Abstract
Purpose
Compared to medical treatment, bariatric surgery reduces long-term mortality in persons with obesity. Some studies indicate that the effect only applies to patients above median age of cohorts, not to younger patients. Our objective was to assess the role of age in the reduction of mortality (global mortality and mortality for specific causes) through bariatric surgery.
Materials and Methods
Data sources: PubMed, Cochrane Library, MEDLINE, and Embase. Study selection: studies reporting mortality in relation to median age of patients. Data extraction and synthesis: pooled random effects of estimates of the risk of mortality in participants undergoing surgery compared with controls, as function of median age.
Results
Mortality was lower in patients undergoing surgery than in controls (OR = 0.29, 95% CI 0.17–0.49). Below median age, the difference between surgery patients and controls was nonsignificant (OR = 0.78, 95% CI 0.57–1.06). Above median age, the difference was significant (OR = 0.23, 95% CI 0.12–0.44). In a subset of 5 studies, deaths due to various causes were less, and external causes-related deaths were more frequent in surgery than in controls. Below median age, deaths due to CVD were less frequent in surgery than in controls. Above median age, total deaths and deaths due to various causes (cardiovascular, diabetes, cancer, and other causes) were less in surgery than in controls. Publication bias was absent.
Conclusion
Compared with controls, bariatric surgery reduces long-term global mortality only above median age, not below median age. Also mortality due to specific causes is mainly reduced in persons above median age.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Persons with obesity have a longer life expectancy undergoing bariatric surgery (BS) than receiving medical treatment of obesity. This applies to both diabetic and nondiabetic persons and has been shown in several papers [1,2,3,4,5,6,7,8], analyzed in two meta-analyses [9, 10], and confirmed later [11,12,13,14,15,16,17,18,19,20], with one noticeable exception [16]. Also, BS improves quality of life in morbid obesity [21], is associated with lower development of medical complications of obesity, with reduced frequency of comorbidities, reduced necessity of several kinds of drugs, improved cardiovascular (CV) risk profile [22,23,24,25,26,27,28], and is cost-effective in the management of obesity [29, 30]. Studies have been performed through restrictive or mixed techniques [gastric banding (LAGB), vertical banded gastroplasty (VGB), Roux-en-Y gastric bypass (RYGB), sleeve gastrectomy (LSG)] and through malabsorptive surgery [biliointestinal bypass (BIBP) and biliopancreatic diversion (BPD)].
Age is probably important for BS under several aspects, but has not been fully explored so far. BS is commonly performed in severely obese middle-aged patients, with a median age of 39 years (16–64 years) [21]; data on BS have been published in obese adolescents, with positive results, so that it has been proposed that “bariatric surgery in severely obese adolescents should be considered standard of care” [31], but figures on numbers of interventions performed in the USA are overall small [32].
A few studies indicate that the preventive effect of BS on mortality is significant for patients above median age of cohorts (i.e., aged > 42 or > 43 or > 44 years in different cohorts), but not for younger patients: this has been shown for LAGB, RYGB, BPD, and BIBP [5, 11, 12, 15]; in the Swedish Obesity Study (SOS), subjects aged < 37 years were excluded a priori [4]. Given the epidemics of obesity and the increasing number of BS interventions, these data are probably of great importance, but have not been formally analyzed. The basic question is if BS should be performed as soon as possible to prevent obesity-related morbidity and mortality or if should be performed later in the life age of patients, when health conditions have deteriorated. The aim of this study is to analyze systemically the effect of age on the preventive effect of BS against mortality, be it global mortality and mortality due to specific causes.
Material and Methods
Strategy
Eligible controlled clinical studies (CCS) were those comparing bariatric surgery versus no-surgery in persons with morbid obesity, irrespective of publication status or language. Measures of treatment efficacy were as follows: (1) global mortality, (2) cardiovascular (CV) cause-related mortality, (3) cancer-related mortality, (4) diabetes-related mortality, (5) mortality due to external causes (i.e., accidents, suicide, poisoning, complications of surgery), and (6) other causes-related mortality (i.e., mortality not due to the above causes, be it infection, gastroenterological or respiratory diseases, etc.).
