmer Biomet, Johnson & Johnson, and Procter & Gamble), IP royalties (Innomed), and board or committee member (AAOS, AAHKS, and Anterior Hip Foundation). A.J.A., L.T.S., B.M., and A.K.E. have nothing to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Samuel, L.T., Acuña, A.J., Mahmood, B. et al. Comparing early and mid-term outcomes between robotic-arm assisted and manual total hip arthroplasty: a systematic review. J Robotic Surg 16, 735–748 (2022). https://doi.org/10.1007/s11701-021-01299-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11701-021-01299-0