Abstract
Background
Bariatric surgery has been applied for weight loss and comorbidity control in China since 2000. Recent studies have shown positive results for bariatric surgery in patients with a body mass index (BMI) of less than 35 kg/m2. However, the effect of surgery on Chinese patients with type II diabetes mellitus (T2DM) has not yet been systematically investigated.
Methods
A comprehensive literature search was performed in the Cochrane Library, Embase, PubMed, and Web of Science from January 2014 to March 2020. All studies examined bariatric surgery outcomes on Chinese patients at 12-, 36-, and 60-month follow-up. The research followed the guidance of Preferred Reporting Items for Systematic Reviews and Meta-Analyses Protocols (PRISMA-P) recommendations.
Results
Eleven studies containing 611 patients were included in this meta-analysis. Clinical indices at 12-, 36-, and 60-month follow-up were analyzed. Significant decreases were identified in body weight, BMI, waist circumference (WC), blood pressure (BP), fasting plasma glucose (FPG), glycosylated hemoglobin A1c (hemoglobin A1c, or HbA1c), triglyceride (TG), low-density lipoprotein (LDL), and high-density lipoprotein (HDL) postoperatively. An increasing trend in the T2DM remission rate was discovered. The remission group was observed to have significantly lower HbA1c and C-peptide level, a shorter duration of T2DM, and a higher BMI than the nonremission group at 12 months.
Conclusions
Bariatric surgery successfully provided significant BMI control as well as a reduction and normalization of glucose- and lipid-related metabolism at 12, 36, and 60 months postoperatively in Chinese patients with T2DM with a preoperative BMI of less than 35 kg/m2. An increasing trend in the T2DM remission rate suggested promising future applications in this population.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Type II diabetes mellitus (T2DM), a universal burden on both health care and life quality, has a complicated pathophysiology that combines mixed genetic and environmental factors. It is estimated that by 2040, 642 million people will be affected by T2DM [1]. The treatment first became an area of research interest in developed Western countries. Early in 1995, Pories et al. mentioned that bariatric surgery was more effective in T2DM control than conventional medical therapy [2]. When conservative methods are no longer effective, bariatric surgery serves as the possible treatment for T2DM.
During past 20 years, there has been rising concern over the prevalence of T2DM in eastern China, such as Beijing, Guangzhou, and Shanghai, as most bariatric surgeries have been performed in these areas. At the same time, a 148.7-fold increase in the quantity of bariatric surgeries during 2011–2015 were recorded compared with 2001–2005, most of which were for relieving metabolic syndrome [3]. On the one hand, research showed that only 10% of T2DM patients have comorbid obesity in China, compared with one-third of the T2DM population in the USA [4] and the mean BMI of Chinese patients with T2DM is 25 kg/m2, according to the China Guidelines for Type II Diabetes. On the other hand, the National Institutes of Health (NIH) recommends patients with BMI larger than 40 or 35 kg/m2 with comorbidities to undergo bariatric surgery and it has been well established that bariatric surgery promotes remarkable remission of T2DM for this group.
Therefore, many patients in China suffering from T2DM with BMI lower than 35 kg/m2 do not meet the criteria of bariatric surgery. With regard to these patients, exploring the effect of bariatric surgery on them is of crucial significance. This meta-analysis is mainly focused on surgical outcomes published in the last 5 years, on the basis of developments over the last 10 years. The goal of this study was to obtain a summary of the efficacy of bariatric surgery at 12, 36, and 60 months for Chinese patients with T2DM initial BMI < 35 kg/m2.
Material and Methods
Search Strategy
We performed a comprehensive search of the literature in PubMed, Web of Science, Cochrane Library, and Embase from January 1, 2014, to March 31, 2020. The terms used for article matching were ((BARIATRIC SURGERY[MeSH]) AND (gastric bypass or sleeve gastrectomy effect) and (China) and (ENGLISH[Language])) AND (diabetes mellitus[MeSH]).