Retrieval of CCSs was based on the PubMed, Cochrane Library, MEDLINE, and Embase (until October 2019) using the terms obesity, bariatric surgery, mortality, long-term mortality, survival, age, young, old, elderly, efficacy, complications, mortality, gastric bypass, gastric banding, biliopancreatic diversion, biliointestinal bypass, sleeve gastrectomy, and associations of two or more of the above terms and limiting the search to controlled clinical human studies. A manual search was also performed using the reference lists from articles, reviews, editorials, and the proceedings of international congresses. When the results of a single study were reported in more than 1 publication, only the most recent and complete data were included in the meta-analysis; an exception was applied to papers [11 and 18], with the same database, but with median observation periods of 13 and of 17 years, respectively; to keep variance of observation periods at a minimum, the former study was considered for the meta-analyses, and simulations were performed in which the latter study was considered. Decisions on which trials to include were taken unblindly by the three authors; the SOS study [4] was not considered since subjects aged < 37 years were excluded. Disagreements were resolved by discussion.
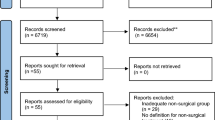
Figure 1 reports the flowchart of clinical studies included in the systematic review and meta-analysis: non-relevant papers were excluded first, together with reviews and meta-analyses; studies without measures of dispersion of data were excluded at a second step, as well as studies without comparisons between surgery and control patients. Twenty-two papers reporting comparisons between surgery and standard medical treatment on long-term (i.e., greater than 1 year follow-up) mortality were identified as of possible interest. Then, as only five papers reported differences in mortality between young and aged patients [5, 11, 12, 15, 17, 18], an invitation was sent to a total of twenty-two authors requesting to provide data about outcomes (number and cause of death) divided according to median age of their cohorts. As a result, nine papers, all published as full reports, could be meta-analyzed with reference to item 1 (number of deaths) and five with reference to item 2 (causes of death) (Tables 1 and 2). The Appendix (Supplemental Table 1) indicates papers considered of interest for which details were requested to authors and the answers received. Data concerning deaths were derived as such from all studies. Appropriate methodology according to the preferred reporting items for systematic reviews and meta-analyses (PRISMA) statement [33] was adhered to. The methodologic quality of the trials was assessed by criteria previously validated [34] (Appendix, Supplemental Table 2). Each quality component was rated for bias as high risk, unclear, or low risk. Data concerning trials, patient characteristics, and treatment outcome (Table 1) were abstracted by the three authors, and discrepancies were resolved by discussion.
Flowchart of clinical studies included in the systematic review and meta-analysis: non-relevant papers were excluded first, together with reviews and meta-analyses; studies without measures of dispersion of data were excluded at a third step, as well as studies without comparisons between surgery and control patients
Statistical Analysis
Treatment outcomes were expressed as odds ratios (ORs) with 95% confidence intervals (CIs) and pooled into an overall OR using a random effects model according to DerSimonian and Laird [35]. The I2 index for heterogeneity was calculated for each assessed outcome, and potential sources of heterogeneity were discussed where appropriate. A p < 0.05 was considered indicative of statistically significant heterogeneity.
To explore the potential effect of several patients or trial characteristics on the pooled estimates, a meta-regression analysis was performed for all outcomes showing a statistically significant heterogeneity. The dependent variable was the observed mortality rate from each study for the outcome of interest. The role of each covariate in heterogeneity was expressed by Wald test estimated by the meta-regression. The following covariates were included in the meta-regression analysis, including covariates previously shown to be associated with survival in patients, age, sex proportion, presence of diabetes, presence of CV diseases (CVD), BMI of each study (weighted medians of surgery and control participants), year of starting of study (as a proxy for technical improvement with time), number of patients enrolled, kind of controls (clinics vs community patients), duration of follow-up, and efficacy of treatment (vs controls) in each study.
Sensitivity Analyses
The first sensitivity analysis assessed the role of age itself on global mortality and on different causes of mortality in BS and in control patients considered separately. The second sensitivity analysis assessed results with different surgical techniques. In a further analysis, we evaluated the existence of a potential publication bias, defined as the tendency of authors and editors to handle studies in which the experimental results achieved statistical significance more favorably than in studies in which the results failed to reach significance, which would ultimately introduce bias into the overall published literature [36]. Funnel plot asymmetry was evaluated by using Egger’s test for small study effects through the metabias routine [36]. All statistical analyses were performed by STATA 12 for Macintosh (Stata Corporation, College Station, TX). The protocol of the meta-analysis has been registered (Prospero).