Inclusion and Exclusion Criteria
The following criteria were applied for inclusion: (1) Chinese patients with T2DM whose BMI < 35 kg/m2; (2) clearly defined study outcomes and diagnosis of comorbidities; (3) latest follow-up data at the same institution and written in English; and (4) sample size larger than 10 with at least a 1-year follow-up. The exclusion criteria were as follows: (1) failure to classify patients by BMI or the use of classification criteria that extend across 35 kg/m2 (for example, BMI > 32.5 kg/m2 will be excluded but BMI < 32.5 kg/m2 will be included.); (2) any type of study other than original follow-up studies such as reports, reviews, or abstracts; and (3) unrepeatable papers, i.e., showing results only in figures.
Data Extraction and Quality Assessment
After the search was conducted and the appropriate literature was selected, two researchers independently extracted data from the included literature. A third researcher checked the fidelity of both researchers’ data to reduce avoidable error. The following information was extracted from each article: first author, publication year, study design, surgery type, sample size, gender, age, comorbidity, follow-up duration, and surgical complications. Study outcomes including weight, BMI, waist circumference (WC), blood pressure (BP), remission of comorbidities, and other metabolic values were recorded.
A quality and risk-of-bias assessment was applied with the Newcastle-Ottawa Scale (NOS), a 9-star scoring system including patient selection, comparability, and outcome. The regression-based Egger’s test and a nonparametric rank correlation (Begg’s) test were performed to evaluate publication bias.
Statistical Analysis
Review Manager version 5.3 was used for meta-analysis, and publication bias was assessed with Stata 16. Continuous value changes were calculated as the mean difference (MD) and 95% confidence interval (CI). For dichotomous data, odds ratios (ORs) with 95% CIs were calculated. To evaluate heterogeneity among studies, the chi-square test was deployed and a Higgins I2 greater than 50% was considered to indicate significant heterogeneity. When significant heterogeneity was present, a random-effects model was applied; otherwise, a fixed-effects model was used. The overall effect was estimated by z-test, with significance defined as P < 0.05.
Results
Search Results
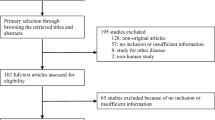
The search process is shown in Fig. 1. Eleven studies that met the inclusion criteria are listed in Table 1, eight [5,6,7,8,9,10,11,12] of which were carried out in university teaching hospitals in mainland China and three [13,14,15] of which were conducted in Taiwan. Seven of the 11 studies were retrospective and the remaining four were prospective.
Quality Assessments
Quality assessments are shown in Table 2; all the studies were evaluated with the NOS.
Systematic Review
We identified eleven studies with 611 patients with T2DM who had a mean age of 47.2 years. Most of the studies reported laparoscopic Roux-en-Y gastric bypass (LRYGB) [5,6,7,8, 10, 11, 13, 15], and the remaining studies reported laparoscopic sleeve gastrectomy (LSG) or both procedures. The mean T2DM duration was 6.8 years, and the average baseline BMI, glycosylated hemoglobin A1c (hemoglobin A1c, or HbA1c), fasting plasma glucose (FPG), triglyceride (TG), high-density lipoprotein (HDL), and low-density lipoprotein (LDL) were 27.9 kg/m2, 8.4%, 9.5 mmol/L, 2.51 mmol/L, 1.1 mmol/L, and 2.83 mmol/L, respectively. According to the American Diabetes Association remission criteria [16], complete remission of T2DM was defined as HbA1c less than 6%, FPG less than 5.6 mmol/L, and no use of diabetes medication for a year. Partial remission was defined as an FPG of 5.6 to 6.9 mmol/L for at least 1 year in the absence of active pharmacological therapy. In this meta-analysis, six studies [5, 6, 10,11,12, 14] followed the complete remission criteria, one [13] did not apply FPG < 5.6 mmol/L, and one [15] with only HbA1c < 6%. The remaining two studies [8, 9] used a threshold of HbA1c < 6.5% and without medication for a year. In this study, combined complete and partial remission were pooled together as T2DM remission.
Meta-analysis
BMI, WC, and BP are shown in Table 3. A significant reduction in BMI occurred at the 12-, 36-, and 60-month visits, with MD values of − 4.97 kg/m2 (95% CI − 6.03 to − 3.92), − 3.50 kg/m2 (95% CI − 7.55 to − 0.55), and − 4.96 kg/m2 (95% CI − 7.15 to − 2.77). Heterogeneity was present in these three outcomes and a random-effects model was used. Fewer than half of these studies included WC, especially in the long-term results, and heterogeneity was not present at the 60-month follow-up, at which time the value was − 14.75 cm (95% CI − 17.5 to − 12). The value was smaller than the WC of − 17.3 cm (95% CI − 20.37 to − 14.23) observed at 12 months and larger than the WC of − 13.97 cm (95% CI − 16.23 to − 11.71) observed at 36 months. A fixed-effects model was used for 60-month WC. Significant decreases in both systolic and diastolic BP were detected, and no significant heterogeneity was found; thus, a fixed-effects model was used.
Outcomes of T2DM are analyzed in Table 4. There was a significant reduction in FPG at each follow-up visit compared to baseline. The MD was − 3.68 mmol/L (95% CI − 3.84 to − 3.52), − 1.78 mmol/L (95% CI − 2.32 to − 1.24), and − 2.18 mmol/L (95% CI − 4.28 to − 0.09). A fixed-effects model was used for the 36-month results. For all the patients, a significant decrease in HbA1c was achieved; the difference was especially notable at 12 months, with a result of − 2.12% (95% CI − 2.5 to − 1.73), which was larger in magnitude than the 36-month or 60-month results. A fixed-effects model was applied to fasting insulin and C-peptide levels at 60 months. Homeostasis model assessment for insulin resistance (HOMA-IR) was reported only at the 12-month result and no significant result was detected. Ten studies included data on T2DM remission. The remission rates at 12, 36, and 60 months were 42.4%, 50.7%, and 53.1%, respectively. In an analysis of three papers [12, 15, 17] comparing baseline indices between remission and nonremission patients which were analyzed, significant differences were recognized in preoperative BMI, HbA1c, C-peptide, and T2DM duration (Table 5).
Lipid metabolism is addressed in Table 6. Six papers reported a significant change in TG, and the results showed a − 1.21 mmol/L (95% CI − 2.01 to − 0.5) and − 1.04 mmol/L (95% CI − 1.94 to − 1.21) decrease at 12 and 36 months, respectively. One paper reported a TG reduction of 1.58 mmol/L at 60 months. Heterogeneity was low at the 36-month study, and a fixed-effects model was used. Given a relatively high preoperative LDL, significantly reduced LDL levels were achieved at 12, 36, and 60 months postoperatively. Low heterogeneity was found at 36 and 60 months and a fixed effect model was used. The HDL level rapidly increased at the 12-month follow-up but dropped later.
Safety and adverse reactions are both important outcomes to observe. Overall, 10 of 11 studies reported complications and mortality. The mortality rate was 0.3% and complication rate was 15.9% including 13 cases of major complications (2.4%). In the Swedish obese subject (SOS) study, in which patients were morbidly obese, the major complication and mortality rate were 2.2% and 0.2%.
Additionally, the efficacy of Roux-en-Y gastric bypass (RYGB) and conservative medical treatment was compared in Table 7. Significantly reduced values of BMI, weight, systolic pressure, diastolic pressure, HbA1c, FPG, and TG were observed in the RYGB group. The remission rate of T2DM was significantly higher in the RYGB group than in the conservative treatment group, with an OR of 37.86 (95% CI 16.02–89.49). There was no significant heterogeneity not for BP, FPG, or the remission rate of T2DM.