Results
The total sample size was 607,643 persons (surgery = 72,267, controls = 535,376); the total number of deaths was 79,134 (surgery deaths = 2274, control deaths = 76,860), and duration of follow-up was 8.7 ± 3.27 years (median ± SD). Mortality was significantly lower in persons undergoing BS than in persons receiving standard medical treatment of obesity (Fig. 2a). Below median age, sample size was 301,787 (surgery = 35,627, controls = 266,160), and the total number of deaths was 7416 (surgery deaths 721, control deaths = 6695). The difference between BS persons and controls was nonsignificant in persons below mean age (Fig. 2b). Above mean age, sample size was 302,771 (surgery = 35,674, controls = 267,097), and the total number of deaths was 71,718 (surgery deaths = 1553, control deaths = 70,165). The difference between BS persons and controls was highly significant above mean age (Fig. 2c). In addition, both in BS persons and in controls, mortality was higher above than below median age (Appendix Supplemental Fig. 1). All the above comparisons were based on nine studies. Median age, frequency of diabetes and of cardiovascular diseases, and sex ratio of BS persons and controls were not different (Table 1); also body mass index was similar in BS persons and in controls (not shown).
Forest plots of meta-analysis of global mortality in surgery vs no-surgery patients (a to c). OR indicates odds ratio (with 95% CI). I2 indicates heterogeneity. Egger’s test for small-study effects is shown in each panel. a Studies without dividing for age. b Studies considering patients below mean age. c Studies considering patients above mean age
In a subset of five studies, causes of death were compared (in BS and in controls) with no division for median age; deaths from CVD, from cancer, and from diabetes were significantly fewer in BS than in controls, while deaths from external causes were higher in BS than in controls (Table 3). In both BS persons and controls considered separately, age-related mortality due to CVD causes, cancer, diabetes, and to other causes in controls was lower below median age than above median age (Supplemental Table 3).
When these five studies were analyzed for median age, only deaths from CVD causes were significantly fewer in BS than in controls below median age (Table 3). Above median age, global deaths and deaths from CVD, cancer, diabetes, and other diseases were significantly fewer in BS than in controls (Table 3, Supplemental Fig. 4). In summary, most of the significance of difference in mortality between surgery and controls came from persons above median age.
All studies were replicated with substitution of study 11 by study 18; results of meta-analyses and meta-regressions did not change (not shown). Heterogeneity was elevated in several meta-analyses, both for non-age-dependent meta-analyses (global mortality, mortality due to CVD causes, and diabetes) and for below median age meta-analyses (global mortality) and for above median age meta-analyses (global mortality, mortality due to CVD causes, due to cancer, due to diabetes) (Table 3). At meta-regression, global mortality was associated with the size of studies, with mortality due to CVD causes, to cancer, and to diabetes in both surgery and in controls, and mortality due to external causes in controls (Supplemental Fig. 3). Publication bias was absent (Table 3).
The first sensitivity analysis showed that in both BS persons and in controls, mortality was lower below than above median age (Supplemental Table 3, Supplemental Fig. 1). However, the ORs and their 95% CI were different in BS persons (OR = 0.46, 95% CI 0.33–0.64) and in controls (OR = 0.16, 95% CI 0.09–0.29), with no overlapping.
Sensitivity analysis evaluating outcomes after different surgical techniques was only possible for global mortality and for causes-related deaths in all patients and showed no statistically different outcome; no other comparison could be performed, as only one study with LAGB (vs 4 with RYGB or BPD or with more techniques together) analyzed causes of death.
Discussion
In this meta-analysis, global mortality as well as mortality due to CVD causes, cancer, and diabetes was reduced in BS persons compared with controls. In addition, mortality due to age was also lower below than above median age in both BS persons and in controls, considered separately. Other studies reporting mortality in BS persons and in controls were not considered since they were without data on age-dependent mortality, but these data are not different from the majority of other studies [9, 10].
This meta-analysis shows that reduced mortality of BS (global mortality, mortality due to CVD, cancer, diabetes, and other causes) mainly applies to persons above median age, not below median. One exception is represented by mortality due to CVD, reduced in BS persons also below median age.