Publication Bias
Egger’s test and Begg’s test were performed based on HbA1c levels. A p value larger than 0.1 was considered to reflect a lack of publication bias; the two tests yielded p values of 0.19 and 0.15, respectively, indicating no publication bias.
Discussion
Since the beginning of the millennium, the application of metabolic surgery targeting T2DM has been increasing in China. The number of bariatric surgeries performed in 2010–2015 was 6937, with 4916 of them being for metabolic diseases [3]. Among all the bariatric surgery techniques, RYGB and SG were the most commonly performed in 2014, composing 85.5% of all such surgeries around the globe [18], these procedures have also been the most frequently performed in China over the last 5 years. At the same time, laparoscopic surgeries have almost completely replaced open procedures, accounting for 89.4% of surgeries, and the former have been found to be a cost-effective alternative to open surgery [19]. With the rapid introduction in bariatric surgery, the procedure has also been deployed for patients with T2DM. Most studies follow the criteria of BMI > 35 kg/m2 while considering the large number of patients with T2DM in China who have relatively lower BMI; there is a need for exploring the effect bariatric surgery on this group.
This meta-analysis was composed of the longest follow-up bariatric studies in Chinese patients with T2DM whose initial BMI were less than 35 kg/m2. This comprehensive analysis showed an encouraging outcome, with significant weight loss postoperatively and the remission rate of T2DM trending upward of at 12, 36, and 60 months (42.4%, 50.3%, 53.1%). The 5-year data showed a T2DM remission rate similar to the 54.5% rate reported by Ikramuddin et al. in a randomized controlled study [20] of patients whose BMI was controlled between 30 and 39.9 kg/m2. This could be an encouraging result as it indicates that even patients with BMI lower than 35 kg/m2 could still benefit from surgery, as the key mechanism of bariatric surgery is weight loss and this could assist satisfied glycemic control in patients with lower BMI. Moreover, significant decreases were observed in weight, BMI, BP, FPG, HbA1c, LDL, and TG at all visits, without large rebounds (Figs. 2 and 3), indicating a durable and effective impact of bariatric surgery and an improvement in co-occurring metabolic syndrome.
No consistent results demonstrated which surgical technique was superior [21], although RYGB is considered the gold standard procedure for the treatment of T2DM. Based on the recent increase in the use of SG for T2DM patients in different BMI groups, cumulative evidence has revealed propitious outcomes in terms of T2DM remission and control. In a recent meta-analysis comparing SG and RYGB in nonmorbidly obese T2DM patients, the results showed a comparative effect between the two procedures, and both improved patients’ quality of life [22]; however, BMI and WC were lower in the RYGB group than in the SG group, which was consistent with the findings of another meta-analysis by Yang et al. [17] and those of the SLEEVEPASS trial [23]. More detailed studies should be performed on the effects of SG effect in Chinese patients to confirm such results, as a recent long-term study showed a high rate of disease recurrence (75%) following SG in Taiwan [24]. In this study, the small number of studies in each research made it unfeasible to explore subgroup effect between RYGB and SG in Chinese group, which expects more study data for a subgroup study.
In 2015, Lee et al. created the “ABCD” scoring system (Table 8), which was used for the prediction of T2DM remission based on four preoperative variables: A, age; B, BMI; C, C-peptide; and D, duration of T2DM [25]. This system offers a brand-new perspective for surgeons in Asia to screen patients’ eligibility for metabolic surgery and to provide maximum personalization for an improved surgical outcome. In this meta-analysis, significantly reduced values were also detected in BMI, C-peptide, and T2DM duration at 12 months, which corresponds to most of the model. In addition, HbA1c was significantly different between the two groups. HbA1c is part of the DiaRem scoring system introduced by Still et al. [26]. Debate has arisen over the relative merits of different score systems [27, 28], and Kam et al. reported that DiaRem was more effective than ABCD in predicting T2DM outcomes in Chinese patients [29]. In the future, additional follow-up studies should include comparisons between remission and nonremission groups to verify the system in the long term and improve discriminatory power. Researchers should also note that at present, with the aid of artificial intelligence technology, a more precise prediction model could be built based upon a massive amount of data in the Chinese population and even the Asian population in general, which requires multicenter cooperation.
When addressing T2DM surgically, safety should always be taken into consideration. In the 11 studies included, 10 studies reported complications and mortality. The overall 30-day mortality rate ranges from 0.2 to 0.6%. Liang and Hsu reported two cases of death at 15 months and 5 years after surgery, one for gastric cancer. The incidence of mortality was 0.3% and no 30-day death case was reported. The general complications include bleeding, leakage, and stenosis of the anastomosis. Postoperative gastrointestinal bleeding (12 cases) were most frequently reported and all were managed successfully without surgery. Compared with other database studies and meta-analysis whose patients with BMI > 35 kg/m2, the major complication rate in this study is 2.4%, which is compatible [30,31,32,33]. However, more high-quality evidence is still warranted in studies of complications for patients whose BMI is lower than 35 kg/m2.
This meta-analysis has some limitations. First, studies with higher level evidence should be included in further meta-analyses as we included only nonrandomized studies. Other limiting factors included the small sample size, the lack of young patients, the variety of T2DM remission criteria among studies, and the mixed surgical techniques represented. Meanwhile, the evaluation of β-cell function was inadequate, for the lack of data reported in most studies. Moreover, heterogeneity existed in some aspects, which unavoidably limited the accuracy of this study. Finally, no weight regaining and T2DM reoccurrence cases were reported, which is an inevitable problem to consider in the long term.
In conclusion, bariatric surgery is effective on Chinese patients in terms of weight loss, BMI reduction, and metabolism control at 12, 36, and 60 months of follow-up. Bariatric surgery can be considered an optional treatment for patients with a BMI of less than 35 kg/m2, as the T2DM remission rate endures over time and is higher than the rate achieved with conservative medical treatment. Future study with high-level evidence and larger number of patients are necessary to better address effect of bariatric surgery in Chinese patients with BMI lower than 35 kg/m2. Results of bariatric surgery in American-born Chinese and/or Indian population with BMI lower than 35 kg/m2 could also be analyzed to reflect a Western lifestyle on innate Asian genetics via-a-vis metabolic syndrome.
References
Zheng Y, Ley SH, Hu FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol. Feb 2018;14(2):88–98. https://doi.org/10.1038/nrendo.2017.151.
Pories WJ, Swanson MS, MacDonald KG, et al. Who would have thought it? An operation proves to be the most effective therapy for adult-onset diabetes mellitus. Ann Surg. 1995;222(3):339–50; discussion 350-2. https://doi.org/10.1097/00000658-199509000-00011.
Du X, Dai R, Zhou HX, et al. Bariatric surgery in China: how is this new concept going? Obes Surg. 2016;26(12):2906–12. https://doi.org/10.1007/s11695-016-2204-2.
Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. Jama. 2004;292(14):1724–37. https://doi.org/10.1001/jama.292.14.1724.
Feng W, Yin T, Chu X, et al. Metabolic effects and safety of Roux-en-Y gastric bypass surgery vs. conventional medication in obese Chinese patients with type 2 diabetes. Diabetes Metab Res Rev. 2019;35(5):e3138. https://doi.org/10.1002/dmrr.3138.
Ji G, Li W, Li P, et al. Effect of Roux-en-Y gastric bypass for patients with type 2 diabetes mellitus and a BMI < 32.5 kg/m2: a 6-year study in Chinese patients. Obes Surg. 2020;30(7):2631–6. https://doi.org/10.1007/s11695-020-04534-z.
Ren Y, Yang W, Yang J, et al. Effect of Roux-en-Y gastric bypass with different pouch size in Chinese T2DM patients with BMI 30-35 kg/m2. Obes Surg. 2015;25(3):457–63. https://doi.org/10.1007/s11695-014-1411-y.