The finding that only above a given age bariatric surgery is more effective in reducing long-term mortality in comparison with controls (coming from registries and from communities or receiving standard medical treatment) has been reported (3–5) but has never been analyzed systemically. As a matter of fact, in the SOS study, persons aged < 37 years were not included [4]. This is probably linked to a low overall mortality of younger persons; this is supported by our data showing a significant increase of mortality, global and for several causes, linked to aging, in both BS persons and controls, and by older data [37]. In particular, when comparing mortality below and above median age, the ORs and their 95% CI were different in BS persons and in controls, with no overlapping.
Additional causes for the nonsignificant effect of BS in persons below median age might be represented by deaths due to external causes, more frequent in BS persons than in controls. One confounding factor might be the increased rate of suicides reported in some studies [13, 16, 17] in persons undergoing bariatric surgery; unfortunately these data could not be meta-analyzed due to the lack of specific information about age of suicide. Methodologic quality of studies was good, even though all studies were retrospective, as it usually happens with studies dealing with bariatric surgery [34]. Publication bias was absent [36].
Limitations
The first limitation is that only nine papers could be analyzed out of many others that could not be included, although data suggesting our conclusions have been published years ago [2,3,4]. Duration of follow-up might be considered short, but data were confirmed when the follow-up period was extended [11, 18]. This meta-analysis only reflects reduction of mortality through bariatric surgery, and does not reflect other possible beneficial effects of bariatric surgery such as reduction or prevention of comorbidities or improvement of quality of life. At present, there is no data on the effect of age on prevention/reduction of comorbidities in persons with obesity or on the effect of age on improvement of quality of life. Future studies might explore these items, but in our experience [38], age does not affect prevention of comorbidities such as diabetes, CV diseases, and cancer; in addition, prolonged observations are accompanied by a progressive increase of efficacy of BS in prevention of comorbidities [18], and the preventive effect of bariatric surgery on incident diabetes is greater in younger subjects [39]. Furthermore, due to the typical nature of meta-analysis, we could only analyze aggregated data so that we could not test interaction between BS and age or implement a global multivariable model for the difference between BS persons and controls, adjusting for age and other possible confounders. Heterogeneity was high for many comparisons, probably due, among other causes, to the great difference in size of the studies analyzed; in addition, it is possibly due also to the different nature of controls. Future randomized clinical trials of long duration are required to confirm the data of this meta-analysis.
Conclusion
Bariatric surgery seems to prevent long-term mortality, compared with controls, only in persons above median age of cohorts, not in younger persons, and a similar effect applies to most causes of mortality. This effect is probably due to a low overall mortality of persons with obesity of young ages. In contrast, no conclusion can be drawn for the effect of age on prevention or disappearance of comorbidities through bariatric surgery.
References
MacDonald Jr KG, Long SD, Swanson MS, et al. The gastric bypass operation reduces the progression and mortality of non-insulin-dependent diabetes mellitus. J Gastrointest Surg. 1997;1:213–20.
Christou NV, Sampalis JS, Liberman M, et al. Surgery decreases long-term mortality, morbidity, and health care use in morbidly obese patients. Ann Surg. 2004;240:416–23.
Flum DR, Dellinger EP. Impact of gastric bypass operation on survival: a population-based analysis. J Am Coll Surg. 2004;199:543–51.
Sjöström L, Narbro K, Sjöström CD, et al. Swedish obese subjects study. Swedish obese patients study. Effects of bariatric surgery on mortality in Swedish obese patients. N Engl J Med. 2007;357:741–52.
Busetto L, Mirabelli D, Petroni ML, et al. Comparative long-term mortality after laparoscopic adjustable gastric banding versus nonsurgical controls. Surg Obes Relat Dis. 2007;3:496–502.
Adams TD, Gress RE, Smith SC, et al. Long-term-mortality after gastric bypass surgery. N Engl J Med. 2007;357:753–61.
Sowemimo OA, Yood SM, Courtney J, et al. Natural history of morbid obesity without surgical intervention. Surg Obes Relat Dis. 2007;3:73–7.
Peeters A, O'Brien PE, Laurie C, et al. Substantial intentional weight loss and mortality in the severely obese. Ann Surg. 2007;246:1028–33.
Pontiroli AE, Morabito A. Long-term prevention of mortality in morbid obesity through bariatric surgery. A systematic review and meta-analysis of trials performed with gastric banding and gastric bypass. Ann Surg. 2011;253:484–7.
Cardoso L, Rodrigues D, Gomes L, et al. Short- and long-term mortality after bariatric surgery: a systematic review and meta-analysis. Diabetes Obes Metab. 2017;19:1223–32.
Pontiroli AE, Zakaria AS, Mantegazza E, Morabito A, Saibene A, Mozzi E, Micheletto G; LAGB10 working group. Long-term mortality and incidence of cardiovascular diseases and type 2 diabetes in diabetic and nondiabetic obese patients undergoing gastric banding: a controlled study. Cardiovasc Diabetol 2016; 15: 39.
Davidson LE, Adams TD, Kim J, et al. Association of patient age at gastric bypass surgery with long-term all-cause and cause-specific mortality. JAMA Surg. 2016;151:631–7.
Lent MR, Benotti PN, Mirshahi T, et al. All-cause and specific-cause mortality risk after Roux-en-Y gastric bypass in patients with and without diabetes. Diabetes Care. 2017;40:1379–85.
Reges O, Greenland P, Dicker D, et al. Association of bariatric surgery using laparoscopic banding, Roux-en-Y gastric bypass, or laparoscopic sleeve gastrectomy vs usual care obesity management with all-cause mortality. JAMA. 2018;319:279–90.
Ceriani V, Sarro G, Micheletto G, Giovanelli A, Zakaria AS, Fanchini M, Osio C, Nosari I, Morabito A, Pontiroli AE; LAGB10 working group. Long-term mortality in obese subjects undergoing malabsorptive surgery (biliopancreatic diversion and biliointestinal bypass) versus medical treatment. Int J Obes 2019; 43:1147–1153.
Gribsholt SB, Thomsen RW, Svensson E, et al. Overall and cause-specific mortality after Roux-en-Y gastric bypass surgery: a nationwide cohort study. Surg Obes Relat Dis. 2017;13:581–7.
Kauppila JH, Tao W, Santoni G, et al. Effects of obesity surgery on overall and disease-specific mortality in a 5-country population-based study. Gastroenterology. 2019;157:119–27.
Pontiroli AE, Zakaria AS, Fanchini M, et al. A 23-year study of mortality and development of co-morbidities in patients with obesity undergoing bariatric surgery (laparoscopic gastric banding) in comparison with medical treatment of obesity. Cardiovasc Diabetol. 2018;17:161.
Moussa OM, Erridge S, Chidambaram S, et al. Mortality of the severely obese: a population study. Ann Surg. 2019;269:1087–91.
Aminian A, Zajichek A, Arterburn DE, et al. Association of metabolic surgery with major adverse cardiovascular outcomes in patients with type 2 diabetes and obesity. JAMA. 2019; https://doi.org/10.1001/jama.2019.14231. [Epub ahead of print]
Raaijmakers LC, Pouwels S, Thomassen SE, et al. Quality of life and bariatric surgery: a systematic review of short- and long-term results and comparison with community norms. Eur J Clin Nutr. 2017;71:441–9.
Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA. 2004;292:1724–37.
Buchwald H, Estok R, Fahrbach K, et al. Weight and type 2 diabetes after bariatric surgery: systematic review and meta-analysis. Am J Med. 2009;122:248–56.
Heneghan HM, Meron-Eldar S, Brethauer SA, et al. Effect of bariatric surgery on cardio-vascular risk profile. Am J Cardiol. 2011;108:1499–507.
Romeo S, Maglio C, Burza MA, et al. Cardiovascular events after bariatric surgery in obese patients with type 2 diabetes. Diabetes Care. 2012;35:2613–7.
Johnson BL, Blackhurst DW, Latham BB, et al. Bariatric surgery is associated with a reduction in major macrovascular and microvascular complications in moderately to severely obese patients with type 2 diabetes mellitus. J Am Coll Surg. 2013;216:545–56.
Busetto L, De Stefano F, Pigozzo S, et al. Long-term cardiovascular risk and coronary events in morbidly obese patients treated with laparoscopic gastric banding. Surg Obes Relat Dis. 2014;10:112–20.