Wang C, Zhang H, Yu H, et al. Roux-en-Y gastric bypass for T2D treatment in Chinese patients with low BMI: 5-year outcomes. Obes Surg. 2020;30(7):2588–97. https://doi.org/10.1007/s11695-020-04520-5.
Wang L, Wang J, Jiang T. Effect of laparoscopic sleeve gastrectomy on type 2 diabetes mellitus in patients with body mass index less than 30 kg/m(2). Obes Surg. 2019;29(3):835–42. https://doi.org/10.1007/s11695-018-3602-4.
Yin J, Xu L, Mao Z, et al. Laparoscopic Roux-en-Y gastric bypass for type 2 diabetes mellitus in nonobese Chinese patients. Surg Laparosc Endosc Percutan Tech. 2014;24(6):e200–6. https://doi.org/10.1097/sle.0000000000000068.
Zhang H, Han X, Yu H, et al. Effect of Roux-en-Y gastric bypass on remission of T2D: medium-term follow-up in Chinese patients with different BMI obesity class. Obes Surg. 2017;27(1):134–42. https://doi.org/10.1007/s11695-016-2262-5.
Liang H, Guan W, Yang Y, et al. Roux-en-Y gastric bypass for Chinese type 2 diabetes mellitus patients with a BMI < 28 kg/m(2): a multi-institutional study. J Biomed Res. 2015;29(2):112–7. https://doi.org/10.7555/jbr.29.20140109.
Hsu CC, Almulaifi A, Chen JC, et al. Effect of bariatric surgery vs medical treatment on type 2 diabetes in patients with body mass index lower than 35: five-year outcomes. JAMA Surg. 2015;150(12):1117–24. https://doi.org/10.1001/jamasurg.2015.2602.
Huang CK, Tai CM, Chang PC, et al. Loop duodenojejunal bypass with sleeve gastrectomy: comparative study with Roux-en-Y gastric bypass in type 2 diabetic patients with a BMI <35 kg/m(2), first year results. Obes Surg. 2016;26(10):2291–301. https://doi.org/10.1007/s11695-016-2118-z.
Lee WJ, Almulaifi A, Chong K, et al. The effect and predictive score of gastric bypass and sleeve gastrectomy on type 2 diabetes mellitus patients with BMI < 30 kg/m(2). Obes Surg. 2015;25(10):1772–8. https://doi.org/10.1007/s11695-015-1603-0.
Buse JB, Caprio S, Cefalu WT, et al. How do we define cure of diabetes? Diabetes Care. 2009;32(11):2133–5. https://doi.org/10.2337/dc09-9036.
Yang P, Chen B, Xiang S, et al. Long-term outcomes of laparoscopic sleeve gastrectomy versus Roux-en-Y gastric bypass for morbid obesity: results from a meta-analysis of randomized controlled trials. Surg Obes Relat Dis. 2019;15(4):546–55. https://doi.org/10.1016/j.soard.2019.02.001.
Angrisani L, Santonicola A, Iovino P, et al. Bariatric surgery and endoluminal procedures: IFSO Worldwide Survey 2014. Obes Surg. 2017;27(9):2279–89. https://doi.org/10.1007/s11695-017-2666-x.
Paxton JH, Matthews JB. The cost effectiveness of laparoscopic versus open gastric bypass surgery. Obes Surg. 2005;15(1):24–34. https://doi.org/10.1381/0960892052993477.
Ikramuddin S, Korner J, Lee WJ, et al. Lifestyle intervention and medical management with vs without Roux-en-Y gastric bypass and control of hemoglobin A1c, LDL cholesterol, and systolic blood pressure at 5 years in the diabetes surgery study. Jama. 2018;319(3):266–78. https://doi.org/10.1001/jama.2017.20813.
Colquitt JL, Pickett K, Loveman E, et al. Surgery for weight loss in adults. Cochrane Database Syst Rev. 2014;8:Cd003641. https://doi.org/10.1002/14651858.CD003641.pub4.
Sha Y, Huang X, Ke P, et al. Laparoscopic Roux-en-Y gastric bypass versus sleeve gastrectomy for type 2 diabetes mellitus in nonseverely obese patients: a systematic review and meta-analysis of randomized controlled trials. Obes Surg. 2020;30(5):1660–70. https://doi.org/10.1007/s11695-019-04378-2.
Salminen P, Helmiö M, Ovaska J, et al. Effect of laparoscopic sleeve gastrectomy vs laparoscopic Roux-en-Y gastric bypass on weight loss at 5 years among patients with morbid obesity: the SLEEVEPASS randomized clinical trial. Jama. 2018;319(3):241–54. https://doi.org/10.1001/jama.2017.20313.
Lee MH, Almalki OM, Lee WJ, et al. Laparoscopic sleeve gastrectomy for type 2 diabetes mellitus: long-term result and recurrence of diabetes. Obes Surg. 2020; https://doi.org/10.1007/s11695-020-04737-4.
Lee WJ, Almulaifi A. Recent advances in bariatric/metabolic surgery: appraisal of clinical evidence. J Biomed Res. 2015;29(2):98–104. https://doi.org/10.7555/jbr.28.20140120.
Still CD, Wood GC, Benotti P, et al. Preoperative prediction of type 2 diabetes remission after Roux-en-Y gastric bypass surgery: a retrospective cohort study. Lancet Diabetes Endocrinol. 2014;2(1):38–45. https://doi.org/10.1016/s2213-8587(13)70070-6.
Lee WJ, Chong K, Chen SC, et al. Preoperative prediction of type 2 diabetes remission after gastric bypass surgery: a comparison of DiaRem scores and ABCD scores. Obes Surg. 2016;26(10):2418–24. https://doi.org/10.1007/s11695-016-2120-5.
Debédat J, Sokolovska N, Coupaye M, et al. Long-term relapse of type 2 diabetes after Roux-en-Y gastric bypass: prediction and clinical relevance. Diabetes Care. 2018;41(10):2086–95. https://doi.org/10.2337/dc18-0567.
Kam H, Tu Y, Pan J, et al. Comparison of four risk prediction models for diabetes remission after Roux-en-Y gastric bypass surgery in obese Chinese patients with type 2 diabetes mellitus. Obes Surg. 2020;30(6):2147–57. https://doi.org/10.1007/s11695-019-04371-9.
Sjöström L, Lindroos AK, Peltonen M, et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 2004;351(26):2683–93. https://doi.org/10.1056/NEJMoa035622.
Małczak P, Pisarska M, Piotr M, et al. Enhanced recovery after bariatric surgery: systematic review and meta-analysis. Obes Surg. 2017;27(1):226–35. https://doi.org/10.1007/s11695-016-2438-z.
Wolter S, Duprée A, ElGammal A, et al. Mentorship programs in bariatric surgery reduce perioperative complication rate at equal short-term outcome-results from the OPTIMIZE trial. Obes Surg. 2019;29(1):127–36. https://doi.org/10.1007/s11695-018-3495-2.
Chang SH, Freeman NLB, Lee JA, et al. Early major complications after bariatric surgery in the USA, 2003-2014: a systematic review and meta-analysis. Obes Rev. 2018;19(4):529–37. https://doi.org/10.1111/obr.12647.
Acknowledgements
The authors appreciated Mengchu Xu, from Fudan University, for providing full access to the comprehensive database search.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, Y., Gu, Y., Jin, Y. et al. Is Bariatric Surgery Effective for Chinese Patients with Type 2 Diabetes Mellitus and Body Mass Index < 35 kg/m2? A Systematic Review and Meta-analysis. OBES SURG 31, 4083–4092 (2021). https://doi.org/10.1007/s11695-021-05520-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-021-05520-9