Chang SH, Stoll CR, Song J, et al. The effectiveness and risks of bariatric surgery: an updated systematic review and meta-analysis, 2003–2012. JAMA Surg. 2014;149:275–87.
Picot J, Jones J, Colquitt JL, et al. The clinical effectiveness and cost-effectiveness of bariatric (weight loss) surgery for obesity: a systematic review and economic evaluation. Health Technol Assess. 2009;13:1–190.
Keating CL, Dixon JB, Moodie ML, et al. Cost-effectiveness of surgically induced weight loss for the management of type 2 diabetes: modeled lifetime analysis. Diabetes Care. 2009;32:567–74.
Pratt JSA, Browne A, Browne NT, et al. ASMBS pediatric metabolic and bariatric surgery guidelines, 2018. Surg Obes Relat Dis. 2018;14:882–901.
Campoverde Reyes KJ, Misra M, Lee H, et al. Weight loss surgery utilization in patients aged 14-25 with severe obesity among several healthcare institutions in the United States. Front Pediatr. 2018;6:251.
Moher DA, Tetzlaff J, Altman DG, PRISMA group preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement PLoS Med 2009; 6: e1000097
Kim SY, Park JE, Lee YJ, et al. Testing a tool for assessing the risk of bias for nonrandomized studies showed moderate reliability and promising validity. J Clin Epidemiol. 2013;66:408–14.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–80.
Easterbrook PJ, Berlin JA, Gopalan R, et al. Publication bias in clinical research. Lancet. 1991;337:867–72.
Stevens J, Cai J, Pamuk ER, et al. The effect of age on the association between body-mass index and mortality. N Engl J Med. 1998;338:1–7.
Pontiroli AE, Ceriani V, Tagliabue E, et al. Bariatric surgery, compared to medical treatment, reduces morbidity but does not reduce mortality in patients aged < 43 years, especially if diabetes mellitus is present. A post-hoc analysis of two retrospective cohort studies. Acta Diabetol. 2019 Oct 9; https://doi.org/10.1007/s00592-019-01433-3. [Epub ahead of print]
Merlotti C, Morabito A, Pontiroli AE. Prevention of type 2 diabetes; a systematic review and meta-analysis of different intervention strategies. Diabetes Obes Metab. 2014;16:719–27.
Acknowledgments
The authors wish to thank the following persons, who went through their databases and further examined their databases to answer our request to yield details about age-related mortality: Sigrid B. Gribsholt, Department of Clinical Epidemiology, Aarhus University, Denmark; Steven C. Hunt, Department of Genetic Medicine, Weill Cornell Medicine, Doha, Qatar; G. Craig Wood, Obesity Institute, 100 N. Academy Ave, Danville, PA, USA; Luca Busetto, Dipartimento di Medicina Interna, Università di Padova, Padova, Italy; Valerio Ceriani, IRCCS MultiMedica, Milan, Italy; Antonio E. Pontiroli, Dipartimento di Scienze della Salute, Università di Milano, Milan, Italy; Orna Reges, Clalit Research Institute, Clalit Health Services, Tel Aviv, Israel; Department of Molecular Medicine and Surgery, Karolinska Institutet, Karolinska University Hospital, Stockholm, Sweden; and Medical Research Center Oulu, University of Oulu and Oulu University Hospital, Oulu, Finland. Without their help, this systematic review and meta-analysis could not have been completed.
Funding
This work was supported by IRCCS MultiMedica, by Università degli Studi di Milano, and by a Grant “Ricerca Corrente” to IRCCS Istituto MultiMedica from Ministero della Salute (Italian Ministry of Health), Italy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
All authors declare that they have no conflict of interest.
Ethical Approval Statement
This manuscript does not contain studies with human participants or animals performed by any of the authors.
Informed Consent Statement
Informed consent does not apply to this manuscript
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 1075 kb)
Rights and permissions
About this article
Cite this article
Pontiroli, A.E., Ceriani, V. & Tagliabue, E. Compared with Controls, Bariatric Surgery Prevents Long-Term Mortality in Persons with Obesity Only Above Median Age of Cohorts: a Systematic Review and Meta-Analysis. OBES SURG 30, 2487–2496 (2020). https://doi.org/10.1007/s11695-020-04530-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-020-04530-3